Abstract
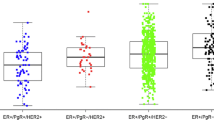
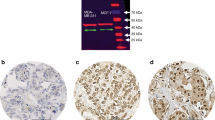
Gli1 is a transcription factor and oncogene with documented roles in the progression of several cancer types, including cancers of the skin and pancreas. The contribution of Gli1 to the progression of breast cancer is less established. In order to investigate the functional impact of Gli1 in breast cancer, expression of Gli1 and its contribution to cell growth was assessed in breast cancer cell lines. These in vitro results were compared to expression of Gli1, determined by immunohistochemistry, in 171 breast cancers. In these cancers, the association of Gli1 with expression of estrogen receptor α (ERα) and progesterone receptor (PR), ErbB2, p53, the rate of proliferation, and clinicopathologic parameters and outcome was assessed. Expression of Gli1 and ERα mRNA was strongly correlated in ERα-positive cell lines (r = 0.999). Treatment with estrogen increased expression of Gli1 in 2 of 3 ERα-positive cell lines; this increase was prevented by treatment with the ERα-specific antagonist MPP. Silencing of Gli1 by shRNA markedly reduced the survival of two ERα-negative cell lines, but caused only a modest reduction in ERα-positive cell lines. In breast cancer tissues, cancers with nuclear localization of Gli1 had a higher ERα (P=0.027) and lower p53 expression (P=0.017) than those without nuclear localization of Gli1. However, nuclear localization of Gli1 was predictive of a poorer cancer-specific survival in ERα-negative, including triple negative, cancers (P = 0.005), but not ERα-positive cancers. In conclusion, we demonstrate a positive association between expression of Gli1 and ERα; however, our data indicate a greater functional effect of Gli1 in ERα-negative cancers.





Similar content being viewed by others
References
Ruiz i Altaba A, Mas C, Stecca B (2007) The Gli code: an information nexus regulating cell fate, stemness and cancer. Trends Cell Biol 17:438–447
Fernandez-Zapico ME (2008) Primers on molecular pathways GLI: more than just hedgehog? Pancreatology 8:227–229
Lipinski RJ, Gipp JJ, Zhang J, Doles JD, Bushman W (2006) Unique and complimentary activities of the Gli transcription factors in hedgehog signaling. Exp Cell Res 312:1925–1938
Kasper M, Regl G, Frischauf AM, Aberger F (2006) GLI transcription factors: mediators of oncogenic hedgehog signalling. Eur J Cancer 42:437–445
Dennler S, Andre J, Alexaki I, Li A, Magnaldo T, ten Dijke P, Wang XJ, Verrecchia F, Mauviel A (2007) Induction of sonic hedgehog mediators by transforming growth factor-beta: Smad3-dependent activation of Gli2 and Gli1 expression in vitro and in vivo. Cancer Res 67:6981–6986
Schnidar H, Eberl M, Klingler S, Mangelberger D, Kasper M, Hauser-Kronberger C, Regl G, Kroismayr R, Moriggl R, Sibilia M, Aberger F (2009) Epidermal growth factor receptor signaling synergizes with Hedgehog/GLI in oncogenic transformation via activation of the MEK/ERK/JUN pathway. Cancer Res 69:1284–1292
Ji Z, Mei FC, Xie J, Cheng X (2007) Oncogenic KRAS activates hedgehog signaling pathway in pancreatic cancer cells. J Biol Chem 282:14048–14055
Stecca B, Mas C, Clement V, Zbinden M, Correa R, Piguet V, Beermann F, Ruiz IAA (2007) Melanomas require HEDGEHOG-GLI signaling regulated by interactions between GLI1 and the RAS-MEK/AKT pathways. Proc Natl Acad Sci USA 104:5895–5900
Ruppert JM, Vogelstein B, Kinzler KW (1991) The zinc finger protein GLI transforms primary cells in cooperation with adenovirus E1A. Mol Cell Biol 11:1724–1728
Pasca di Magliano M, Hebrok M (2003) Hedgehog signalling in cancer formation and maintenance. Nat Rev 3:903–911
