Abstract
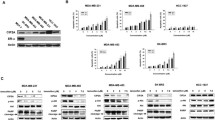
Antiestrogen therapies arrest susceptible estrogen receptor (ER)-positive breast cancers by increasing p27. Since Src phosphorylates p27 to promote p27 proteolysis, Src activation observed in up to 40% of ER-positive cancers may contribute to antiestrogen resistance. In this article, we show that treatment with the Src-inhibitor saracatinib (AZD0530) together with ER-blocking drugs increased breast cancer cell cycle arrest via p27. Saracatinib and fulvestrant together more effectively increased p27, reduced Ki67, and impaired MDA-MB-361 xenograft tumor growth in vivo than either of the drugs alone. In contrast, saracatinib monotherapy rapidly gave rise to drug resistance. Since combined ER and Src inhibition delays development of resistance in vivo, these data support further clinical investigation of saracatinib in combination with fulvestrant for women with ER-positive breast cancer. Proteomic analysis revealed striking bypass activation of the mTOR pathway in saracatinib-resistant tumors. mTORC1 activation also arose following long-term culture of ER-positive breast cancer lines in the presence of saracatinib. These data indicate the utility of proteomic analysis of drug-resistant tumors to identify potential means of drug resistance. The use of mTOR kinase inhibitors with saracatinib may subvert drug resistance and prove to be more effective than saracatinib alone.





Similar content being viewed by others
References
Howell A, Robertson JF, Abram P, Lichinitser MR, Elledge R, Bajetta E, Watanabe T, Morris C, Webster A, Dimery I, Osborne CK (2004) Comparison of fulvestrant versus tamoxifen for the treatment of advanced breast cancer in postmenopausal women previously untreated with endocrine therapy: a multinational, double-blind, randomized trial. J Clin Oncol 22(9):1605–1613
Chia S, Gradishar W, Mauriac L, Bines J, Amant F, Federico M, Fein L, Romieu G, Buzdar A, Robertson JF, Brufsky A, Possinger K, Rennie P, Sapunar F, Lowe E, Piccart M (2008) Double-blind, randomized placebo controlled trial of fulvestrant compared with exemestane after prior nonsteroidal aromatase inhibitor therapy in postmenopausal women with hormone receptor-positive, advanced breast cancer: results from EFECT. J Clin Oncol 26(10):1664–1670
Sutherland RL, Green MD, Hall RE, Reddel RR, Taylor IW (1983) Tamoxifen induces accumulation of MCF7 human mammary carcinoma cells in the G0/G1 phase of the cell cycle. Eur J Cancer Clin Oncol 19:615–621
Cariou S, Donovan JC, Flanagan WM, Milic A, Bhattacharya N, Slingerland JM (2000) Down-regulation of p21WAF1/CIP1 or p27Kip1 abrogates antiestrogen-mediated cell cycle arrest in human breast cancer cells. Proc Natl Acad Sci USA 97(16):9042–9046
Osborne CK, Boldt DH, Clark GM, Trent JM (1983) Effects of tamoxifen on human breast cancer cell cycle kinetics: accumulation of cells in early G1 phase. Cancer Res 43:3583–3585
Watts CKW, Brady A, Sarcevic B, deFazio A, Sutherland RL (1996) Antiestrogens inhibition of cell cycle progression in breast cancer cells is associated with inhibition of cyclin-dependent kinase activity and decreased retinoblastoma protein phosphorylation. Mol Endocrinol 9:1804–1813
Nicholson RI, Francis AB, McClelland RA, Manning DL, Gee JMW (1994) Pure anti-oestrogens (ICI 164384 and ICI 182780) and breast cancer: is the attainment of complete estrogen withdrawal worthwhile? Endocr Relat Cancer 3:1–13
Henderson IC, Patek AJ (1998) The relationship between prognostic and predictive factors in the management of breast cancer. Breast Cancer Res Treat 52(1–3):261–288
