Abstract
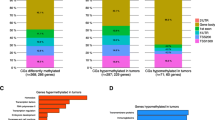
The Vimentin gene plays a pivotal role in epithelial-to-mesenchymal transition and is known to be overexpressed in the prognostically poor basal-like breast cancer subtype. Recent studies have reported Vimentin DNA methylation in association with poor clinical outcomes in other solid tumors, but not in breast cancer. We therefore quantified Vimentin DNA methylation using MALDI-TOF mass spectrometry in breast tumors and matched normal pairs in association with gene expression and survival in a hospital-based study of breast cancer patients. Gene expression data via qRT-PCR in cell lines and oligomicroarray data from breast tissues were correlated with percent methylation in the Vimentin promoter. A threshold of 20 percent average methylation compared with matched normal pairs was set for bivariate and multivariate tests of association between methylation and tumor subtype, tumor histopathology, and survival. Vimentin was differentially methylated in luminal breast cancer cell lines, and in luminal A, luminal B, and HER2-enriched breast tumor subtypes, but was rare in basal-like cell lines and tumors. Increased methylation was strongly correlated with decreased mRNA expression in cell lines, and had a moderate inverse correlation in breast tumors. Vimentin methylation predicted poor overall survival independent of race, subtype, stage, nodal status, or metastatic disease and holds promise as a new prognostic biomarker for breast cancer patients.





Similar content being viewed by others
Abbreviations
- EMT:
-
Epithelial-to-mesenchymal transition
- ER:
-
Estrogen receptor
- HR:
-
Hormone receptor
- HER2:
-
Epidermal growth factor receptor
- OS:
-
Overall survival
- RFS:
-
Recurrence free survival
- mRNA:
-
messenger RNA
- IHC:
-
Immunohistochemical stain
- UNC:
-
University of North Carolina
References
Challa AA, Stefanovic B (2011) A novel role of vimentin filaments: binding and stabilization of collagen mRNAs. Mol Cell Biol 31:3773–3789. doi:10.1128/MCB.05263-11
Chen WD, Han ZJ, Skoletsky J, Olson J, Sah J, Myeroff L, Platzer P, Lu S, Dawson D, Willis J, Pretlow TP, Lutterbaugh J, Kasturi L, Willson JK, Rao JS, Shuber A, Markowitz SD (2005) Detection in fecal DNA of colon cancer-specific methylation of the nonexpressed vimentin gene. J Natl Cancer Inst 97:1124–1132. doi:10.1093/jnci/dji204
Coolen MW, Statham AL, Gardiner-Garden M, Clark SJ (2007) Genomic profiling of CpG methylation and allelic specificity using quantitative high-throughput mass spectrometry: critical evaluation and improvements. Nucleic Acids Res 35:e119. doi:10.1093/nar/gkm662
Costa VL, Henrique R, Danielsen SA, Duarte-Pereira S, Eknaes M, Skotheim RI, Rodrigues A, Magalhaes JS, Oliveira J, Lothe RA, Teixeira MR, Jeronimo C, Lind GE (2010) Three epigenetic biomarkers, GDF15, TMEFF2, and VIM, accurately predict bladder cancer from DNA-based analyses of urine samples. Clin Cancer Res 16:5842–5851. doi:10.1158/1078-0432.CCR-10-1312
Dumont N, Wilson MB, Crawford YG, Reynolds PA, Sigaroudinia M, Tlsty TD (2008) Sustained induction of epithelial to mesenchymal transition activates DNA methylation of genes silenced in basal-like breast cancers. Proc Natl Acad Sci USA 105:14867–14872. doi:10.1073/pnas.0807146105
Ehrich M, Bocker S, van den Boom D (2005) Multiplexed discovery of sequence polymorphisms using base-specific cleavage and MALDI-TOF MS. Nucleic Acids Res 33:e38. doi:10.1093/nar/gni038
