Abstract
Purpose
Dedicated breast positron emission tomography (DbPET) provides detailed high-resolution images and can detect intratumoral heterogeneity using 18F-fluorodeoxyglucose (FDG). We aimed to evaluate the correlation between FDG uptake on DbPET and the clinicopathological features of breast cancer, particularly those with an intratumoral heterogeneous distribution of FDG on DbPET.
Methods
We evaluated 195 consecutive patients with invasive breast cancer who underwent preoperative whole-body PET (WBPET) and DbPET concurrently between January 2016 and March 2017. The relationships between clinicopathological factors and the maximum standard uptake values (SUVmax) of DbPET and WBPET, including clinical stage, nuclear grade, Ki67 proliferation index, estrogen receptor (ER) and human epidermal growth factor receptor type 2 (HER2) statuses, and the intratumoral heterogeneous distribution of FDG on DbPET, were evaluated.
Results
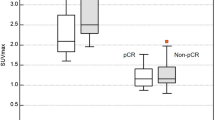
The SUVmax of DbPET was significantly correlated with clinical T stage, N stage, nuclear grade, and Ki67 proliferation index (all p < 0.001) as well as the ER (p = 0.006) and HER2 (p = 0.040) statuses. Intratumoral heterogeneous distribution of FDG on DbPET was significantly related with high nuclear grade (p = 0.016) and high Ki67 proliferation index (p = 0.015) but not with clinical T stage, N stage, and ER and HER2 statuses.
Conclusions
The SUVmax of DbPET correlates with clinicopathological factors and also WBPET does. In addition, intratumoral heterogeneity on DbPET provides predictive value for malignancy grade and could inform therapeutic decisions.



Similar content being viewed by others
Abbreviations
- ER:
-
Estrogen receptor
- DbPET:
-
Dedicated breast positron emission tomography
- FDG:
-
18F-fluorodeoxyglucose
- HER2:
-
Human epidermal growth factor receptor type 2
- WBPET:
-
Whole-body positron emission tomography
- PET/CT:
-
Positron emission tomography/computed tomography
- SUVmax:
-
Maximum standardized uptake value
References
Vansteenkiste JF, Stroobants SG, Dupont PJ, De Leyn PR, Verbeken EK, Deneffe GJ et al (1999) Prognostic importance of the standardized uptake value on (18)F-fluoro-2-deoxy-glucosepositron emission tomography scan in non-small-cell lung cancer: an analysis of 125 cases. Leuven Lung Cancer Group. J Clin Oncol 17:3201–3206
Okazumi S, Isono K, Enomoto K, Kikuchi T, Ozaki M, Yamamoto H et al (1992) Evaluation of liver tumors using fluorine-18-fluorodeoxyglucose PET: characterization of tumor and assessment of effect of treatment. J Nucl Med 33:333–339
Fuster D, Duch J, Paredes P, Velasco M, Mun˜oz M, Santamaria G et al (2008) Preoperative staging of large primary breast cancer with [18F]fluorodeoxyglucose positron emission tomography/computed tomography compared with conventional imaging procedures. J Clin Oncol 26:4746–4751
Groheux D, Hindie E, Delord M, Giacchetti S, Hamy AS, de Bazelaire C et al (2012) Prognostic impact of (18)FDG-PET-CT findings in clinical stage III and IIB breast cancer. J Natl Cancer Inst 104:1879–1887
Okada J, Yoshikawa K, Itami M, Imaseki K, Uno K, Itami J et al (1992) Positron emission tomography using fluorine-18-fluorodeoxyglucose in malignant lymphoma: a comparison with proliferative activity. J Nucl Med 33:325–329