Chen Y, Guggisberg N, Jorda M, Gonzalez-Angulo A, Hennessy B, Mills GB, Tan CK, Slingerland JM (2009) Combined Src and aromatase inhibition impairs human breast cancer growth in vivo and bypass pathways are activated in AZD0530-resistant tumors. Clin Cancer Res 15(10):3396–3405
Migliaccio A, DiDomenico M, Castona C, DeFalco A, Bontempo P, Nola E, Auricchio F (1996) Tyrosine kinase/p21ras/MAP-kinase pathway activation by estradiol receptor complex in MCF-7 cells. EMBO J 15:1292–1300
Collins P, Webb C (1999) Estrogen hits the surface. Nat Med 5(10):1130–1131
Castoria G, Migliaccio A, Bilancio A, Di Domenico M, de Falco A, Lombardi M, Fiorentino R, Varricchio L, Barone MV, Auricchio F (2001) PI3-kinase in concert with Src promotes the S-phase entry of oestradiol-stimulated MCF-7 cells. EMBO J 20(21):6050–6059
Migliaccio A, Pagano M, Auricchio F (1993) Immediate and transient stimulation of protein tyrosine phosphorylation by estradiol in MCF-7 cells. Oncogene 8:2183–2191
Migliaccio A, Castoria G, Di Domenico M, de Falco A, Bilancio A, Lombardi M, Barone MV, Ametrano D, Zannini MS, Abbondanza C, Auricchio F (2000) Steroid-induced androgen receptor-oestradiol receptor beta-Src complex triggers prostate cancer cell proliferation. EMBO J 19(20):5406–5417
Castoria G, Lombardi M, Barone MV, Bilancio A, Di DM, Bottero D, Vitale F, Migliaccio A, Auricchio F (2003) Androgen-stimulated DNA synthesis and cytoskeletal changes in fibroblasts by a nontranscriptional receptor action. J Cell Biol 161(3):547–556
Musgrove EA, Sutherland RL (1994) Cell cycle control by steroid hormones. Semin Cancer Biol 5(5):381–389
Belsches-Jablonski AP, Biscardi JS, Peavy DR, Tice DA, Romney DA, Parsons SJ (2001) Src family kinases and HER2 interactions in human breast cancer cell growth and survival. Oncogene 20(12):1465–1475
Hiscox S, Jordan NJ, Smith C, James M, Morgan L, Taylor KM, Green TP, Nicholson RI (2008) Dual targeting of Src and ER prevents acquired antihormone resistance in breast cancer cells. Breast Cancer Res Treat 115(1):57–67
Chu I, Sun J, Arnaout A, Kahn H, Hanna W, Narod S, Sun P, Tan CK, Hengst L, Slingerland J (2007) p27 phosphorylation by Src regulates inhibition of cyclin E-Cdk2. Cell 128(2):281–294
Grimmler M, Wang Y, Mund T, Cilensek Z, Keidel EM, Waddell MB, Jakel H, Kullmann M, Kriwacki RW, Hengst L (2007) Cdk-inhibitory activity and stability of p27Kip1 are directly regulated by oncogenic tyrosine kinases. Cell 128(2):269–280
Coleman KM, Smith CL (2001) Intracellular signaling pathways: nongenomic actions of estrogens and ligand-independent activation of estrogen receptors. Front Biosci 6:D1379–D1391
Song RX, Barnes CJ, Zhang ZG, Bao YD, Kumar R, Santen RJ (2004) The role of Shc and insulin-like growth factor 1 receptor in mediating the translocation of estrogen receptor α to the plasma membrane. Proc Natl Acad Sci USA 101(7):2076–2081
Ple PA, Green TP, Hennequin LF, Curwen J, Fennell M, Allen J, Lambert-Van Der Brempt C, Costello G (2004) Discovery of a new class of anilinoquinazoline inhibitors with high affinity and specificity for the tyrosine kinase domain of c-Src. J Med Chem 47(4):871–887
Green TP, Fennell M, Whittaker R, Curwen J, Jacobs V, Allen J, Logie A, Hargreaves J, Hickinson DM, Wilkinson RW, Elvin P, Boyer B, Carragher N, Ple PA, Bermingham A, Holdgate GA, Ward WH, Hennequin LF, Davies BR, Costello GF (2009) Preclinical anticancer activity of the potent, oral Src inhibitor AZD0530. Mol Oncol 3:248–261
Herynk MH, Beyer AR, Cui Y, Weiss H, Anderson E, Green TP, Fuqua SA (2006) Cooperative action of tamoxifen and c-Src inhibition in preventing the growth of estrogen receptor-positive human breast cancer cells. Mol Cancer Ther 5(12):3023–3031
