Abstract
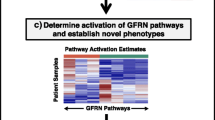
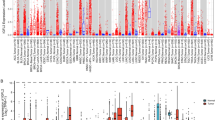
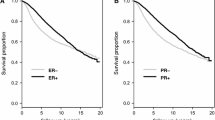
The insulin-like growth factor (IGF) axis is fundamentally important in cell growth, development and cancer. We used genomic technologies to better characterize the activity of the IGF axis in human breast cancer and to identify predictors of response to IGF targeted therapies. Analysis of the gene expression patterns and pathway analysis in 204 clinically annotated primary breast cancers were performed and compared to levels of mRNA for IGF ligands and receptors. Pathway activation scores were calculated by Pearson correlation (+1, −1). Network analysis was performed using Ingenuity software. IGF-1 ligand levels were strongly negatively correlated (P < 10−6) with a published IGF-IR activation signature. A signature of high IGF-1 ligand was associated with better prognosis (P = 0.025–1.5 × 10−8) in several public datasets. Pathway analysis revealed upregulation of pathways associated with breast differentiation (adipocyte growth factors, PPAR-gamma) and down-regulation of proliferation pathways (AKT/MAPK) in the IGF-1 ligand high group. Of note, the IGF-1 ligand signature was anti-correlated with IGFIR receptor levels (P = 0.07). In conclusion, a breast tumor-derived signature of high IGF-1 ligand is associated with favorable outcome, in contrast to a previously reported IGF-IR activation signature. The prognostic value of the IGF-I ligand signature is validated in three independent datasets. These signatures should be applied in study of IGF1-R targeted therapy.




Similar content being viewed by others
References
Bonneterre J, Peyrat JP, Beuscart R, Demaille A (1990) Prognostic significance of insulin-like growth factor 1 receptors in human breast cancer. Cancer Res 50(21):6931–6935
Blum G, Gazit A, Levitzki A (2003) Development of new insulin-like growth factor-1 receptor kinase inhibitors using catechol mimics. J Biol Chem 278(42):40442–40454
Burtrum D, Zhu Z, Lu D, Anderson DM, Prewett M, Pereira DS et al (2003) A fully human monoclonal antibody to the insulin-like growth factor I receptor blocks ligand-dependent signaling and inhibits human tumor growth in vivo. Cancer Res 63(24):8912–8921
Pollak MN, Schernhammer ES, Hankinson SE (2004) Insulin-like growth factors and neoplasia. Nat Rev Cancer 4(7):505–518
Surmacz E (2003) Growth factor receptors as therapeutic targets: strategies to inhibit the insulin-like growth factor I receptor. Oncogene. 22(42):6589–6597
Baserga R, Glazer PM, Turner BC, Haffty BG, Narayanan L, Yuan J et al (1997) Insulin-like growth factor-I receptor overexpression mediates cellular radioresistance and local breast cancer recurrence after lumpectomy and radiation. Cancer Res 57(15):3079–3083
Sachdev D, Hartell JS, Lee AV, Zhang X, Yee D (2004) A dominant negative type I insulin-like growth factor receptor inhibits metastasis of human cancer cells. J Biol Chem 279(6):5017–5024
Creighton CJ, Casa A, Lazard Z, Huang S, Tsimelzon A, Hilsenbeck SG et al (2008) Insulin-like growth factor-I activates gene transcription programs strongly associated with poor breast cancer prognosis. J Clin Oncol 26(25):4078–4085
Mu L, Katsaros D, Wiley A, Lu L, de la Longrais I, Smith S et al (2008) Peptide concentrations and mRNA expression of IGF-I, IGF-II and IGFBP-3 in breast cancer and their associations with disease characteristics. Breast Cancer Res Treat [Internet] (cited 2008 Sep 27). http://www.ncbi.nlm.nih.gov/pubmed/18481170
Gentleman R, Carey V, Bates D, Bolstad B, Dettling M, Dudoit S et al (2004) Bioconductor: open software development for computational biology and bioinformatics. Genome Biol 5(10):R80
