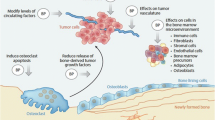
Abstract
Adjuvant therapies for early breast cancer are associated with substantial decreases in bone mineral density. Bisphosphonates are antiresorptive agents that have an established role in preventing skeletal morbidity in patients with bone metastases and in the treatment of osteoporosis. Recently, several trials have demonstrated the efficacy of bone-directed agents for prevention of cancer treatment-induced bone loss in both premenopausal and postmenopausal women with early stage breast cancer. Moreover, it is now becoming evident that bisphosphonates may also exert anticancer effects in the adjuvant setting. For example, long-term follow-up of a study in patients with bone marrow micrometastases from breast cancer revealed overall survival benefits for clodronate versus placebo, and an ongoing large trial may provide further insights. Addition of twice-yearly zoledronic acid to standard adjuvant endocrine therapy significantly improved disease-free survival and decreased disease recurrence compared with standard therapy alone in 3 clinical trials involving nearly 3,500 patients with stage I-IIIA breast cancer, and monthly zoledronic acid during neoadjuvant therapy decreased residual tumor volume and improved pathologic response in patients with stage II/III breast cancer. Overall, a large and growing body of evidence suggests the potential adjuvant benefits of bisphosphonates in early breast cancer.

Similar content being viewed by others
Abbreviations
- ABCSG-12:
-
Austrian Breast and Colorectal Cancer Study Group Trial 12
- AI:
-
Aromatase inhibitor
- AIBL:
-
Aromatase inhibitor-associated bone loss
- BC:
-
Breast cancer
- BMD:
-
Bone mineral density
- BMFS:
-
Bone-metastases-free survival
- BP:
-
Bisphosphonate
- CTIBL:
-
Cancer therapy-induced bone loss
- DFS:
-
Disease-free survival
- DTCs:
-
Disseminated tumor cells
- EBC:
-
Early breast cancer
- HR:
-
Hazard ratio
- IV:
-
Intravenous(ly)
- LS:
-
Lumbar spine
- NNT:
-
Number needed to treat
- ONJ:
-
Osteonecrosis of the jaw
- q3mo:
-
Every 3 months
- q6mo:
-
Every 6 months
- RANKL:
-
Receptor activator of nuclear factor kappa B ligand
- RFS:
-
Recurrence-free survival
- SRE:
-
Skeletal-related event
- TAM:
-
Tamoxifen
- TH:
-
Total hip
- ZOL:
-
Zoledronic acid
References
Garcia M, Jemal A, Ward EM et al (2007) Global cancer facts & figures, 2007. American Cancer Society, Atlanta
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG) (2005) Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet 365:1687–1717
Hadji P (2009) Aromatase inhibitor-associated bone loss in breast cancer patients is distinct from postmenopausal osteoporosis. Crit Rev Oncol Hematol 69:73–82
Siris ES, Chen Y-T, Abbott TA et al (2004) Bone mineral density thresholds for pharmacological intervention to prevent fractures. Arch Intern Med 164:1108–1112
Bliuc D, Nguyen ND, Milch VE et al (2009) Mortality risk associated with low-trauma osteoporotic fracture and subsequent fracture in men and women. JAMA 301:513–521
Boyle WJ, Simonet WS, Lacey DL (2003) Osteoclast differentiation and activation. Nature 423:337–342
Powles TJ, Hickish T, Kanis JA et al (1996) Effect of tamoxifen on bone mineral density measured by dual-energy X-ray absorptiometry in healthy premenopausal and postmenopausal women. J Clin Oncol 14:78–84
Sverrisdottir A, Fornander T, Jacobsson H et al (2004) Bone mineral density among premenopausal women with early breast cancer in a randomized trial of adjuvant endocrine therapy. J Clin Oncol 22:3694–3699
Vehmanen L, Elomaa I, Blomqvist C et al (2006) Tamoxifen treatment after adjuvant chemotherapy has opposite effects on bone mineral density in premenopausal patients depending on menstrual status. J Clin Oncol 24:675–680
Gnant M, Mlineritsch B, Luschin-Ebengreuth G et al (2008) Adjuvant endocrine therapy plus zoledronic acid in premenopausal women with early-stage breast cancer: 5-year follow-up of the ABCSG-12 bone-mineral density substudy. Lancet Oncol 9:840–849
Pfeilschifter J, Diel IJ (2000) Osteoporosis due to cancer treatment: pathogenesis and management. J Clin Oncol 18:1570–1593
Shapiro CL, Manola J, Leboff M (2001) Ovarian failure after adjuvant chemotherapy is associated with rapid bone loss in women with early-stage breast cancer. J Clin Oncol 19:3306–3311
