Abstract
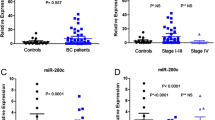
MicroRNAs play a role in breast cancer development and progression by post-transcriptional repression of the expression of important genes, such as the tumor suppressor gene phosphatase and tensin homolog (PTEN). The focus of the current study was to examine the diagnostic potential of circulating cell-free microRNAs targeting PTEN in breast cancer. Our analyses were performed on preoperative serum samples of 102 patients with early breast cancer and a subset of 34 postoperative samples, as well as of 32 patients with benign breast disease and 53 healthy women. The relative concentrations of four circulating microRNAs (miR-19a, miR-20a, miR-21, and miR-214) in blood serum were measured by TaqMan MicroRNA assays. Levels of preoperative serum miR-20a and miR-21 were significantly higher in patients with breast cancer and benign disease than in healthy women (p = 0.0001), but only serum miR-214 could discriminate malignant from benign tumors and healthy controls (p = 0.0001) with an area under the curve of 0.878 and 0.883 in ROC analysis, respectively. Moreover, miR-214 levels significantly decreased in the postoperative serum samples (p = 0.0001) as compared to the preoperative samples. The comparison with the clinicopathologic data of the breast cancer patients showed that increased miR-214 levels were associated with a positive lymph node status (p = 0.039). Our data show that circulating, cell-free miR-214 has diagnostic potential in breast cancer as indicator of malignant disease and metastatic spread to regional lymph nodes. Since PTEN is an important target gene of miR-214, this finding could also have potential implications for therapeutic approaches.


Similar content being viewed by others
References
Colozza M, Cardoso F, Sotiriou C, Larsimont D, Piccart MJ (2005) Bringing molecular prognosis and prediction to the clinic. Clin Breast Cancer 6(1):61–76
Pantel K, Alix-Panabieres C, Riethdorf S (2009) Cancer micrometastases. Nat Rev Clin Oncol 6(6):339–351
Pantel K, Brakenhoff RH (2004) Dissecting the metastatic cascade. Nat Rev Cancer 4(6):448–456
Dumitrescu RG, Cotarla I (2005) Understanding breast cancer risk—where do we stand in 2005? J Cell Mol Med 9(1):208–221
Blanco-Aparicio C, Renner O, Leal JF, Carnero A (2007) PTEN, more than the AKT pathway. Carcinogenesis 28(7):1379–1386
Dillon RL, White DE, Muller WJ (2007) The phosphatidyl inositol 3-kinase signaling network: implications for human breast cancer. Oncogene 26(9):1338–1345
Kloosterman WP, Plasterk RH (2006) The diverse functions of microRNAs in animal development and disease. Dev Cell 11(4):441–450
Stefani G, Slack FJ (2008) Small non-coding RNAs in animal development. Natl Rev Mol Cell Biol 9(3):219–230
Bernstein E, Kim SY, Carmell MA, Murchison EP, Alcorn H, Li MZ, Mills AA, Elledge SJ, Anderson KV, Hannon GJ (2003) Dicer is essential for mouse development. Nat Genet 35(3):215–217
Heneghan HM, Miller N, Lowery AJ, Sweeney KJ, Kerin MJ (2009) MicroRNAs as novel biomarkers for breast cancer. J Oncol 2009:950201
Lewis BP, Shih IH, Jones-Rhoades MW, Bartel DP, Burge CB (2003) Prediction of mammalian microRNA targets. Cell 115(7):787–798
Cao Y, Yu SL, Wang Y, Guo GY, Ding Q, An RH (2011) MicroRNA-dependent regulation of PTEN after arsenic trioxide treatment in bladder cancer cell line T24. Tumour Biol 32(1):179–188
Trompeter HI, Abbad H, Iwaniuk KM, Hafner M, Renwick N, Tuschl T, Schira J, Muller HW, Wernet P (2011) MicroRNAs MiR-17, MiR-20a, and MiR-106b act in concert to modulate E2F activity on cell cycle arrest during neuronal lineage differentiation of USSC. PLoS ONE 6(1):e16138
Wickramasinghe NS, Manavalan TT, Dougherty SM, Riggs KA, Li Y, Klinge CM (2009) Estradiol downregulates miR-21 expression and increases miR-21 target gene expression in MCF-7 breast cancer cells. Nucleic Acids Res 37(8):2584–2595
Yang H, Kong W, He L, Zhao JJ, O’Donnell JD, Wang J, Wenham RM, Coppola D, Kruk PA, Nicosia SV, Cheng JQ (2008) MicroRNA expression profiling in human ovarian cancer: miR-214 induces cell survival and cisplatin resistance by targeting PTEN. Cancer Res 68(2):425–433
Schwarzenbach H, Hoon DS, Pantel K (2011) Cell-free nucleic acids as biomarkers in cancer patients. Nat Rev Cancer 11(6):426–437
Lawrie CH, Gal S, Dunlop HM, Pushkaran B, Liggins AP, Pulford K, Banham AH, Pezzella F, Boultwood J, Wainscoat JS, Hatton CS, Harris AL (2008) Detection of elevated levels of tumour-associated microRNAs in serum of patients with diffuse large B-cell lymphoma. Br J Haematol 141(5):672–675
Esquela-Kerscher A, Slack FJ (2006) Oncomirs—microRNAs with a role in cancer. Nat Rev Cancer 6(4):259–269