Berman DM, Karhadkar SS, Maitra A, Montes De Oca R, Gerstenblith MR, Briggs K, Parker AR, Shimada Y, Eshleman JR, Watkins DN, Beachy PA (2003) Widespread requirement for hedgehog ligand stimulation in growth of digestive tract tumours. Nature 425:846–851
Karhadkar SS, Steven Bova G, Abdallah N, Dhara S, Gardner D, Maitra A, Isaacs JT, Berman DM, Beachy PA (2004) Hedgehog signalling in prostate regeneration, neoplasia and metastasis. Nature 431:707–712
Kayed H, Kleeff J, Keleg S, Guo J, Ketterer K, Berberat PO, Giese N, Esposito I, Giese T, Buchler MW, Friess H (2004) Indian hedgehog signaling pathway: expression and regulation in pancreatic cancer. Int J Cancer 110:668–676
Watkins DN, Berman DM, Burkholder SG, Wang B, Beachy PA, Baylin SB (2003) Hedgehog signalling within airway epithelial progenitors and in small-cell lung cancer. Nature 422:313–317
Thayer SP, di Magliano MP, Heiser PW, Nielsen CM, Roberts DJ, Lauwers GY, Qi YP, Gysin S, Fernandez-del Castillo C, Yajnik V, Antoniu B, McMahon M, Warshaw AL, Hebrok M (2003) Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature 425:851–856
Wolf I, Bose S, Desmond JC, Lin BT, Williamson EA, Karlan BY, Koeffler HP (2007) Unmasking of epigenetically silenced genes reveals DNA promoter methylation and reduced expression of PTCH in breast cancer. Breast Cancer Res Treat 105(2):139–155
Ma X, Chen K, Huang S, Zhang X, Adegboyega PA, Evers BM, Zhang H, Xie J (2005) Frequent activation of the hedgehog pathway in advanced gastric adenocarcinomas. Carcinogenesis 26:1698–1705
Ma X, Sheng T, Zhang Y, Zhang X, He J, Huang S, Chen K, Sultz J, Adegboyega PA, Zhang H, Xie J (2005) Hedgehog signaling is activated in subsets of esophageal cancers. Int J Cancer 118(1):139–148
Qualtrough D, Buda A, Gaffield W, Williams AC, Paraskeva C (2004) Hedgehog signalling in colorectal tumour cells: induction of apoptosis with cyclopamine treatment. Int J Cancer 110:831–837
Das S, Harris LG, Metge BJ, Liu S, Riker AI, Samant RS, Shevde LA (2009) The hedgehog pathway transcription factor, GLI1 promotes malignant behavior of cancer cells by upregulating osteopontin. J Biol Chem 284(34):22888–22897
Mukherjee S, Frolova N, Sadlonova A, Novak Z, Steg A, Page GP, Welch DR, Lobo-Ruppert SM, Ruppert JM, Johnson MR, Frost AR (2006) Hedgehog signaling and response to cyclopamine differs in epithelial and stromal cells in benign breast and breast cancer. Cancer Biol Ther 5:674–683
Zhang X, Harrington N, Moraes RC, Wu MF, Hilsenbeck SG, Lewis MT (2009) Cyclopamine inhibition of human breast cancer cell growth independent of smoothened (Smo). Breast Cancer Res Treat 115(3):505–521
Kubo M, Nakamura M, Tasaki A, Yamanaka N, Nakashima H, Nomura M, Kuroki S, Katano M (2004) Hedgehog signaling pathway is a new therapeutic target for patients with breast cancer. Cancer Res 64:6071–6074
Hatsell S, Frost AR (2007) Hedgehog signaling in mammary gland development and breast cancer. J Mammary Gland Biol Neoplasia 12:163–173
Liu S, Dontu G, Mantle ID, Patel S, Ahn NS, Jackson KW, Suri P, Wicha MS (2006) Hedgehog signaling and Bmi-1 regulate self-renewal of normal and malignant human mammary stem cells. Cancer Res 66:6063–6071
Neve RM, Chin K, Fridlyand J, Yeh J, Baehner FL, Fevr T, Clark L, Bayani N, Coppe JP, Tong F, Speed T, Spellman PT, DeVries S, Lapuk A, Wang NJ, Kuo WL, Stilwell JL, Pinkel D, Albertson DG, Waldman FM, McCormick F, Dickson RB, Johnson MD, Lippman M, Ethier S, Gazdar A, Gray JW (2006) A collection of breast cancer cell lines for the study of functionally distinct cancer subtypes. Cancer Cell 10:515–527
Cailleau R, Olive M, Cruciger QV (1978) Long-term human breast carcinoma cell lines of metastatic origin: preliminary characterization. In vitro 14:911–915
