Abstract
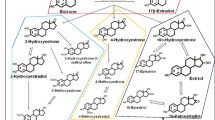
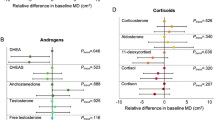
Mammographic density is a strong and independent risk factor for breast cancer and is considered an intermediate marker of risk. The major predictors of premenopausal mammographic density, however, have yet to be fully elucidated. To test the hypothesis that urinary estrogen metabolism profiles are associated with mammographic density, we conducted a cross-sectional study among 352 premenopausal women in the Nurses’ Health Study II (NHSII). We measured average percent mammographic density using a computer-assisted method. In addition, we assayed 15 estrogens and estrogen metabolites (jointly termed EM) in luteal-phase urine samples. We used multivariable linear regression to quantify the association of average percent density with quartiles of each individual EM as well as the sum of all EM (total EM), EM groups defined by metabolic pathway, and pathway ratios. In multivariable models controlling for body mass index and other predictors of breast density, women in the top quartile of total EM had an average percent density 3.4 percentage points higher than women in the bottom quartile (95 % confidence interval: −1.1, 8.0; p trend = 0.08). A non-significant positive association was noted for the 2-hydroxylation pathway catechols (breast density was 4.0 percentage points higher in top vs. bottom quartile; p trend = 0.06). In general, we observed no associations with parent estrogens or the 4- or 16-hydroxylation pathways or pathway ratios. These results suggest that urinary luteal estrogen profiles are not strongly associated with premenopausal mammographic density. If these profiles are associated with breast cancer risk, they may not act through influences on breast density.
Similar content being viewed by others
References
Key T, Appleby P, Barnes I, Reeves G (2002) Endogenous sex hormones and breast cancer in postmenopausal women: reanalysis of nine prospective studies. J Natl Cancer Inst 94(8):606–616
Missmer SA, Eliassen AH, Barbieri RL, Hankinson SE (2004) Endogenous estrogen, androgen, and progesterone concentrations and breast cancer risk among postmenopausal women. J Natl Cancer Inst 96(24):1856–1865. doi:10.1093/jnci/djh336
Walker K, Bratton DJ, Frost C (2011) Premenopausal endogenous oestrogen levels and breast cancer risk: a meta-analysis. Br J Cancer 105(9):1451–1457. doi:10.1038/bjc.2011.358
Kaaks R, Berrino F, Key T, Rinaldi S, Dossus L, Biessy C, Secreto G, Amiano P, Bingham S, Boeing H, Bueno de Mesquita HB, Chang-Claude J, Clavel-Chapelon F, Fournier A, van Gils CH, Gonzalez CA, Gurrea AB, Critselis E, Khaw KT, Krogh V, Lahmann PH, Nagel G, Olsen A, Onland-Moret NC, Overvad K, Palli D, Panico S, Peeters P, Quiros JR, Roddam A, Thiebaut A, Tjonneland A, Chirlaque MD, Trichopoulou A, Trichopoulos D, Tumino R, Vineis P, Norat T, Ferrari P, Slimani N, Riboli E (2005) Serum sex steroids in premenopausal women and breast cancer risk within the European Prospective Investigation into Cancer and Nutrition (EPIC). J Natl Cancer Inst 97(10):755–765. doi:10.1093/jnci/dji132
Eliassen AH, Missmer SA, Tworoger SS, Spiegelman D, Barbieri RL, Dowsett M, Hankinson SE (2006) Endogenous steroid hormone concentrations and risk of breast cancer among premenopausal women. J Natl Cancer Inst 98(19):1406–1415. doi:10.1093/jnci/djj376
McCormack VA, dos Santos Silva I (2006) Breast density and parenchymal patterns as markers of breast cancer risk: a meta-analysis. Cancer Epidemiol Biomarkers Prev 15(6):1159–1169. doi:10.1158/1055-9965.EPI-06-0034
Boyd NF, Martin LJ, Bronskill M, Yaffe MJ, Duric N, Minkin S (2010) Breast tissue composition and susceptibility to breast cancer. J Natl Cancer Inst 102(16):1224–1237. doi:10.1093/jnci/djq239
Martin LJ, Boyd NF (2008) Mammographic density. Potential mechanisms of breast cancer risk associated with mammographic density: hypotheses based on epidemiological evidence. Breast Cancer Res 10 (1):201. doi:10.1186/bcr1831