Ehrich M, Nelson MR, Stanssens P, Zabeau M, Liloglou T, Xinarianos G, Cantor CR, Field JK, van den Boom D (2005) Quantitative high-throughput analysis of DNA methylation patterns by base-specific cleavage and mass spectrometry. Proc Natl Acad Sci USA 102:15785–15790. doi:10.1073/pnas.0507816102
Fackler MJ, Umbricht CB, Williams D, Argani P, Cruz LA, Merino VF, Teo WW, Zhang Z, Huang P, Visvananthan K, Marks J, Ethier S, Gray JW, Wolff AC, Cope LM, Sukumar S (2011) Genome-wide methylation analysis identifies genes specific to breast cancer hormone receptor status and risk of recurrence. Cancer Res 71:6195–6207. doi:10.1158/0008-5472.CAN-11-1630
Hu Z, Fan C, Oh DS, Marron JS, He X, Qaqish BF, Livasy C, Carey LA, Reynolds E, Dressler L, Nobel A, Parker J, Ewend MG, Sawyer LR, Wu J, Liu Y, Nanda R, Tretiakova M, Ruiz Orrico A, Dreher D, Palazzo JP, Perreard L, Nelson E, Mone M, Hansen H, Mullins M, Quackenbush JF, Ellis MJ, Olopade OI, Bernard PS, Perou CM (2006) The molecular portraits of breast tumors are conserved across microarray platforms. BMC Genomics 7:96. doi:10.1186/1471-2164-7-96
Itzkowitz SH, Jandorf L, Brand R, Rabeneck L, Schroy PC 3rd, Sontag S, Johnson D, Skoletsky J, Durkee K, Markowitz S, Shuber A (2007) Improved fecal DNA test for colorectal cancer screening. Clin Gastroenterol Hepatol 5:111–117. doi:10.1016/j.cgh.2006.10.006
Ivaska J (2011) Vimentin: central hub in EMT induction? Small Gtpases 2:51–53. doi:10.4161/sgtp.2.1.15114
Jung S, Yi L, Kim J, Jeong D, Oh T, Kim CH, Kim CJ, Shin J, An S, Lee MS (2011) The role of vimentin as a methylation biomarker for early diagnosis of cervical cancer. Mol Cells 31:405–411. doi:10.1007/s10059-011-0229-x
Katz E, Dubois-Marshall S, Sims AH, Gautier P, Caldwell H, Meehan RR, Harrison DJ (2011) An in vitro model that recapitulates the epithelial to mesenchymal transition (EMT) in human breast cancer. PLoS ONE 6:e17083. doi:10.1371/journal.pone.0017083
Kisiel JB, Yab TC, Taylor WR, Chari ST, Petersen GM, Mahoney DW, Ahlquist DA (2011) Stool DNA testing for the detection of pancreatic cancer: assessment of methylation marker candidates. Cancer. doi:10.1002/cncr.26558
Kitamura Y, Shirahata A, Sakuraba K, Goto T, Mizukami H, Saito M, Ishibashi K, Kigawa G, Nemoto H, Sanada Y, Hibi K (2011) Aberrant methylation of the Vimentin gene in hepatocellular carcinoma. Anticancer Res 31:1289–1291
Kitamura YH, Shirahata A, Sakata M, Goto T, Mizukami H, Saito M, Ishibashi K, Kigawa G, Nemoto H, Sanada Y, Hibi K (2009) Frequent methylation of Vimentin in well-differentiated gastric carcinoma. Anticancer Res 29:2227–2229
Kokkinos MI, Wafai R, Wong MK, Newgreen DF, Thompson EW, Waltham M (2007) Vimentin and epithelial-mesenchymal transition in human breast cancer—observations in vitro and in vivo. Cells Tissues Organs 185:191–203. doi:10.1159/000101320
Livasy CA, Karaca G, Nanda R, Tretiakova MS, Olopade OI, Moore DT, Perou CM (2006) Phenotypic evaluation of the basal-like subtype of invasive breast carcinoma. Mod Pathol 19:264–271. doi:10.1038/modpathol.3800528
McShane LM, Altman DG, Sauerbrei W, Taube SE, Gion M, Clark GM (2005) Reporting recommendations for tumor marker prognostic studies. J Clin Oncol 23:9067–9072. doi:10.1200/JCO.2004.01.0454