Bines J, Oleske DM, Cobleigh MA (1996) Ovarian function in premenopausal women treated with adjuvant chemotherapy for breast cancer. J Clin Oncol 14:1718–1729
Swenson KK, Nissen MJ, Anderson E et al (2009) Effects of exercise vs. bisphosphonates on bone mineral density in breast cancer patients receiving chemotherapy. J Support Oncol 7:101–107
Bock O, Felsenberg D (2008) Bisphosphonates in the management of postmenopausal osteoporosis—optimizing efficacy in clinical practice. Clin Interv Aging 3:279–297
Pavlakis N, Schmidt R, Stockler M (2005) Bisphosphonates for breast cancer. Cochrane Database Syst Rev CD003474
Hadji P, Bundred N (2007) Reducing the risk of cancer treatment-associated bone loss in patients with breast cancer. Semin Oncol 34(suppl 4):S4–S10
Hershman DL, McMahon DJ, Crew KD et al (2008) Zoledronic acid prevents bone loss in premenopausal women undergoing adjuvant chemotherapy for early-stage breast cancer. J Clin Oncol 26:4739–4745
Brufsky A, Bundred N, Coleman R et al (2008) Integrated analysis of zoledronic acid for prevention of aromatase inhibitor-associated bone loss in postmenopausal women with early breast cancer receiving adjuvant letrozole. Oncologist 13:503–514
Frassoldati A, Brufsky A, Bundred N, et al (2008) The effect of zoledronic acid on aromatase inhibitor-associated bone loss in postmenopausal women with early breast cancer receiving adjuvant letrozole: 24-month integrated follow-up of the Z-FAST and ZO-FAST trials [poster]. Presented at: 33rd Congress of the European Society for Medical Oncology, Stockholm, Sweden, 12–16 September 2008 (Abstr 185PD)
Gnant M, Mlineritsch B, Schippinger W et al (2009) Endocrine therapy plus zoledronic acid in premenopausal breast cancer. N Engl J Med 360:679–691
Ellis GK, Bone HG, Chlebowski R et al (2008) Randomized trial of denosumab in patients receiving adjuvant aromatase inhibitors for nonmetastatic breast cancer. J Clin Oncol 26:4875–4882
Vehmanen L, Saarto T, Elomaa I et al (2001) Long-term impact of chemotherapy-induced ovarian failure on bone mineral density (BMD) in premenopausal breast cancer patients. The effect of adjuvant clodronate treatment. Eur J Cancer 37:2373–2378
Delmas PD, Balena R, Confravreux E et al (1997) Bisphosphonate risedronate prevents bone loss in women with artificial menopause due to chemotherapy of breast cancer: a double-blind, placebo-controlled study. J Clin Oncol 15:955–962
Hines SL, Mincey BA, Sloan JA, et al (2008) N02C1: a phase III randomized, placebo-controlled, double-blind trial of risedronate for prevention of bone loss in premenopausal women undergoing adjuvant chemotherapy for breast cancer (BC) [abstract]. J Clin Oncol 26(suppl):12s (Abstr 525)
Saarto T, Vehmanen L, Elomaa I et al (2001) The effect of clodronate and antioestrogens on bone loss associated with oestrogen withdrawal in postmenopausal women with breast cancer. Br J Cancer 84:1047–1051
Confavreux CB, Fontana A, Guastalla JP et al (2007) Estrogen-dependent increase in bone turnover and bone loss in postmenopausal women with breast cancer treated with anastrozole. Prevention with bisphosphonates. Bone 41:346–352
Greenspan SL, Brufsky A, Lembersky BC et al (2008) Risedronate prevents bone loss in breast cancer survivors: a 2-year, randomized, double-blind, placebo-controlled clinical trial. J Clin Oncol 26:2644–2652
Van Poznak C, Hannon RA, Mackey JR et al (2010) Prevention of aromatase inhibitor-induced bone loss using risedronate: the SABRE trial. J Clin Oncol 28:967–975
Singh S, Cuzick J, Edwards R, et al (2007) Effect of anastrozole on bone mineral density after one year of treatment: results from bone sub-study of the International Breast Cancer Intervention Study (IBIS-II) [abstract]. Breast Cancer Res Treat 106(suppl 1):S9 (Abstr 28)
Lester JE, Dodwell D, Purohit OP et al (2008) Prevention of anastrozole-induced bone loss with monthly oral ibandronate during adjuvant aromatase inhibitor therapy for breast cancer. Clin Cancer Res 14:6336–6342
Fuleihan GE, Salamoun M, Mourad YA et al (2005) Pamidronate in the prevention of chemotherapy-induced bone loss in premenopausal women with breast cancer: a randomized controlled trial. J Clin Endocrinol Metab 90:3209–3214