Bradlow HL, Hershcopf RJ, Martucci CP, Fishman J (1985) Estradiol 16 alpha-hydroxylation in the mouse correlates with mammary tumor incidence and presence of murine mammary tumor virus: a possible model for the hormonal etiology of breast cancer in humans. Proc Natl Acad Sci USA 82(18):6295–6299
Eliassen AH, Spiegelman D, Xu X, Keefer LK, Veenstra TD, Barbieri RL, Willett WC, Hankinson SE, Ziegler RG (2012) Urinary estrogens and estrogen metabolites and subsequent risk of breast cancer among premenopausal women. Cancer Res 72(3):696–706. doi:10.1158/0008-5472.CAN-11-2507
Riza E, dos Santos Silva I, De Stavola B, Bradlow HL, Sepkovic DW, Linos D, Linos A (2001) Urinary estrogen metabolites and mammographic parenchymal patterns in postmenopausal women. Cancer Epidemiol Biomarkers Prev 10(6):627–634
Maskarinec G, Williams AE, Rinaldi S, Kaaks R (2005) Mammographic densities and urinary hormones in healthy women with different ethnic backgrounds. In: Li JJ, Li S, Daling JR (eds) Hormonal carcinogensis IV: Proceedings of the fourth international symposium. Springer, New York, pp 277–286
Fuhrman BJ, Brinton LA, Pfeiffer RM, Xu X, Veenstra TD, Teter BE, Byrne C, Dallal CM, Barba M, Muti P, Gierach GL (2012) Estrogen metabolism and mammographic density in postmenopausal women: a cross-sectional study. Cancer Epidemiol Biomarkers Prev. doi:10.1158/1055-9965.EPI-12-0247
Maskarinec G, Heak S, Morimoto Y, Custer L, Franke AA (2012) The relation of urinary estrogen metabolites with mammographic densities in premenopausal women. Cancer Epidemiol. doi:10.1016/j.canep. 2012.03.014
Yong M, Schwartz SM, Atkinson C, Makar KW, Thomas SS, Newton KM, Aiello Bowles EJ, Holt VL, Leisenring WM, Lampe JW (2010) Associations between polymorphisms in glucuronidation and sulfation enzymes and mammographic breast density in premenopausal women in the United States. Cancer Epidemiol Biomarkers Prev 19(2):537–546. doi:10.1158/1055-9965.EPI-09-0898
Byng JW, Boyd NF, Little L, Lockwood G, Fishell E, Jong RA, Yaffe MJ (1996) Symmetry of projection in the quantitative analysis of mammographic images. Eur J Cancer Prev 5(5):319–327
Pettersson A, Hankinson SE, Willett WC, Lagiou P, Trichopoulos D, Tamimi RM (2011) Nondense mammographic area and risk of breast cancer. Breast Cancer Res 13 (5):R100. doi:10.1186/bcr3041
Rosner B, Cook N, Portman R, Daniels S, Falkner B (2008) Determination of blood pressure percentiles in normal-weight children: some methodological issues. Am J Epidemiol 167(6):653–666. doi:10.1093/aje/kwm348
Byrne C, Colditz GA, Willett WC, Speizer FE, Pollak M, Hankinson SE (2000) Plasma insulin-like growth factor (IGF) I, IGF-binding protein 3, and mammographic density. Cancer Res 60(14):3744–3748
Tamimi RM, Hankinson SE, Colditz GA, Byrne C (2005) Endogenous sex hormone levels and mammographic density among postmenopausal women. Cancer Epidemiol Biomarkers Prev 14(11 Pt 1):2641–2647. doi:10.1158/1055-9965.EPI-05-0558
Stone J, Ding J, Warren RM, Duffy SW, Hopper JL (2010) Using mammographic density to predict breast cancer risk: dense area or percentage dense area. Breast Cancer Res 12(6):R97. doi:10.1186/bcr2778
Lokate M, Peeters PH, Peelen LM, Haars G, Veldhuis WB, van Gils CH (2011) Mammographic density and reast cancer risk: the role of the fat surrounding the fibroglandular tissue. Breast Cancer Res 13(5):R103. doi:10.1186/bcr3044
Falk RT, Xu X, Keefer L, Veenstra TD, Ziegler RG (2008) A liquid chromatography-mass spectrometry method for the simultaneous measurement of 15 urinary estrogens and estrogen metabolites: assay reproducibility and interindividual variability. Cancer Epidemiol Biomarkers Prev 17(12):3411–3418. doi:10.1158/1055-9965.EPI-08-0355