Mendez MG, Kojima S, Goldman RD (2010) Vimentin induces changes in cell shape, motility, and adhesion during the epithelial to mesenchymal transition. The FASEB J 24:1838–1851. doi:10.1096/fj.09-151639
Mor-Vaknin N, Punturieri A, Sitwala K, Markovitz DM (2003) Vimentin is secreted by activated macrophages. Nat Cell Biol 5:59–63. doi:10.1038/ncb898
Nagaraja GM, Othman M, Fox BP, Alsaber R, Pellegrino CM, Zeng Y, Khanna R, Tamburini P, Swaroop A, Kandpal RP (2006) Gene expression signatures and biomarkers of noninvasive and invasive breast cancer cells: comprehensive profiles by representational difference analysis, microarrays and proteomics. Oncogene 25:2328–2338. doi:10.1038/sj.onc.1209265
Ned RM, Melillo S, Marrone M (2011) Fecal DNA testing for colorectal cancer screening: the ColoSure test. PLoS Currents 3:RRN1220. doi:10.1371/currents.RRN1220
Neve RM, Chin K, Fridlyand J, Yeh J, Baehner FL, Fevr T, Clark L, Bayani N, Coppe JP, Tong F, Speed T, Spellman PT, DeVries S, Lapuk A, Wang NJ, Kuo WL, Stilwell JL, Pinkel D, Albertson DG, Waldman FM, McCormick F, Dickson RB, Johnson MD, Lippman M, Ethier S, Gazdar A, Gray JW (2006) A collection of breast cancer cell lines for the study of functionally distinct cancer subtypes. Cancer Cell 10:515–527. doi:10.1016/j.ccr.2006.10.008
Park SY, Lee HE, Li H, Shipitsin M, Gelman R, Polyak K (2010) Heterogeneity for stem cell-related markers according to tumor subtype and histologic stage in breast cancer. Clin Cancer Res 16:876–887. doi:10.1158/1078-0432.CCR-09-1532
Parker JS, Mullins M, Cheang MC, Leung S, Voduc D, Vickery T, Davies S, Fauron C, He X, Hu Z, Quackenbush JF, Stijleman IJ, Palazzo J, Marron JS, Nobel AB, Mardis E, Nielsen TO, Ellis MJ, Perou CM, Bernard PS (2009) Supervised risk predictor of breast cancer based on intrinsic subtypes. J Clin Oncol 27:1160–1167. doi:10.1200/JCO.2008.18.1370
Perou CM (2011) Molecular stratification of triple-negative breast cancers. Oncologist 16(Suppl 1):61–70. doi:10.1634/theoncologist.2011-S1-61
Prat A, Ellis MJ, Perou CM (2012) Practical implications of gene-expression-based assays for breast oncologists. Nat Rev. Clin Oncol 9:48–57. doi:10.1038/nrclinonc.2011.178
Prat A, Parker JS, Karginova O, Fan C, Livasy C, Herschkowitz JI, He X, Perou CM (2010) Phenotypic and molecular characterization of the claudin-low intrinsic subtype of breast cancer. Breast Cancer Res 12:R68. doi:10.1186/bcr2635
Pruitt K, Zinn RL, Ohm JE, McGarvey KM, Kang SH, Watkins DN, Herman JG, Baylin SB (2006) Inhibition of SIRT1 reactivates silenced cancer genes without loss of promoter DNA hypermethylation. PLoS Genet 2:e40. doi:10.1371/journal.pgen.0020040
Radpour R, Haghighi MM, Fan AX, Torbati PM, Hahn S, Holzgreve W, Zhong XY (2008) High-throughput hacking of the methylation patterns in breast cancer by in vitro transcription and thymidine-specific cleavage mass array on MALDI-TOF silico-chip. Mol Cancer Res 6:1702–1709. doi:10.1158/1541-7786.MCR-08-0262
Radpour R, Kohler C, Haghighi MM, Fan AX, Holzgreve W, Zhong XY (2009) Methylation profiles of 22 candidate genes in breast cancer using high-throughput MALDI-TOF mass array. Oncogene 28:2969–2978. doi:10.1038/onc.2009.149
Saeed AI, Bhagabati NK, Braisted JC, Liang W, Sharov V, Howe EA, Li J, Thiagarajan M, White JA, Quackenbush J (2006) TM4 microarray software suite. Methods Enzymol 411:134–193. doi:10.1016/S0076-6879(06)11009-5
