Abstract
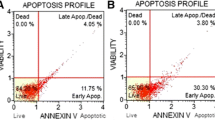
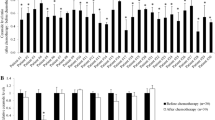
The sphingolipid ceramide is known to play a central role in chemo- and radiation-induced cell death. Acid ceramidase (AC) hydrolyzes ceramide, and thus reduces intracellular levels of this proapoptotic lipid. The role of AC as a putative anticancer target is supported by reports of upregulation in prostate cancer and in some breast tumors. In this study, we determined whether the introduction of an AC inhibitor would enhance the apoptosis-inducing effects of C6-ceramide (C6-cer) in breast cancer cells. Cultured breast cancer cells were treated with DM102 [(2R,3Z)-N-(1-hydroxyoctadec-3-en-2-yl)pivalamide, C6-cer, or the combination. Cell viability and cytotoxic synergy were assessed. Activation of apoptotic pathways, generation of reactive oxygen species, and mitochondrial transmembrane potential were determined. DM102 was a more effective AC inhibitor than N-oleoylethanolamine (NOE) and (1R,2R)-2-N-(tetradecanoylamino)-1-(4′-nitrophenyl)-1,3-propandiol (B-13) in MDA-MB-231, MCF-7, and BT-474 cells. As single agents, C6-cer (IC50 5–10 μM) and DM102 (IC50 20 μM) were only moderately cytotoxic in MDA-MB-231, MCF-7, and SK-BR-3 cells. Co-administration, however, produced synergistic decreases in viability (combination index <0.5) in all cell lines. Apoptosis was confirmed in MDA-MB-231 cells by detection of caspase 3 cleavage and a >3-fold increase in caspase 3/7 activation, PARP cleavage, and a >70% increase in Annexin-V positive cells. C6-cer/DM102 increased ROS levels 4-fold in MDA-MB-231 cells, shifted the ratio of Bax:Bcl-2 to >9-fold that of control cells, and resulted in mitochondrial membrane depolarization. DM102 also increased the synthesis of 3H-palmitate-labeled long-chain ceramides by 2-fold when C6-cer was present. These data support the effectiveness of targeting AC in combination with exogenous short-chain ceramide as an anticancer strategy, and warrant continued investigation into the utility of the C6-cer/DM102 drug duo in human breast cancer.








Similar content being viewed by others
References
Howlader N, Noone AM, Krapcho M, Neyman N, Aminou R, Waldron W, Altekruse SF, Kosary CL, Ruhl J, Tatalovich Z, Cho H, Mariotto A, Eisner MP, Lewis DR, Chen HS, Feuer EJ, Cronin KA, Edwards BK (eds) (2011) SEER Cancer Statistics Review, 1975-2008, National Cancer Institute, Bethesda, MD.http://seer.cancer.gov/csr/1975_2008/
Jemal A, Siegel R, Xu J, Ward E (2010) Cancer statistics, 2010. CA Cancer J Clin 60(5):277–300
Gonzalez-Angulo AM, Morales-Vasquez F, Hortobagyi GN (2007) Overview of resistance to systemic therapy in patients with breast cancer. Adv Exp Med Biol 608:1–22
Liu YY, Han TY, Giuliano AE, Cabot MC (2001) Ceramide glycosylation potentiates cellular multidrug resistance. FASEB J 15(3):719–730
Lavie Y, Cao H, Bursten SL, Giuliano AE, Cabot MC (1996) Accumulation of glucosylceramides in multidrug-resistant cancer cells. J Biol Chem 271(32):19530–19536
Liu YY, Han TY, Giuliano AE, Cabot MC (1999) Expression of glucosylceramide synthase, converting ceramide to glucosylceramide, confers adriamycin resistance in human breast cancer cells. J Biol Chem 274(2):1140–1146
Gouaze-Andersson V, Yu JY, Kreitenberg AJ, Bielawska A, Giuliano AE, Cabot MC (2007) Ceramide and glucosylceramide upregulate expression of the multidrug resistance gene MDR1 in cancer cells. Biochim Biophys Acta 1771(12):1407–1417