Xu X, Veenstra TD, Fox SD, Roman JM, Issaq HJ, Falk R, Saavedra JE, Keefer LK, Ziegler RG (2005) Measuring fifteen endogenous estrogens simultaneously in human urine by high-performance liquid chromatography-mass spectrometry. Anal Chem 77(20):6646–6654. doi:10.1021/ac050697c
Ziegler RG, Faupel-Badger JM, Sue LY, Fuhrman BJ, Falk RT, Boyd-Morin J, Henderson MK, Hoover RN, Veenstra TD, Keefer LK, Xu X (2010) A new approach to measuring estrogen exposure and metabolism in epidemiologic studies. J Steroid Biochem Mol Biol 121(3–5):538–545. doi:10.1016/j.jsbmb.2010.03.068
Matthews CE, Fortner RT, Xu X, Hankinson SE, Eliassen AH, Ziegler RG (2012) Association between physical activity and urinary estrogens and estrogen metabolites in premenopausal women. J Clin Endocrinol Metab. doi:10.1210/jc.2012-1732
Xie J, Eliassen AH, Xu X, Mathews CE, Hankinson SE, Ziegler RG, Tworoger SS (in press) Body size in relation to urinary estrogens and estrogen metabolites (EM) among premenopausal women during the luteal phase. Hormones Cancer
Fortner RT, Hankinson SE, Schairer C, Xu X, Ziegler RG, Eliassen AH (2012) Association between reproductive factors and urinary estrogens and estrogen metabolites in premenopausal women. Cancer Epidemiol Biomarkers Prev. doi:10.1158/1055-9965.EPI-12-0171
Rosner B (1983) Percentage points for a generalized ESD many-outlier procedure. Technometrics 25:165–172
Vachon CM, Kuni CC, Anderson K, Anderson VE, Sellers TA (2000) Association of mammographically defined percent breast density with epidemiologic risk factors for breast cancer (United States). Cancer Causes Control 11(7):653–662
Boyd NF, Lockwood GA, Byng JW, Little LE, Yaffe MJ, Tritchler DL (1998) The relationship of anthropometric measures to radiological features of the breast in premenopausal women. Br J Cancer 78(9):1233–1238
Samimi G, Colditz GA, Baer HJ, Tamimi RM (2008) Measures of energy balance and mammographic density in the Nurses’ Health Study. Breast Cancer Res Treat 109(1):113–122. doi:10.1007/s10549-007-9631-7
Eliassen AH, Ziegler RG, Rosner B, Veenstra TD, Roman JM, Xu X, Hankinson SE (2009) Reproducibility of fifteen urinary estrogens and estrogen metabolites over a 2- to 3-year period in premenopausal women. Cancer Epidemiol Biomarkers Prev 18(11):2860–2868. doi:10.1158/1055-9965.EPI-09-0591
Taioli E, Im A, Xu X, Veenstra TD, Ahrendt G, Garte S (2010) Comparison of estrogens and estrogen metabolites in human breast tissue and urine. Reprod Biol Endocrinol 8:93. doi:10.1186/1477-7827-8-93
Fuhrman BJ, Schairer C, Gail MH, Boyd-Morin J, Xu X, Sue LY, Buys SS, Isaacs C, Keefer LK, Veenstra TD, Berg CD, Hoover RN, Ziegler RG (2012) Estrogen metabolism and risk of breast cancer in postmenopausal women. J Natl Cancer Inst 104(4):326–339. doi:10.1093/jnci/djr531
Acknowledgments
We thank Barbara DeSouza and Divya Prithviraj for their assistance with data collection. This work was supported in part by the Breast Cancer Research Foundation, the National Institutes of Health, National Cancer Institute (NCI) (CA124865, CA67262, and CA50385), the Intramural Research Program of the NCI Division of Cancer Epidemiology and Genetics, and with federal funds of the NCI awarded under Contract HHSN261200800001E to SAIC-Frederick. K.A.B. was supported by the Nutritional Epidemiology of Cancer Training Grant (R25 CA098566). The content of this publication does not necessarily reflect the views or policies of the U.S. Department of Health and Human Services; nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. Government.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Regina G. Ziegler, Rulla M. Tamimi—co-last authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bertrand, K.A., Eliassen, A.H., Hankinson, S.E. et al. Urinary estrogens and estrogen metabolites and mammographic density in premenopausal women. Breast Cancer Res Treat 136, 277–287 (2012). https://doi.org/10.1007/s10549-012-2240-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-012-2240-0