Hackett AJ, Smith HS, Springer EL, Owens RB, Nelson-Rees WA, Riggs JL, Gardner MB (1977) Two syngeneic cell lines from human breast tissue: the aneuploid mammary epithelial (hs578t) and the diploid myoepithelial (hs578bst) cell lines. J Natl Cancer Inst 58:1795–1806
Lacroix M, Leclercq G (2004) Relevance of breast cancer cell lines as models for breast tumours: an update. Breast Cancer Res Treat 83:249–289
Blick T, Widodo E, Hugo H, Waltham M, Lenburg ME, Neve RM, Thompson EW (2008) Epithelial mesenchymal transition traits in human breast cancer cell lines. Clin Exp Metastasis 25:629–642
Guise TA, Yin JJ, Mohammad KS (2003) Role of endothelin-1 in osteoblastic bone metastases. Cancer 97:779–784
Yoneda T, Michigami T, Yi B, Williams PJ, Niewolna M, Hiraga T (2000) Actions of bisphosphonate on bone metastasis in animal models of breast carcinoma. Cancer 88:2979–2988
Zhang RD, Fidler IJ, Price JE (1991) Relative malignant potential of human breast carcinoma cell lines established from pleural effusions and a brain metastasis. Invasion Metastasis 11:204–215
Li X, Deng W, Nail CD, Bailey SK, Kraus MH, Ruppert JM, Lobo-Ruppert SM (2006) Snail induction is an early response to Gli1 that determines the efficiency of epithelial transformation. Oncogene 25:609–621
Yanai K, Nagai S, Wada J, Yamanaka N, Nakamura M, Torata N, Noshiro H, Tsuneyoshi M, Tanaka M, Katano M (2007) Hedgehog signaling pathway is a possible therapeutic target for gastric cancer. J Surg Oncol 95:55–62
Kayed H, Kleeff J, Keleg S, Buchler MW, Friess H (2003) Distribution of Indian hedgehog and its receptors patched and smoothened in human chronic pancreatitis. J Endocrinol 178:467–478
Thomas MK, Rastalsky N, Lee JH, Habener JF (2000) Hedgehog signaling regulation of insulin production by pancreatic beta-cells. Diabetes 49:2039–2047
Talley LI, Grizzle WE, Waterbor JW, Brown D, Weiss H, Frost AR (2002) Hormone receptors and proliferation in breast carcinomas of equivalent histologic grades in pre- and postmenopausal women. Int J Cancer 98:118–127
Talley L, Chhieng DC, Bell WC, Grizzle WE, Frost AR (2008) Immunohistochemical detection of egfr, p185(erbb-2), bcl-2 and p53 in breast carcinomas in pre-menopausal and post-menopausal women. Biotech Histochem 83:5–14
Tapia C, Glatz K, Novotny H, Lugli A, Horcic M, Seemayer CA, Tornillo L, Terracciano L, Spichtin H, Mirlacher M, Simon R, Sauter G (2007) Close association between HER-2 amplification and overexpression in human tumors of non-breast origin. Mod Pathol 20:192–198
Kaplan E, Meier P (1958) Nonparametric estimation from incomplete observations. J Am Stat Assoc 53:457–481
Katayama S, Ashizawa K, Gohma H, Fukuhara T, Narumi K, Tsuzuki Y, Tatemoto H, Nakada T, Nagai K (2006) The expression of hedgehog genes (ihh, dhh) and hedgehog target genes (ptc1, gli1, coup-tfii) is affected by estrogenic stimuli in the uterus of immature female rats. Toxicol Appl Pharmacol 217:375–383
Koga K, Nakamura M, Nakashima H, Akiyoshi T, Kubo M, Sato N, Kuroki S, Nomura M, Tanaka M, Katano M (2008) Novel link between estrogen receptor alpha and hedgehog pathway in breast cancer. Anticancer Res 28:731–740
Nolan-Stevaux O, Lau J, Truitt ML, Chu GC, Hebrok M, Fernandez-Zapico ME, Hanahan D (2009) GLI1 is regulated through smoothened-independent mechanisms in neoplastic pancreatic ducts and mediates PDAC cell survival and transformation. Genes Dev 23:24–36
Kameda C, Tanaka H, Yamasaki A, Nakamura M, Koga K, Sato N, Kubo M, Kuroki S, Tanaka M, Katano M (2009) The hedgehog pathway is a possible therapeutic target for patients with estrogen receptor-negative breast cancer. Anticancer Res 29:871–879
Munro AJ, Lain S, Lane DP (2005) P53 abnormalities and outcomes in colorectal cancer: a systematic review. Br J Cancer 92:434–444