Schwarzenbach H, Muller V, Beeger C, Gottberg M, Stahmann N, Pantel K (2007) A critical evaluation of loss of heterozygosity detected in tumor tissues, blood serum and bone marrow plasma from patients with breast cancer. Breast Cancer Res 9(5):R66
Mostert B, Sieuwerts AM, Martens JW, Sleijfer S (2011) Diagnostic applications of cell-free and circulating tumor cell-associated miRNAs in cancer patients. Expert Rev Mol Diagn 11(3):259–275
Muller V, Alix-Panabieres C, Pantel K (2010) Insights into minimal residual disease in cancer patients: implications for anti-cancer therapies. Eur J Cancer 46(7):1189–1197
Tsujiura M, Ichikawa D, Komatsu S, Shiozaki A, Takeshita H, Kosuga T, Konishi H, Morimura R, Deguchi K, Fujiwara H, Okamoto K, Otsuji E (2010) Circulating microRNAs in plasma of patients with gastric cancers. Br J Cancer 102(7):1174–1179
Ng EK, Chong WW, Jin H, Lam EK, Shin VY, Yu J, Poon TC, Ng SS, Sung JJ (2009) Differential expression of microRNAs in plasma of patients with colorectal cancer: a potential marker for colorectal cancer screening. Gut 58(10):1375–1381
Jindra PT, Bagley J, Godwin JG, Iacomini J (2010) Costimulation-dependent expression of microRNA-214 increases the ability of T cells to proliferate by targeting Pten. J Immunol 185(2):990–997
Li LM, Hou DX, Guo YL, Yang JW, Liu Y, Zhang CY, Zen K (2011) Role of microRNA-214-targeting phosphatase and tensin homolog in advanced glycation end product-induced apoptosis delay in monocytes. J Immunol 186(4):2552–2560
Razis E, Bobos M, Kotoula V, Eleftheraki AG, Kalofonos HP, Pavlakis K, Papakostas P, Aravantinos G, Rigakos G, Efstratiou I, Petraki K, Bafaloukos D, Kostopoulos I, Pectasides D, Kalogeras KT, Skarlos D, Fountzilas G (2011) Evaluation of the association of PIK3CA mutations and PTEN loss with efficacy of trastuzumab therapy in metastatic breast cancer. Breast Cancer Res Treat 128(2):447–456
Pohlmann PR, Mayer IA, Mernaugh R (2009) Resistance to trastuzumab in breast cancer. Clin Cancer Res 15(24):7479–7491
Gotte M (2010) MicroRNAs in breast cancer pathogenesis. Minerva Ginecol 62(6):559–571
Calin GA, Dumitru CD, Shimizu M, Bichi R, Zupo S, Noch E, Aldler H, Rattan S, Keating M, Rai K, Rassenti L, Kipps T, Negrini M, Bullrich F, Croce CM (2002) Frequent deletions and down-regulation of micro- RNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc Natl Acad Sci USA 99(24):15524–15529
Olive V, Jiang I, He L (2010) mir-17-92, a cluster of miRNAs in the midst of the cancer network. Int J Biochem Cell Biol 42(8):1348–1354
Hayashita Y, Osada H, Tatematsu Y, Yamada H, Yanagisawa K, Tomida S, Yatabe Y, Kawahara K, Sekido Y, Takahashi T (2005) A polycistronic microRNA cluster, miR-17-92, is overexpressed in human lung cancers and enhances cell proliferation. Cancer Res 65(21):9628–9632
Moussay E, Wang K, Cho JH, van Moer K, Pierson S, Paggetti J, Nazarov PV, Palissot V, Hood LE, Berchem G, Galas DJ (2011) MicroRNA as biomarkers and regulators in B-cell chronic lymphocytic leukemia. Proc Natl Acad Sci USA 108(16):6573–6578
Asaga S, Kuo C, Nguyen T, Terpenning M, Giuliano AE, Hoon DS (2011) Direct serum assay for microRNA-21 concentrations in early and advanced breast cancer. Clin Chem 57(1):84–91
Song B, Wang C, Liu J, Wang X, Lv L, Wei L, Xie L, Zheng Y, Song X (2010) MicroRNA-21 regulates breast cancer invasion partly by targeting tissue inhibitor of metalloproteinase 3 expression. J Exp Clin Cancer Res 29:29
Ota D, Mimori K, Yokobori T, Iwatsuki M, Kataoka A, Masuda N, Ishii H, Ohno S, Mori M (2011) Identification of recurrence-related microRNAs in the bone marrow of breast cancer patients. Int J Oncol 38(4):955–962
Komatsu S, Ichikawa D, Takeshita H, Tsujiura M, Morimura R, Nagata H, Kosuga T, Iitaka D, Konishi H, Shiozaki A, Fujiwara H, Okamoto K, Otsuji E (2011) Circulating microRNAs in plasma of patients with oesophageal squamous cell carcinoma. Br J Cancer 105(1):104–111
Wei J, Gao W, Zhu CJ, Liu YQ, Mei Z, Cheng T, Shu YQ (2011) Identification of plasma microRNA-21 as a biomarker for early detection and chemosensitivity of non-small cell lung cancer. Chin J Cancer 30(6):407–414
Acknowledgment
We are grateful to the European Research Council (Advanced Investigators Grant 269081-DISSECT) and the Erich und Gertrud Roggenbuck-Stiftung, Hamburg, for supporting this study. We thank Ms. Carina Roth for their excellent technical assistance.
Conflict of interest
All authors disclose any financial and personal relationships with other people or organizations that could inappropriately influence (bias) their study.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Schwarzenbach, H., Milde-Langosch, K., Steinbach, B. et al. Diagnostic potential of PTEN-targeting miR-214 in the blood of breast cancer patients. Breast Cancer Res Treat 134, 933–941 (2012). https://doi.org/10.1007/s10549-012-1988-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-012-1988-6