Ruckhaberle E, Rody A, Engels K, Gaetje R, von Minckwitz G, Schiffmann S, Grosch S, Geisslinger G, Holtrich U, Karn T, Kaufmann M (2008) Microarray analysis of altered sphingolipid metabolism reveals prognostic significance of sphingosine kinase 1 in breast cancer. Breast Cancer Res Treat 112(1):41–52
Watson C, Long JS, Orange C, Tannahill CL, Mallon E, McGlynn LM, Pyne S, Pyne NJ, Edwards J (2010) High expression of sphingosine 1-phosphate receptors, S1P1 and S1P3, sphingosine kinase 1, and extracellular signal-regulated kinase-1/2 is associated with development of tamoxifen resistance in estrogen receptor-positive breast cancer patients. Am J Pathol 177(5):2205–2215
Malavaud B, Pchejetski D, Mazerolles C, de Paiva GR, Calvet C, Doumerc N, Pitson S, Rischmann P, Cuvillier O (2010) Sphingosine kinase-1 activity and expression in human prostate cancer resection specimens. Eur J Cancer 46(18):3417–3424
Shirai K, Kaneshirao T, Wada M, Furuya H, Bielawski J, Hannun YA, Obeid L, Ogretmen B, Kawamori T (2011) A role of sphingosine kinase 1 in head & neck carcinogenesis. Cancer Prev Res (Phila) 4(3):454–462
Liu G, Zheng H, Zhang Z, Wu Z, Xiong H, Li J, Song L (2010) Overexpression of sphingosine kinase 1 is associated with salivary gland carcinoma progression and might be a novel predictive marker for adjuvant therapy. BMC Cancer 10:495
Seelan RS, Qian C, Yokomizo A, Bostwick DG, Smith DI, Liu W (2000) Human acid ceramidase is overexpressed but not mutated in prostate cancer. Genes Chromosom Cancer 29(2):137–146
Ruckhaberle E, Holtrich U, Engels K, Hanker L, Gatje R, Metzler D, Karn T, Kaufmann M, Rody A (2009) Acid ceramidase 1 expression correlates with a better prognosis in ER-positive breast cancer. Climacteric 12(6):502–513
Meacham WD, Antoon JW, Burow ME, Struckhoff AP, Beckman BS (2009) Sphingolipids as determinants of apoptosis and chemoresistance in the MCF-7 cell model system. Exp Biol Med (Maywood) 234(11):1253–1263
Holman DH, Turner LS, El-Zawahry A, Elojeimy S, Liu X, Bielawski J, Szulc ZM, Norris K, Zeidan YH, Hannun YA, Bielawska A, Norris JS (2008) Lysosomotropic acid ceramidase inhibitor induces apoptosis in prostate cancer cells. Cancer Chemother Pharmacol 61(2):231–242
Antoon JW, White MD, Slaughter EM, Driver JL, Khalili HS, Elliott S, Smith CD, Burow ME, Beckman BS (2011) Targeting NFkB mediated breast cancer chemoresistance through selective inhibition of sphingosine kinase-2. Cancer Biol Ther 11(7):678–689
Gouaze-Andersson V, Flowers M, Karimi R, Fabrias G, Delgado A, Casas J, Cabot MC (2011) Inhibition of acid ceramidase by a 2-substituted aminoethanol amide synergistically sensitizes prostate cancer cells to N-(4-hydroxyphenyl) retinamide. Prostate 71(10):1064–1073
Morales A, Paris R, Villanueva A, Llacuna L, Garcia-Ruiz C, Fernandez-Checa JC (2007) Pharmacological inhibition or small interfering RNA targeting acid ceramidase sensitizes hepatoma cells to chemotherapy and reduces tumor growth in vivo. Oncogene 26(6):905–916
Pchejetski D, Bohler T, Brizuela L, Sauer L, Doumerc N, Golzio M, Salunkhe V, Teissie J, Malavaud B, Waxman J, Cuvillier O (2010) FTY720 (fingolimod) sensitizes prostate cancer cells to radiotherapy by inhibition of sphingosine kinase-1. Cancer Res 70(21):8651–8661
Liu YY, Han TY, Yu JY, Bitterman A, Le A, Giuliano AE, Cabot MC (2004) Oligonucleotides blocking glucosylceramide synthase expression selectively reverse drug resistance in cancer cells. J Lipid Res 45(5):933–940
