Abstract
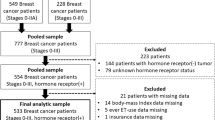
Adjuvant hormonal therapy for non-metastatic hormone receptor (HR)-positive breast cancer decreases risk of breast cancer recurrence and increases survival. However, some women do not initiate this life-saving treatment. We used a prospective cohort design to investigate factors related to non-initiation of hormonal therapy among women with newly diagnosed, non-metastatic HR-positive breast cancer recruited from three U.S. sites. Serial interviews were conducted at baseline and during treatment to examine sociodemographic factors, tumor characteristics, and treatment decision-making factors. Multivariate modeling assessed associations between variables of interest and hormonal therapy initiation. Of 1,050 breast cancer patients recruited, 725 (69 %) had HR-positive breast cancer, of whom 87 (12.0 %) based on self-report and 122 (16.8 %) based on medical record/pharmacy fill rates did not initiate hormonal therapy. In a multivariable analysis, non-initiation of hormonal therapy, defined by medical record/pharmacy, was associated with having greater negative beliefs about efficacy of treatment (OR 1.42, 95 % CI 1.18–1.70). Non-initiation was less likely in those who found the quality of patient/physician communication to be higher (OR 0.96, 95 % CI 0.93–0.99), the hormonal therapy treatment decision an easy one to make (OR 0.45, 95 % CI 0.23–0.90) or neither easy nor difficult (OR 0.34, 95 % CI 0.20–0.58); and had more positive beliefs about hormonal therapy efficacy (OR 0.40, 95 % CI 0.34–0.62). Factors influencing non-initiation of adjuvant hormonal therapy are complex and influenced by patient beliefs regarding treatment efficacy and side effects. Educational interventions to women about the benefits of hormonal therapy may decrease negative beliefs and increase hormone therapy initiation.
Similar content being viewed by others
References
Clarke M, Collins R, Darby S et al (2005) Effects of radiotherapy and of differences in the extent of surgery for early breast cancer on local recurrence and 15-year survival: an overview of the randomised trials. Lancet 366(9503):2087–2106
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG) (2005) Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet 365(9472):1687–1717
Hershman D, McBride R, Jacobson JS et al (2005) Racial disparities in treatment and survival among women with early stage breast cancer. J Clin Oncol 23(27):6639–6646
Lyman GH, Dale DC, Crawford J (2003) Incidence and predictors of low dose-intensity in adjuvant breast cancer chemotherapy: a nationwide study of community practices. J Clin Oncol 21(24):4524–4531
Partridge AH, Wang PS, Winer EP, Avorn J (2003) Nonadherence to adjuvant tamoxifen therapy in women with primary breast cancer. J Clin Oncol 21(4):602–606
Bickell NA, McEvoy MD (2003) Physicians’ reasons for failing to deliver effective breast cancer care: a framework for underuse. Med Care 41(3):442–446
Hershman DL, Wang X, McBride R, Jacobson JS, Grann VR, Neugut AI (2006) Delay of adjuvant chemotherapy initiation following breast cancer surgery among elderly women. Breast Cancer Res Treat 99(3):313–321
Hershman DL, Wang X, McBride R, Jacobson JS, Grann VR, Neugut AI (2006) Delay in initiating adjuvant radiotherapy following breast conservation surgery and its impact on survival. Int J Radiat Oncol Biol Phys 65(5):1353–1360
Fedewa SA, Ward EM, Stewart AK, Edge SB (2010) Delays in adjuvant chemotherapy treatment among patients with breast cancer are more likely in African American and Hispanic populations: a national cohort study 2004–2006. J Clin Oncol 28(27):4135–4141
Silliman RA, Guadagnoli E, Weitberg AB, Mor V (1989) Age as a predictor of diagnostic and initial treatment intensity in newly diagnosed breast cancer patients. J Gerontol 44(2):M46–M50