Groheux D, Hindie´ E, Giacchetti S, Hamy AS, Berger F, Merlet P et al (2014) Early assessment with 18F-fluorodeoxyglucose positron emission tomography/computed tomography can help predict the outcome of neoadjuvant chemotherapy in triple negative breast cancer. Eur J Cancer 50:1864–1871
Berriolo-Riedinger A, Touzery C, Riedinger JM, Toubeau M, Coudert B, Arnould L et al (2007) [18F]FDG-PET predicts complete pathological response of breast cancer to neoadjuvant chemotherapy. Eur J Nucl Med Mol Imaging 34:1915–1924
Humbert O, Berriolo-Riedinger A, Riedinger JM, Coudert B, Arnould L, Cochet A et al (2012) Changes in 18F-FDG tumor metabolism after a first course of neoadjuvant chemotherapy in breast cancer: influence of tumor subtypes. Ann Oncol 23:2572–2577
Buck A, Schirrmeister H, Kuhn T, Shen C, Kalker T, Kotzerke J et al (2002) FDG uptake in breast cancer: correlation with biological and clinical prognostic parameters. Eur J Nucl Med Mol Imaging 29:1317–1323
Smith TA, Sharma RI, Thompson AM, Paulin FE (2006) Tumor 18F-FDG incorporation is enhanced by attenuation of P53 function in breast cancer cells in vitro. J Nucl Med 47:1525–1530
Dehdashti F, Mortimer JE, Siegel BA, Griffeth LK, Bonasera TJ, Fusselman MJ et al (1995) Positron tomographic assessment of estrogen receptors in breast cancer: comparison with FDG-PET and in vitro receptor assays. J Nucl Med 36:1766–1774
Crippa F, Seregni E, Agresti R, Chiesa C, Pascali C, Bogni A et al (1998) Association between [18F]fluorodeoxyglucose uptake and postoperative histopathology, hormone receptor status, thymidine labelling index and p53 in primary breast cancer: a preliminary observation. Eur J Nucl Med 25:1429–1434
Avril N, Menzel M, Dose J, Schelling M, Weber W, Ja¨nicke F et al (2001) Glucose metabolism of breast cancer assessed by 18F-FDG PET: histologic and immunohistochemical tissue analysis. J Nucl Med 42:9–16
Bos R, van Der Hoeven JJ, van Der Wall E, van Der Groep P, van Diest PJ, Comans EF et al (2002) Biologic correlates of [18]fluorodeoxyglucose uptake in human breast cancer measured by positron emission tomography. J Clin Oncol 20:379–387
Kumar R, Chauhan A, Zhuang H, Chandra P, Schnall M, Alavi A (2006) Clinicopathologic factors associated with false negative FDG-PET in primary breast cancer. Breast Cancer Res Treat 98:267–274
Groheux D, Giacchetti S, Moretti JL, Porcher R, Espie´ M, Lehmann- Che J et al (2011) Correlation of high 18F-FDG uptake to clinical, pathological and biological prognostic factors in breast cancer. Eur J Nucl Med Mol Imaging 38:426–435
Fasanella S, Leonardi E, Cantaloni C, Eccher C, Bazzanella I, Aldovini D et al (2011) Proliferative activity in human breast cancer: Ki-67 automated evaluation and the influence of different Ki-67 equivalent antibodies. Diagn Pathol 6:S7
Tchou J, Sonnad SS, Bergey MR, Basu S, Tomaszewski J, Alavi A et al (2010) Degree of tumor FDG uptake correlates with proliferation index in triple negative breast cancer. Mol Imaging Biol 12:657–662
Oshida M, Uno K, Suzuki M, Nagashima T, Hashimoto H, Yagata H et al (1998) Predicting the prognoses of breast carcinoma patients with positron emission tomography using 2-deoxy-2-fluoro[18F]-D-glucose. Cancer 82:2227–2234
Na F, Wang J, Li C, Deng L, Xue J, Lu Y (2014) Primary tumor standardized uptake value measured on F18-Fluorodeoxyglucose positron emission tomography is of prediction value for survival and local control in non-small-cell lung cancer receiving radiotherapy: meta-analysis. J Thorac Oncol 9:834–842