Sun Y, Zhang T, Gao P, Meng B, Gao Y, Wang X, Zhang J, Wang H, Wu X, Zheng W, Zhou G (2010) Targeting glucosylceramide synthase downregulates expression of the multidrug resistance gene MDR1 and sensitizes breast carcinoma cells to anticancer drugs. Breast Cancer Res Treat 121(3):591–599
Selzner M, Bielawska A, Morse MA, Rudiger HA, Sindram D, Hannun YA, Clavien PA (2001) Induction of apoptotic cell death and prevention of tumor growth by ceramide analogues in metastatic human colon cancer. Cancer Res 61(3):1233–1240
Stover T, Kester M (2003) Liposomal delivery enhances short-chain ceramide-induced apoptosis of breast cancer cells. J Pharmacol Exp Ther 307(2):468–475
Stover TC, Sharma A, Robertson GP, Kester M (2005) Systemic delivery of liposomal short-chain ceramide limits solid tumor growth in murine models of breast adenocarcinoma. Clin Cancer Res 11(9):3465–3474
Zolnik BS, Stern ST, Kaiser JM, Heakal Y, Clogston JD, Kester M, McNeil SE (2008) Rapid distribution of liposomal short-chain ceramide in vitro and in vivo. Drug Metab Dispos 36(8):1709–1715
Antoon JW, Liu J, Gestaut MM, Burow ME, Beckman BS, Foroozesh M (2009) Design, synthesis, and biological activity of a family of novel ceramide analogues in chemoresistant breast cancer cells. J Med Chem 52(18):5748–5752
Bektas M, Jolly PS, Muller C, Eberle J, Spiegel S, Geilen CC (2005) Sphingosine kinase activity counteracts ceramide-mediated cell death in human melanoma cells: role of Bcl-2 expression. Oncogene 24(1):178–187
Mahdy AE, Cheng JC, Li J, Elojeimy S, Meacham WD, Turner LS, Bai A, Gault CR, McPherson AS, Garcia N, Beckham TH, Saad A, Bielawska A, Bielawski J, Hannun YA, Keane TE, Taha MI, Hammouda HM, Norris JS, Liu X (2009) Acid ceramidase upregulation in prostate cancer cells confers resistance to radiation: AC inhibition, a potential radiosensitizer. Mol Ther 17(3):430–438
Yang F, Foekens JA, Yu J, Sieuwerts AM, Timmermans M, Klijn JG, Atkins D, Wang Y, Jiang Y (2006) Laser microdissection and microarray analysis of breast tumors reveal ER-alpha related genes and pathways. Oncogene 25(9):1413–1419
Elojeimy S, Liu X, McKillop JC, El-Zawahry AM, Holman DH, Cheng JY, Meacham WD, Mahdy AE, Saad AF, Turner LS, Cheng J, Day TA, Dong JY, Bielawska A, Hannun YA, Norris JS (2007) Role of acid ceramidase in resistance to FasL: therapeutic approaches based on acid ceramidase inhibitors and FasL gene therapy. Mol Ther 15(7):1259–1263
Maeda I, Takano T, Matsuzuka F, Maruyama T, Higashiyama T, Liu G, Kuma K, Amino N (1999) Rapid screening of specific changes in mRNA in thyroid carcinomas by sequence specific-differential display: decreased expression of acid ceramidase mRNA in malignant and benign thyroid tumors. Int J Cancer 81(5):700–704
Musumarra G, Barresi V, Condorelli DF, Scire S (2003) A bioinformatic approach to the identification of candidate genes for the development of new cancer diagnostics. Biol Chem 384(2):321–327
Lee SC, Xu X, Lim YW, Iau P, Sukri N, Lim SE, Yap HL, Yeo WL, Tan P, Tan SH, McLeod H, Goh BC (2009) Chemotherapy-induced tumor gene expression changes in human breast cancers. Pharmacogenet Genomics 19(3):181–192
Hara S, Nakashima S, Kiyono T, Sawada M, Yoshimura S, Iwama T, Banno Y, Shinoda J, Sakai N (2004) p53-Independent ceramide formation in human glioma cells during gamma-radiation-induced apoptosis. Cell Death Differ 11(8):853–861
Ray PS, Gouaze-Andersson V, Giuliano AE, Cabot MC (2009) Pathologic response to neoadjuvant chemotherapy for breast cancer. Linked to enzymes that metabolize ceramide? In: Proceedings of the American Association for Cancer Research. AACR, Denver, CO