Hershman DL, Kushi LH, Shao T et al (2010) Early discontinuation and nonadherence to adjuvant hormonal therapy in a cohort of 8,769 early stage breast cancer patients. J Clin Oncol 28(27):4120–4128
Jordan VC (1980) Evaluation of the antitumour activity of the non-steroidal antioestrogen monohydroxytamoxifen in the DMBA-induced rat mammary carcinoma model. Eur J Cancer 16(2):239–251
Baum M, Budzar AU, Cuzick J et al (2002) Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early breast cancer: first results of the ATAC randomised trial. Lancet 359(9324):2131–2139
Coombes RC, Hall E, Gibson LJ et al (2004) A randomized trial of exemestane after two to three years of tamoxifen therapy in postmenopausal women with primary breast cancer. N Engl J Med 350(11):1081–1092
Goss PE, Ingle JN, Martino S et al (2005) Randomized trial of letrozole following tamoxifen as extended adjuvant therapy in receptor-positive breast cancer: updated findings from NCIC CTG MA.17. J Natl Cancer Inst 97(17):1262–1271
Goss PE, Ingle JN, Martino S et al (2003) A randomized trial of letrozole in postmenopausal women after five years of tamoxifen therapy for early stage breast cancer. N Engl J Med 349(19):1793–1802
Howell A, Cuzick J, Baum M et al (2005) Results of the ATAC (Arimidex, Tamoxifen, Alone or in Combination) trial after completion of 5 years’ adjuvant treatment for breast cancer. Lancet 365(9453):60–62
Thurlimann B, Keshaviah A, Coates AS et al (2005) A comparison of letrozole and tamoxifen in postmenopausal women with early breast cancer. N Engl J Med 353(26):2747–2757
Bickell NA, LePar F, Wang JJ, Leventhal H (2007) Lost opportunities: physicians’ reasons and disparities in breast cancer treatment. J Clin Oncol 25(18):2516–2521
Bickell NA, Wang JJ, Oluwole S et al (2006) Missed opportunities: racial disparities in adjuvant breast cancer treatment. J Clin Oncol 24(9):1357–1362
Kimmick G, Anderson R, Camacho F, Bhosle M, Hwang W, Balkrishnan R (2009) Adjuvant hormonal therapy use among insured, low-income women with breast cancer. J Clin Oncol 27(21):3445–3451
Pini T, Griggs J, Hamilton A, Katz S (2011) Patterns and correlates of adjuvant breast cancer endocrine therapy use. J Clin Oncol 29 (suppl; abstr 510)
Quinn V, Strauss J, Schottinger J, Cheetham C, Haque R (2011) When to intervene to increase use of adjuvant hormone therapy among with hormone-sensitive breast cancer. J Clin Oncol 29 (suppl; abstr 511)
Demissie S, Silliman RA, Lash TL (2001) Adjuvant tamoxifen: predictors of use, side effects, and discontinuation in older women. J Clin Oncol 19(2):322–328
Hershman DL, Shao T, Kushi LH et al (2011) Early discontinuation and non-adherence to adjuvant hormonal therapy are associated with increased mortality in women with breast cancer. Breast Cancer Res Treat 126(2):529–537
Fink AK, Gurwitz J, Rakowski W, Guadagnoli E, Silliman RA (2004) Patient beliefs and tamoxifen discontinuance in older women with estrogen receptor-positive breast cancer. J Clin Oncol 22(16):3309–3315
Neugut AI, Clarke Hillyer G, Kushi LH et al (2012) The breast cancer quality of care study (B-QUAL): a multi-center study to determine causes for non-compliance with breast cancer adjuvant therapy. Breast J. doi:10.1111/j.1524-4741.2012.01240.x
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40(5):373–383
Llewellyn-Thomas HA, McGreal MJ, Thiel EC, Fine S, Erlichman C (1991) Patients’ willingness to enter clinical trials: measuring the association with perceived benefit and preference for decision participation. Soc Sci Med 32(1):35–42
Mandelblatt JS, Sheppard VB, Hurria A et al (2010) Breast cancer adjuvant chemotherapy decisions in older women: the role of patient preference and interactions with physicians. J Clin Oncol 28(19):3146–3153
Sherbourne CD, Stewart AL (1991) The MOS social support survey. Soc Sci Med 32(6):705–714