Berghmans T, Dusart M, Paesmans M, Hossein-Foucher C, Buvat I, Castaigne C et al (2008) Primary tumor standardized uptake value (SUVmax) measured on fluorodeoxyglucose positron emission tomography (FDG-PET) is of prognostic value for survival in non-small cell lung cancer (NSCLC): a systematic review and meta-analysis (MA) by the European Lung Cancer Working Party for the IASLC Lung Cancer Staging Project. J Thorac Oncol 3:6–12
Kadoya T, Aogi K, Kiyoto S, Masumoto N, Sugawara Y, Okada M (2013) Role of maximum standardized uptake value in fluorodeoxyglucose positron emission tomography/computed tomography predicts malignancy grade and prognosis of operable breast cancer: a multi-institute study. Breast Cancer Res Treat 141:269–275
Kenjiro Aogi T, Kadoya Y, Sugawara S, Kiyoto H, Shigematsu N, Masumoto et al (2015) Utility of 18F FDG-PET/CT for predicting prognosis of luminal-type breast cancer. Breast Cancer Res Treat 150:209–217
Cintolo JA, Tchou J, Pryma DA (2013) Diagnostic and prognostic application of positron emission tomography in breast imaging: emerging uses and the role of PET in monitoring treatment response. Breast Cancer Res Treat 138(2):331–346
Hosono M, Saga T, Ito K, Kumita S, Sasaki M, Senda M et al (2014) Clinical practice guideline for dedicated breast PET. Ann Nucl Med 28:597–602
Fowler AM (2014) A molecular approach to breast imaging. J Nucl Med 55:177–180
Nishimatsu K, Nakamoto Y, Miyake KK, Ishimori T, Kanao S, Toi M et al (2017) Higher breast cancer conspicuity on dbPET compared to WB-PET/CT. Eur J Radiol 90:138–145
Garcia Hernandez T, Vicedo Gonzalez A, Ferrer Rebolleda J, Sanchez Jurado R, Rosello Ferrando J, Brualla Gonzalez L et al (2016) Performance evaluation of a high resolution dedicated breast PET scanner. Med Phys 43:2261
Shinsuke Sasada N, Masumoto N, Goda K, Kajitani A, Emi T, Kadoya et al (2018) Dedicated breast PET for detecting residual disease after neoadjuvant chemotherapy in operable breast cancer: a prospective cohort study. Eur J Surg Oncol 44:444–448
Iima M, Nakamoto Y, Kanao S, Sugie T, Ueno T, Kawada M et al (2012) Clinical performance of 2 dedicated PET scanners for breast imaging: initial evaluation. J Nucl Med 53:1534–1542
Miyake K, Matsumoto K, Inoue M, Nakamoto Y, Kanao S, Oishi T et al (2014) Performance evaluation of a new dedicated breast PET scanner using NEMA NU4-2008 standards. J Nucl Med 55:1198–1203
Yu L, Yang W, Cai X, Shi D, Fan Y, Lu H (2010) Centrally necrotizing carcinoma of the breast: clinicopathological analysis of 33 cases indicating its basal-like phenotype and poor prognosis. Histopathology 57:193–201
Jimenez RE, Wallis T, Visscher DW (2001) Centrally necrotizing carcinomas of the breast: a distinct histologic subtype with aggressive clinical behavior. Am J Surg Pathol 25:331–337
Acknowledgements
We thank Kazushi Marukawa and Masatsugu Tsujimura of Chuden Hospital for providing data regarding PET examinations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have any conflict of interest.
Rights and permissions
About this article
Cite this article
Masumoto, N., Kadoya, T., Sasada, S. et al. Intratumoral heterogeneity on dedicated breast positron emission tomography predicts malignancy grade of breast cancer. Breast Cancer Res Treat 171, 315–323 (2018). https://doi.org/10.1007/s10549-018-4791-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-018-4791-1