Juul N, Szallasi Z, Eklund AC, Li Q, Burrell RA, Gerlinger M, Valero V, Andreopoulou E, Esteva FJ, Symmans WF, Desmedt C, Haibe-Kains B, Sotiriou C, Pusztai L, Swanton C (2010) Assessment of an RNA interference screen-derived mitotic and ceramide pathway metagene as a predictor of response to neoadjuvant paclitaxel for primary triple-negative breast cancer: a retrospective analysis of five clinical trials. Lancet Oncol 11(4):358–365
Bielawska A, Greenberg MS, Perry D, Jayadev S, Shayman JA, McKay C, Hannun YA (1996) (1S, 2R)-d-erythro-2-(N-myristoylamino)-1-phenyl-1-propanol as an inhibitor of ceramidase. J Biol Chem 271(21):12646–12654
Bedia C, Casas J, Garcia V, Levade T, Fabrias G (2007) Synthesis of a novel ceramide analogue and its use in a high-throughput fluorogenic assay for ceramidases. Chembiochem 8(6):642–648
Mao CQ, Du JZ, Sun TM, Yao YD, Zhang PZ, Song EW, Wang J (2011) A biodegradable amphiphilic and cationic triblock copolymer for the delivery of siRNA targeting the acid ceramidase gene for cancer therapy. Biomaterials 32(11):3124–3133
Samsel L, Zaidel G, Drumgoole HM, Jelovac D, Drachenberg C, Rhee JG, Brodie AM, Bielawska A, Smyth MJ (2004) The ceramide analog, B13, induces apoptosis in prostate cancer cell lines and inhibits tumor growth in prostate cancer xenografts. Prostate 58(4):382–393
Kanto T, Kalinski P, Hunter OC, Lotze MT, Amoscato AA (2001) Ceramide mediates tumor-induced dendritic cell apoptosis. J Immunol 167(7):3773–3784
Bedia C, Canals D, Matabosch X, Harrak Y, Casas J, Llebaria A, Delgado A, Fabrias G (2008) Cytotoxicity and acid ceramidase inhibitory activity of 2-substituted aminoethanol amides. Chem Phys Lipids 156(1–2):33–40
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37(8):911–917
Chou TC, Talalay P (1984) Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzym Regul 22:27–55
Vandenabeele P, Vanden Berghe T, Festjens N (2006) Caspase inhibitors promote alternative cell death pathways. Sci STKE 2006(356):pe44
Valerie K, Yacoub A, Hagan MP, Curiel DT, Fisher PB, Grant S, Dent P (2007) Radiation-induced cell signaling: inside-out and outside-in. Mol Cancer Ther 6(3):789–801
Chen Y, Jungsuwadee P, Vore M, Butterfield DA, St Clair DK (2007) Collateral damage in cancer chemotherapy: oxidative stress in nontargeted tissues. Mol Interv 7(3):147–156
Scatena R, Bottoni P, Botta G, Martorana GE, Giardina B (2007) The role of mitochondria in pharmacotoxicology: a reevaluation of an old, newly emerging topic. Am J Physiol Cell Physiol 293(1):C12–C21
Le Bras M, Clement MV, Pervaiz S, Brenner C (2005) Reactive oxygen species and the mitochondrial signaling pathway of cell death. Histol Histopathol 20(1):205–219
Kong JY, Rabkin SW (2003) Mitochondrial effects with ceramide-induced cardiac apoptosis are different from those of palmitate. Arch Biochem Biophys 412(2):196–206
Matsunaga T, Kotamraju S, Kalivendi SV, Dhanasekaran A, Joseph J, Kalyanaraman B (2004) Ceramide-induced intracellular oxidant formation, iron signaling, and apoptosis in endothelial cells: protective role of endogenous nitric oxide. J Biol Chem 279(27):28614–28624
Ogretmen B, Pettus BJ, Rossi MJ, Wood R, Usta J, Szulc Z, Bielawska A, Obeid LM, Hannun YA (2002) Biochemical mechanisms of the generation of endogenous long chain ceramide in response to exogenous short chain ceramide in the A549 human lung adenocarcinoma cell line. Role for endogenous ceramide in mediating the action of exogenous ceramide. Biol Chem 277(15):12960–12969