Rosen LS, Gordon DH, Dugan W Jr et al (2004) Zoledronic acid is superior to pamidronate for the treatment of bone metastases in breast carcinoma patients with at least one osteolytic lesion. Cancer 100:36–43
Shapiro CL, Halabi S, Hars V, et al (2008) Zoledronic acid (ZA) preserves bone mineral density (BMD) in premenopausal women who develop ovarian failure due to adjuvant chemotherapy: first results from CALBG trial 79809 [oral presentation]. Presented at 44th Annual Meeting of the American Society of Clinical Oncology, Chicago, IL, 30 May–3 June 2008 (Abstr 512)
Hoer A, Gol D, Gothe H et al (2005) Rates and determinants of patient persistency with oral bisphosphonates in cancer patients [abstract]. Support Care Cancer 13:428 Abstract 407-057
Ettinger MP, Gallagher R, MacCosbe PE (2006) Medication persistence with weekly versus daily doses of orally administered bisphosphonates. Endocr Pract 12:522–528
Brufsky AM, Bosserman LD, Caradonna RR et al (2009) Zoledronic acid effectively prevents aromatase inhibitor-associated bone loss in postmenopausal women with early breast cancer receiving adjuvant letrozole: Z-FAST study 36-month follow-up results. Clin Breast Cancer 9:77–85
Eidtmann H, Bundred NJ, de Boer R, et al (2009) The effect of zoledronic acid on aromatase inhibitor associated bone loss in postmenopausal women with early breast cancer receiving adjuvant letrozole: 36 months follow-up of ZO-FAST [abstract]. Cancer Res 69(suppl 2):74s (Abstr 44)
Brufsky A, Harker WG, Beck JT, et al (2009) The effect of zoledronic acid on aromatase inhibitor-associated bone loss in premenopausal women with early breast cancer receiving adjuvant letrozole: the Z-FAST study 5-year final follow-up [poster]. Presented at: 32nd Annual San Antonio Breast Cancer Symposium, San Antonio, TX, 9–13 December 2009 (Abstr 4083). Available at: http://www.posters2view.com/sabcs09/viewp.php?nu=4083. Accessed 4 February 2010
Llombart A, Frassoldati A, Paija O, et al (2009) Zoledronic acid prevents aromatase inhibitor-associated bone loss in postmenopausal women with early breast cancer receiving adjuvant letrozole: E-ZO-FAST 36-month follow up [poster]. Presented at: ASCO 2009 Breast Cancer Symposium, San Francisco, CA, 8–10 October 2009 (Abstr 213)
Hines SL, Mincey B, Dentchev T et al (2009) Immediate versus delayed zoledronic acid for prevention of bone loss in postmenopausal women with breast cancer starting letrozole after tamoxifen-N03CC. Breast Cancer Res Treat 117:603–609
Stopeck A, Body JJ, Fujiwara Y, et al (2009) Denosumab versus zoledronic acid for the treatment of breast cancer patients with bone metastases: results of a randomized phase 3 study [oral presentation]. Presented at: ECCO 15–34th ESMO Multidisciplinary Congress; Berlin, Germany; 20–24 September 2009. Abstract 2LBA
Stopeck A, de Boer R, Fujiwara Y, et al (2009) A comparison of denosumab versus zoledronic acid for the prevention of skeletal-related events in breast cancer patients with bone metastases [abstract]. Cancer Res 69(24 suppl):490s. Abstract 422
Meads MB, Hazlehurst LA, Dalton WS (2008) The bone marrow microenvironment as a tumor sanctuary and contributor to drug resistance. Clin Cancer Res 14:2519–2526
Winter MC, Holen I, Coleman RE (2008) Exploring the anti-tumour activity of bisphosphonates in early breast cancer. Cancer Treat Rev 34:453–475
Lin AY, Park JW, Scott J, et al (2008) Zoledronic acid as adjuvant therapy for women with early stage breast cancer and disseminated tumor cells in bone marrow [abstract]. J Clin Oncol 26(suppl):20s (Abstr 559)
Rack B, Schindlbeck C, Strobl B et al (2008) Efficacy of zoledronate in treating persisting isolated tumor cells in bone marrow in patients with breast cancer. A phase II pilot study. Dtsch Med Wochenschr 133:285–289
Solomayer EF, Gebauer G, Hirnle P et al (2009) Influence of zoledronic acid on disseminated tumor cells (DTC) in primary breast cancer patients [abstract]. Cancer Res 69(suppl 2):170s–171s (Abstr 2048)
Aft R, Naughton M, Trinkaus K et al (2010) Effect of zoledronic acid on disseminated tumour cells in women with locally advanced breast cancer: an open label, randomised, phase 2 trial. Lancet Oncol 11:421–428