Du P, Kibbe WA, Lin SM (2008) lumi: a pipeline for processing Illumina microarray. Bioinformatics 24(13):1547–1548
Lin SM, Du P, Huber W, Kibbe WA (2008) Model-based variance-stabilizing transformation for Illumina microarray data. Nucleic Acids Res 36(2):e11
Smyth GK (2004) Linear models and empirical Bayes methods for assessing differential expression in microarray experiments. Stat Appl Genet Mol Biol. 3(3)
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B (Methodol) 57(1):289–300
The R Project for Statistical Computing. http://www.r-project.org)
Wang Y, Klijn JGM, Zhang Y, Sieuwerts AM, Look MP, Yang F et al (2005) Gene-expression profiles to predict distant metastasis of lymph-node-negative primary breast cancer. Lancet 365(9460):671–679
van de Vijver MJ, He YD, Van’t Veer LJ, Dai H, Hart AAM, Voskuil DW et al (2002) A gene-expression signature as a predictor of survival in breast cancer. N Engl J Med 347(25):1999–2009
Chin K, DeVries S, Fridlyand J, Spellman PT, Roydasgupta R, Kuo W et al (2006) Genomic and transcriptional aberrations linked to breast cancer pathophysiologies. Cancer Cell 10(6):529–541
Parker JS, Mullins M, Cheang MCU, Leung S, Voduc D, Vickery T et al (2009) Supervised risk predictor of breast cancer based on intrinsic subtypes. J Clin Oncol 27(8):1160–1167
Ingenuity Pathways Analysis (IPA; Ingenuity Systems, Redwood City, CA). http://www.ingenuity.com
Resnik JL, Reichart DB, Huey K, Webster NJ, Seely BL (1998) Elevated insulin-like growth factor I receptor autophosphorylation and kinase activity in human breast cancer. Cancer Res 58(6):1159–1164
Kim H, Litzenburger BC, Cui X, Delgado DA, Grabiner BC, Lin X et al (2007) Constitutively active type I insulin-like growth factor receptor causes transformation and xenograft growth of immortalized mammary epithelial cells and is accompanied by an epithelial-to-mesenchymal transition mediated by NF-{kappa}B and snail. Mol Cell Biol 27(8):3165–3175
Dunn SE, Ehrlich M, Sharp NJ, Reiss K, Solomon G, Hawkins R et al (1998) A dominant negative mutant of the insulin-like growth factor-I receptor inhibits the adhesion, invasion, and metastasis of breast cancer. Cancer Res 58(15):3353–3361
Zhang X, Kamaraju S, Hakuno F, Kabuta T, Takahashi S, Sachdev D et al (2004) Motility response to insulin-like growth factor-I (IGF-I) in MCF-7 cells is associated with IRS-2 activation and integrin expression. Breast Cancer Res Treat 83(2):161–170
Shin A, Ren Z, Shu X, Cai Q, Gao Y, Zheng W (2007) Expression patterns of insulin-like growth factor 1 (IGF-I) and its receptor in mammary tissues and their associations with breast cancer survival. Breast Cancer Res Treat 105(1):55–61
Kato S, Masuhiro Y, Watanabe M, Kobayashi Y, Takeyama KI, Endoh H et al (2000) Molecular mechanism of a cross-talk between oestrogen and growth factor signalling pathways. Genes Cells. 5(8):593–601
Huynh H, Nickerson T, Pollak M, Yang X (1996) Regulation of insulin-like growth factor I receptor expression by the pure antiestrogen ICI 182780. Clin Cancer Res 2(12):2037–2042
Lee AV, Weng CN, Jackson JG, Yee D (1997) Activation of estrogen receptor-mediated gene transcription by IGF-I in human breast cancer cells. J Endocrinol 152(1):39–47
Lee AV, Jackson JG, Gooch JL, Hilsenbeck SG, Coronado-Heinsohn E, Osborne CK et al (1999) Enhancement of insulin-like growth factor signaling in human breast cancer: estrogen regulation of insulin receptor substrate-1 expression in vitro and in vivo. Mol Endocrinol 13(5):787–796
Decensi A, Robertson C, Ballardini B, Paggi D, Guerrieri-Gonzaga A, Bonanni B et al (1999) Effect of tamoxifen on lipoprotein(a) and insulin-like growth factor-I (IGF-I) in healthy women. Eur J Cancer 35(4):596–600
Happerfield LC, Miles DW, Barnes DM, Thomsen LL, Smith P, Hanby A (1997) The localization of the insulin-like growth factor receptor 1 (IGFR-1) in benign and malignant breast tissue. J Pathol 183(4):412–417