Maunsell E, Drolet M, Ouhoummane N, Robert J (2005) Breast cancer survivors accurately reported key treatment and prognostic characteristics. J Clin Epidemiol 58(4):364–369
Phillips K-A, Milne RL, Buys S et al (2005) Agreement between self-reported breast cancer treatment and medical records in a population-based breast cancer family registry. J Clin Oncol 23(21):4679–4686
Ruddy K, Mayer E, Partridge A (2009) Patient adherence and persistence with oral anticancer treatment. CA Cancer J Clin 59(1):56–66
Bell RJ, Lijovic M, Fradkin P et al (2009) Lack of knowledge of hormone receptor status and use of endocrine therapy in invasive breast cancer. J Women’s Health 18(12):1975–1980
Bickell NA, Weidmann J, Fei K, Lin JJ, Leventhal H (2009) Underuse of breast cancer adjuvant treatment: patient knowledge, beliefs, and medical mistrust. J Clin Oncol 27(31):5160–5167
Osterberg L, Blaschke T (2005) Adherence to medication. N Engl J Med 353(5):487–497
Maly RC, Leake B, Silliman RA (2004) Breast cancer treatment in older women: impact of the patient-physician interaction. J Am Geriatr Soc 52(7):1138–1145
Elwyn G, O’Connor A, Stacey D et al (2006) Developing a quality criteria framework for patient decision aids: online international Delphi consensus process. Br Med J 333(7565):417
Sepucha KR, Fowler FJ Jr, Mulley AG Jr (2004) Policy support for patient-centered care: the need for measurable improvements in decision quality. Health Aff (Millwood) Suppl Variation:VAR54–62
Siminoff LA, Ravdin P, Colabianchi N, Sturm CM (2000) Doctor–patient communication patterns in breast cancer adjuvant therapy discussions. Health Expect 3(1):26–36
Waters EA, Weinstein ND, Colditz GA, Emmons K (2009) Explanations for side effect aversion in preventive medical treatment decisions. Health Psychol 28(2):201–209
Horne R, Weinman J (1999) Patients’ beliefs about prescribed medicines and their role in adherence to treatment in chronic physical illness. J Psychosom Res 47(6):555–567
Iihara N, Tsukamoto T, Morita S, Miyoshi C, Takabatake K, Kurosaki Y (2004) Beliefs of chronically ill Japanese patients that lead to intentional non-adherence to medication. J Clin Pharm Ther 29(5):417–424
Heisey R, Pimlott N, Clemons M, Cummings S, Drummond N (2006) Women’s views on chemoprevention of breast cancer: qualitative study. Can Fam Physician 52:624–625
Grunfeld EA, Hunter MS, Sikka P, Mittal S (2005) Adherence beliefs among breast cancer patients taking tamoxifen. Patient Educ Couns 59(1):97–102
Lash TL, Fox MP, Westrup JL, Fink AK, Silliman RA (2006) Adherence to tamoxifen over the five-year course. Breast Cancer Res Treat 99(2):215–220
Livaudais JC, Hershman DL, Habel L et al (2012) Racial/ethnic differences in initiation of adjuvant hormonal therapy among women with hormone receptor-positive breast cancer. Breast Cancer Res Treat 131(2):607–617
Acknowledgments
Supported by a Department of Defense Breast Cancer Center of Excellence Award (BC043120) to Dr. Neugut, a NCI R01 (CA105274), a Department of Defense Center for Biobehavioral Breast Cancer Research to Dr. Kushi, an NCI R25 fellowship (CA094601) to N. Leoce, Department of Defense (DAMD-17-01-1-0334) to Dr. Bovbjerg, a NCI R01 (CA100598) to Dr. Ambrosone, and a NCI R01 (CA124924 and 127617), U10 (CA 84131) and K05 (CA96940) to Dr. Mandelblatt. Drs. Hershman and Ambrosone are recipients of funding from the Breast Cancer Research Foundation.
Conflict of interest
The authors declare they have no conflicts of interest which may have influenced the results of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Neugut, A.I., Hillyer, G.C., Kushi, L.H. et al. Non-initiation of adjuvant hormonal therapy in women with hormone receptor-positive breast cancer: The Breast Cancer Quality of Care Study (BQUAL). Breast Cancer Res Treat 134, 419–428 (2012). https://doi.org/10.1007/s10549-012-2066-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-012-2066-9