Zaghloul MS, Boutrus R, El-Hosieny H, et al (2008) A controlled prospective randomized placebo-controlled trial of zoledronic acid in bony metastatic bladder cancer patients [abstract]. J Clin Oncol 26(suppl):257s (Abstr 5033)
Aviles A, Nambo MJ, Neri N et al (2007) Antitumor effect of zoledronic acid in previously untreated patients with multiple myeloma. Med Oncol 24:227–230
Mystakidou K, Katsouda E, Parpa E et al (2005) Randomized, open label, prospective study on the effect of zoledronic acid on the prevention of bone metastases in patients with recurrent solid tumors that did not present with bone metastases at baseline. Med Oncol 22:195–201
Santini D, Vincenzi B, Galluzzo S et al (2007) Repeated intermittent low-dose therapy with zoledronic acid induces an early, sustained, and long-lasting decrease of peripheral vascular endothelial growth factor levels in cancer patients. Clin Cancer Res 13:4482–4486
Diel IJ, Jaschke A, Solomayer EF et al (2008) Adjuvant oral clodronate improves the overall survival of primary breast cancer patients with micrometastases to the bone marrow: a long-term follow-up. Ann Oncol 19:2007–2011
Powles T, Paterson A, McCloskey E et al (2006) Reduction in bone relapse and improved survival with oral clodronate for adjuvant treatment of operable breast cancer [ISRCTN83688026]. Breast Cancer Res 8:R13
Dearnaley DP, Mason MD, Parmar MK, et al (2009) Survival benefit with oral sodium clodronate in metastatic but not localised prostate cancer: Long-term results of MRC PR04 & PR05 [abstract]. Presented at ASCO 2009 Genitourinary Cancers Symposium, Orlando, FL, 26–28 February 2009 (Abstr 6)
Saarto T, Blomqvist C, Virkkunen P et al (2001) Adjuvant clodronate treatment does not reduce the frequency of skeletal metastases in node-positive breast cancer patients: 5-year results of a randomized controlled trial. J Clin Oncol 19:10–17
Bria E, Nistico C, Cuppone F et al (2006) Benefit of taxanes as adjuvant chemotherapy for early breast cancer: pooled analysis of 15,500 patients. Cancer 106:2337–2344
Coleman RE, Winter MC, Cameron D et al (2010) The effects of adding zoledronic acid to neoadjuvant chemotherapy on tumour response: exploratory evidence for direct anti-tumour activity in breast cancer. Br J Cancer 102:1099–1105
Coleman R, Bundred NJ, De Boer R, et al (2009) Impact of zoledronic acid in postmenopausal women with early breast cancer receiving adjuvant letrozole: Z-FAST, ZO-FAST, and E-ZO-FAST [poster]. Presented at 32nd Annual San Antonio Breast Cancer Symposium, San Antonio, TX, 9–13 December 2009 (Abstr 4082). Available at: http://www.posters2view.com/sabcs09/viewp.php?nu=4082. Accessed 3 February 2010
Hillner BE, Ingle JN, Chlebowski RT et al (2003) American Society of Clinical Oncology 2003 update on the role of bisphosphonates and bone health issues in women with breast cancer. J Clin Oncol 21:4042–4057
Hadji P, Body JJ, Aapro MS et al (2008) Practical guidance for the management of aromatase inhibitor-associated bone loss. Ann Oncol 19:1407–1416
Reid DM, Doughty J, Eastell R et al (2008) Guidance for the management of breast cancer treatment-induced bone loss: a consensus position statement from a UK Expert Group. Cancer Treat Rev 34(suppl 1):S3–S18
Acknowledgments
Financial support for medical editorial assistance was provided by Novartis Pharmaceuticals. I thank Shalini Murthy, PhD, for her medical editorial assistance with this manuscript. She helped draft the manuscript according to my direction and integrated the revisions I provided. The sponsor (Novartis Pharmaceuticals) had no influence over the content of the manuscript at any stage.
Conflict of interest statement
Dr. Lipton has participated as a consultant for Amgen and Novartis; received honoraria from Amgen, Novartis, and Genentech; received research funding from Novartis, Monogram Biosciences, and Oncogene Sciences; and provided expert testimony for Novartis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lipton, A. Should bisphosphonates be utilized in the adjuvant setting for breast cancer?. Breast Cancer Res Treat 122, 627–636 (2010). https://doi.org/10.1007/s10549-010-0935-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-010-0935-7