Ye J, Liang S, Guo N, Li S, Wu AM, Giannini S et al (2003) Combined effects of tamoxifen and a chimeric humanized single chain antibody against the type I IGF receptor on breast tumor growth in vivo. Horm Metab Res 35(11–12):836–842
Ciarmatori S, Kiepe D, Haarmann A, Huegel U, Tönshoff B (2007) Signaling mechanisms leading to regulation of proliferation and differentiation of the mesenchymal chondrogenic cell line RCJ3.1C5.18 in response to IGF-I. J Mol Endocrinol 38(4):493–508
Ewton DZ, Kansra S, Lim S, Friedman E (2002) Insulin-like growth factor-I has a biphasic effect on colon carcinoma cells through transient inactivation of forkhead1, initially mitogenic, then mediating growth arrest and differentiation. Int J Cancer 98(5):665–673
Rosenthal SM, Cheng ZQ (1995) Opposing early and late effects of insulin-like growth factor I on differentiation and the cell cycle regulatory retinoblastoma protein in skeletal myoblasts. Proc Natl Acad Sci USA 92(22):10307–10311
Wei Y, Chen Y, Li D, Gu R, Wang W (2004) Dual effect of insulin-like growth factor on the apical 70-pS K channel in the thick ascending limb of rat kidney. Am J Physiol Cell Physiol 286(6):C1258–C1263
Lupu R, Cardillo M, Cho C, Harris L, Hijazi M, Perez C et al (1996) The significance of heregulin in breast cancer tumor progression and drug resistance. Breast Cancer Res Treat 38(1):57–66
Marte BM, Jeschke M, Graus-Porta D, Taverna D, Hofer P, Groner B et al (1995) Neu differentiation factor/heregulin modulates growth and differentiation of HC11 mammary epithelial cells. Mol Endocrinol 9(1):14–23
Nagashima T, Shimodaira H, Ide K, Nakakuki T, Tani Y, Takahashi K et al (2007) Quantitative transcriptional control of ErbB receptor signaling undergoes graded to biphasic response for cell differentiation. J Biol Chem 282(6):4045–4056
Staebler A, Sommers C, Mueller SC, Byers S, Thompson EW, Lupu R (1994) Modulation of breast cancer progression and differentiation by the gp30/heregulin [correction of neregulin]. Breast Cancer Res Treat 31(2–3):175–182
Xu F, Yu Y, Le XF, Boyer C, Mills GB, Bast RC (1999) The outcome of heregulin-induced activation of ovarian cancer cells depends on the relative levels of HER-2 and HER-3 expression. Clin Cancer Res 5(11):3653–3660
Hallstrom TC, Mori S, Nevins JR (2008) An E2F1-dependent gene expression program that determines the balance between proliferation and cell death. Cancer Cell 13(1):11–22
Mueller E, Sarraf P, Tontonoz P, Evans RM, Martin KJ, Zhang M et al (1998) Terminal differentiation of human breast cancer through PPAR[gamma]. Mol Cell 1(3):465–470
Massagué J (2008) TGF[beta] in cancer. Cell 134(2):215–230
Oft M, Peli J, Rudaz C, Schwarz H, Beug H, Reichmann E (1996) TGF-beta1 and Ha-Ras collaborate in modulating the phenotypic plasticity and invasiveness of epithelial tumor cells. Genes Dev 10(19):2462–2477
Danielpour D, Song K (2006) Cross-talk between IGF-I and TGF-[beta] signaling pathways. Cytokine Growth Factor Rev 17(1–2):59–74
Jassem J, Langer CL, Karp DD, Mok T, Novello S, Park K, Strausz J, Benner RJ, Green S, Gualberto A (2010) Randomized, open label, phase III trial of figitumumab in combination with paclitaxel and carboplatin versus paclitaxel and carboplatin in patients with non-small cell lung cancer (NSCLC). American Society of Clinical Oncology Annual Meeting 2010. Abstract #7500, Chicago
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mu, L., Tuck, D., Katsaros, D. et al. Favorable outcome associated with an IGF-1 ligand signature in breast cancer. Breast Cancer Res Treat 133, 321–331 (2012). https://doi.org/10.1007/s10549-012-1952-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-012-1952-5