Sarrio D, Franklin CK, Mackay A, Reis-Filho JS, Isacke CM (2011) Epithelial and mesenchymal subpopulations within normal basal breast cell lines exhibit distinct stem cell/progenitor properties. Stem Cells. doi:10.1002/stem.791
Sarrio D, Rodriguez-Pinilla SM, Hardisson D, Cano A, Moreno-Bueno G, Palacios J (2008) Epithelial-mesenchymal transition in breast cancer relates to the basal-like phenotype. Cancer Res 68:989–997. doi:10.1158/0008-5472.CAN-07-2017
Satelli A, Li S (2011) Vimentin in cancer and its potential as a molecular target for cancer therapy. Cell Mol Life Sci. doi:10.1007/s00018-011-0735-1
Shirahata A, Sakata M, Sakuraba K, Goto T, Mizukami H, Saito M, Ishibashi K, Kigawa G, Nemoto H, Sanada Y, Hibi K (2009) Vimentin methylation as a marker for advanced colorectal carcinoma. Anticancer Res 29:279–281
Shirahata A, Sakuraba K, Goto T, Saito M, Ishibashi K, Kigawa G, Nemoto H, Hibi K (2010) Detection of vimentin (VIM) methylation in the serum of colorectal cancer patients. Anticancer Res 30:5015–5018
Sousa B, Paredes J, Milanezi F, Lopes N, Martins D, Dufloth R, Vieira D, Albergaria A, Veronese L, Carneiro V, Carvalho S, Costa JL, Zeferino L, Schmitt F (2010) P-cadherin, vimentin and CK14 for identification of basal-like phenotype in breast carcinomas: an immunohistochemical study. Histol Histopathol 25:963–974
Swift-Scanlan T, Vang R, Blackford A, Fackler MJ, Sukumar S (2011) Methylated genes in breast cancer: associations with clinical and histopathological features in a familial breast cancer cohort. Cancer Biol Ther 11:853–865. doi:10.4161/cbt.11.10.15177
Vanaja DK, Ehrich M, Van den Boom D, Cheville JC, Karnes RJ, Tindall DJ, Cantor CR, Young CY (2009) Hypermethylation of genes for diagnosis and risk stratification of prostate cancer. Cancer Invest 27:549–560. doi:10.1080/07357900802620794
Veeck J, Esteller M (2010) Breast cancer epigenetics: from DNA methylation to microRNAs. J Mammary Gland Biol Neoplasia 15:5–17. doi:10.1007/s10911-010-9165-1
Zinn RL, Pruitt K, Eguchi S, Baylin SB, Herman JG (2007) hTERT is expressed in cancer cell lines despite promoter DNA methylation by preservation of unmethylated DNA and active chromatin around the transcription start site. Cancer Res 67:194–201. doi:10.1158/0008-5472.Can-06-3396
Acknowledgments
The authors thank Dr. Tim Wiltshire and the Lineberger Cancer Center for permitting the use of the Mass Array Platform to carry out the methylation studies; Dr. Lisa Carey and Amy Drobish for clinical updates and medical records abstraction; and Dr. Scott Bultman for his thoughtful review of the paper. The authors gratefully acknowledge the support of the NIH/NCI Breast SPORE CA058823 to C.M. Perou, and the KL2RR025746 from the National Center for Research Resources and the Susan G. Komen Foundation KG090180 to T. Swift-Scanlan.
Conflicts of interest
The experiments described in this study comply with the current laws of the country in which they were performed. CMP is a major stockholder of Bioclassifier LLC and co-founder and managing partner of University Genomics. The other authors declare no competing interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ulirsch, J., Fan, C., Knafl, G. et al. Vimentin DNA methylation predicts survival in breast cancer. Breast Cancer Res Treat 137, 383–396 (2013). https://doi.org/10.1007/s10549-012-2353-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-012-2353-5