Chu I, Arnaout A, Loiseau S, Sun J, Seth A, McMahon C, Chun K, Hennessy B, Mills GB, Nawaz Z, Slingerland JM (2007) Src promotes estrogen-dependent estrogen receptor alpha proteolysis in human breast cancer. J Clin Investig 117(8):2205–2215
Catzavelos C, Bhattacharya N, Ung YC, Wilson JA, Roncari L, Sandhu C, Shaw P, Yeger H, Morava-Protzner I, Kapusta L, Franssen E, Pritchard KI, Slingerland JM (1997) Decreased levels of the cell-cycle inhibitor p27Kip1 protein: prognostic implications in primary breast cancer. Nat Med 3(2):227–230
Tibes R, Qiu Y, Lu Y, Hennessy B, Andreeff M, Mills GB, Kornblau SM (2006) Reverse phase protein array: validation of a novel proteomic technology and utility for analysis of primary leukemia specimens and hematopoietic stem cells. Mol Cancer Ther 5(10):2512–2521
Hennessy BT, Lu YL, Poradosu E, Yu QH, Yu SX, Hall H, Carey MS, Ravoori M, Gonzalez-Angulo AM, Birch R, Henderson IC, Kundra V, Mills GB (2007) Pharmacodynamic markers of perifosine efficacy. Clin Cancer Res 13(24):7421–7431
Dings RP, Yokoyama Y, Ramakrishnan S, Griffioen AW, Mayo KH (2003) The designed angiostatic peptide anginex synergistically improves chemotherapy and antiangiogenesis therapy with angiostatin. Cancer Res 63(2):382–385
Hu J, He X, Baggerly KA, Coombes KR, Hennessy BTJ, Mills GB (2007) Non-parametric quantification of protein lysate arrays. Bioinformatics 23(15):1986–1994
Jensen EV, Jordan VC (2003) The estrogen receptor: a model for molecular medicine. Clin Cancer Res 9(6):1980–1989
Ali S, Coombes RC (2002) Endocrine-responsive breast cancer and strategies for combating resistance. Nat Rev Cancer 2(2):101–112
Clarke R, Leonessa F, Welch JN, Skaar TC (2001) Cellular and molecular pharmacology of antiestrogen action and resistance. Pharmacol Rev 53(1):25–71
Johnston SR, Martin LA, Leary A, Head J, Dowsett M (2007) Clinical strategies for rationale combinations of aromatase inhibitors with novel therapies for breast cancer. J Steroid Biochem Mol Biol 106(1–5):180–186
Johnston SR, Martin LA, Head J, Smith I, Dowsett M (2005) Aromatase inhibitors: combinations with fulvestrant or signal transduction inhibitors as a strategy to overcome endocrine resistance. J Steroid Biochem Mol Biol 95(1–5):173–181
Chu I, Blackwell K, Chen S, Slingerland J (2005) The dual ErbB1/ErbB2 inhibitor, lapatinib (GW572016), cooperates with tamoxifen to inhibit both cell proliferation- and estrogen-dependent gene expression in antiestrogen-resistant breast cancer. Cancer Res 65(1):18–25
Planas-Silva MD, Weinberg RA (1997) Estrogen-dependent cyclin E-cdk2 activation through p21 redistribution. Mol Cell Biol 17(7):4059–4069
Foster J, Wimalasen J (1996) Estrogen regulates activity of cyclin-dependent kinases and retinoblastoma protein phosphorylation in breast cancer cells. Mol Endocrinol 10:488–496
Prall OWJ, Sarcevic B, Musgrove EA, Watts CKW, Sutherland RL (1997) Estrogen induced activation of Cdk4 and Cdk2 during G1-S phase progression is accompanied by increased cyclin D1 expression and decreased cyclin dependent inhibitor association with cyclin E-Cdk2. J Biol Chem 272:10882–10894
Acknowledgments
This study was supported by grants from the Flight Attendant Medical Research Institute, and the Breast Cancer Research Foundation. YC was supported by an Avon-AACR Fellowship, and SW, NG and JMS were supported in part by the Doris Duke Charitable Foundation.
Conflicts of interest statement
The authors declared no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Chen, Y., Alvarez, E.A., Azzam, D. et al. Combined Src and ER blockade impairs human breast cancer proliferation in vitro and in vivo. Breast Cancer Res Treat 128, 69–78 (2011). https://doi.org/10.1007/s10549-010-1024-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-010-1024-7