Barth BM, Cabot MC, Kester M (2011) Ceramide-based therapeutics for the treatment of cancer. Anticancer Agents Med Chem [Epub ahead of print]
Hannun YA, Luberto C (2000) Ceramide in the eukaryotic stress response. Trends Cell Biol 10(2):73–80
Jayadev S, Liu B, Bielawska AE, Lee JY, Nazaire F, Pushkareva M, Obeid LM, Hannun YA (1995) Role for ceramide in cell cycle arrest. J Biol Chem 270(5):2047–2052
Fillet M, Bentires-Alj M, Deregowski V, Greimers R, Gielen J, Piette J, Bours V, Merville MP (2003) Mechanisms involved in exogenous C2- and C6-ceramide-induced cancer cell toxicity. Biochem Pharmacol 65(10):1633–1642
Lucci A, Han TY, Liu YY, Giuliano AE, Cabot MC (1999) Modification of ceramide metabolism increases cancer cell sensitivity to cytotoxics. Int J Oncol 15(3):541–546
Mehta S, Blackinton D, Omar I, Kouttab N, Myrick D, Klostergaard J, Wanebo H (2000) Combined cytotoxic action of paclitaxel and ceramide against the human Tu138 head and neck squamous carcinoma cell line. Cancer Chemother Pharmacol 46(2):85–92
Chapman JV, Gouaze-Andersson V, Messner MC, Flowers M, Karimi R, Kester M, Barth BM, Liu X, Liu YY, Giuliano AE, Cabot MC (2010) Metabolism of short-chain ceramide by human cancer cells–implications for therapeutic approaches. Biochem Pharmacol 80(3):308–315
Jaffrezou JP, Maestre N, de Mas-Mansat V, Bezombes C, Levade T, Laurent G (1998) Positive feedback control of neutral sphingomyelinase activity by ceramide. FASEB J 12(11):999–1006
Mancinetti A, Di Bartolomeo S, Spinedi A (2009) Long-chain ceramide produced in response to N-hexanoylsphingosine does not induce apoptosis in CHP-100 cells. Lipids 44(11):1039–1046
Charles AG, Han TY, Liu YY, Hansen N, Giuliano AE, Cabot MC (2001) Taxol-induced ceramide generation and apoptosis in human breast cancer cells. Cancer Chemother Pharmacol 47(5):444–450
Nehra R, Riggins RB, Shajahan AN, Zwart A, Crawford AC, Clarke R (2010) BCL2 and CASP8 regulation by NF-kappaB differentially affect mitochondrial function and cell fate in antiestrogen-sensitive and -resistant breast cancer cells. FASEB J 24(6):2040–2055
Engel LW, Young NA (1978) Human breast carcinoma cells in continuous culture: a review. Cancer Res 38(11 Pt 2):4327–4339
Lacroix M, Leclercq G (2004) Relevance of breast cancer cell lines as models for breast tumours: an update. Breast Cancer Res Treat 83(3):249–289
Neve RM, Chin K, Fridlyand J, Yeh J, Baehner FL, Fevr T, Clark L, Bayani N, Coppe JP, Tong F, Speed T, Spellman PT, DeVries S, Lapuk A, Wang NJ, Kuo WL, Stilwell JL, Pinkel D, Albertson DG, Waldman FM, McCormick F, Dickson RB, Johnson MD, Lippman M, Ethier S, Gazdar A, Gray JW (2006) A collection of breast cancer cell lines for the study of functionally distinct cancer subtypes. Cancer Cell 10(6):515–527
Acknowledgments
This research was supported by the Association for Breast and Prostate Cancer Studies (Los Angeles), the Fashion Footwear Association of New York Charitable Foundation (New York, NY), National Institute of General Medical Sciences (grant no. GM77391), and the Spanish Ministerio de Ciencia e Innovación (Grant no. SAF2008-00706). We thank Matthew Bush for compiling the typescript and creating the figures.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Flowers, M., Fabriás, G., Delgado, A. et al. C6-Ceramide and targeted inhibition of acid ceramidase induce synergistic decreases in breast cancer cell growth. Breast Cancer Res Treat 133, 447–458 (2012). https://doi.org/10.1007/s10549-011-1768-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-011-1768-8