Payne SJ, Bowen RL, Jones JL, Wells CA (2008) Predictive markers in breast cancer—the present. Histopathology 52:82–90
Helin ML, Helle MJ, Helin HJ, Isola JJ (1989) Proliferative activity and steroid receptors determined by immunohistochemistry in adjacent frozen sections of 102 breast carcinomas. Arch Pathol Lab Med 113:854–857
Zhao J, Chen G, Cao D, Li Y, Diao F, Cai H, Jin Y, Lu J (2009) Expression of gli1 correlates with the transition of breast cancer cells to estrogen-independent growth. Breast Cancer Res Treat. doi:10.1007/s10549-009-0323-3
Keshamouni VG, Mattingly RR, Reddy KB (2002) Mechanism of 17-beta-estradiol-induced Erk1/2 activation in breast cancer cells. A role for HER2 and PKC-delta. J Biol Chem 277:22558–22565
Tanaka H, Nakamura M, Kameda C, Kubo M, Sato N, Kuroki S, Tanaka M, Katano M (2009) The Hedgehog signaling pathway plays an essential role in maintaining the cd44+cd24− /low subpopulation and the side population of breast cancer cells. Anticancer Res 29:2147–2157
Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF (2003) Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci USA 100:3983–3988
Bartkowiak K, Wieczorek M, Buck F, Harder S, Moldenhauer J, Effenberger KE, Pantel K, Peter-Katalinic J, Brandt BH (2009) Two-dimensional differential gel electrophoresis of a cell line derived from a breast cancer micrometastasis revealed a stem/progenitor cell protein profile. J Proteome Res 8(4):2004–2014
Charafe-Jauffret E, Ginestier C, Iovino F, Wicinski J, Cervera N, Finetti P, Hur MH, Diebel ME, Monville F, Dutcher J, Brown M, Viens P, Xerri L, Bertucci F, Stassi G, Dontu G, Birnbaum D, Wicha MS (2009) Breast cancer cell lines contain functional cancer stem cells with metastatic capacity and a distinct molecular signature. Cancer Res 69:1302–1313
Hurt EM, Farrar WL (2008) Cancer stem cells: the seeds of metastasis? Mol Interv 8:140–142
Honeth G, Bendahl PO, Ringner M, Saal LH, Gruvberger-Saal SK, Lovgren K, Grabau D, Ferno M, Borg A, Hegardt C (2008) The cd44+/cd24− phenotype is enriched in basal-like breast tumors. Breast Cancer Res 10:R53
Nakshatri H, Srour EF, Badve S (2009) Breast cancer stem cells and intrinsic subtypes: controversies rage on. Curr Stem Cell Res Ther 4:50–60
Rakha EA, Ellis IO (2009) Triple-negative/basal-like breast cancer: review. Pathology 41:40–47
Fiaschi M, Rozell B, Bergstrom A, Toftgard R (2009) Development of mammary tumors by conditional expression of GLI1. Cancer Res 69:4810–4817
Stecca B, Ruiz i Altaba A (2009) A GLI1-p53 inhibitory loop controls neural stem cell and tumour cell numbers. EMBO J 28:663–676
Abe Y, Oda-Sato E, Tobiume K, Kawauchi K, Taya Y, Okamoto K, Oren M, Tanaka N (2008) Hedgehog signaling overrides p53-mediated tumor suppression by activating Mdm2. Proc Natl Acad Sci USA 105:4838–4843
Acknowledgments
This study was funded by the American Cancer Society (RSG-05-207-01-TBE), the Susan G. Komen Foundation (BCTR0707453) and the National Institutes of Health/National Cancer Institute (R03CA130057). We thank Drs. Mike Ruppert, Susan Lobo-Ruppert, Danny R. Welch, and Doug Hurst for their helpful advice and discussion. We also thank Dr. Mike Ruppert for the use of the Gli1 rabbit antibody and viral packaging vectors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Lusheng Xu and Yeon-Jin Kwon have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xu, L., Kwon, YJ., Frolova, N. et al. Gli1 promotes cell survival and is predictive of a poor outcome in ERα-negative breast cancer. Breast Cancer Res Treat 123, 59–71 (2010). https://doi.org/10.1007/s10549-009-0617-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-009-0617-5