Abstract
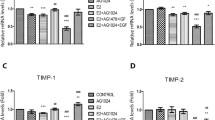
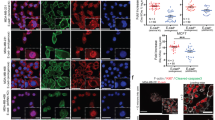
In the present study, we evaluated the ability of 4-hydroxytamoxifen (OHT) and epidermal growth factor (EGF) to regulate homotypic adhesion in MCF7 breast cancer cells. Our results demonstrate that OHT and EGF activate the E-cadherin promoter, increase E-cadherin mRNA and protein expression and enhance homotypic aggregation of MCF7 cells. Interestingly, an ERα and EGFR cross-talk is involved in the E-cadherin expression by OHT and EGF, as demonstrated by knocking down either receptor. On the basis of our findings, the well-established cross-talk between ERα and EGFR could be extended to the modulation of E-cadherin expression by OHT and EGF. Thus, the potential ability of tamoxifen to induce cell–cell aggregation may contribute to the biologic response of pharmacologic intervention in patients with breast cancer.







Similar content being viewed by others
References
Smigal C, Jemal A, Ward E et al (2006) Trends in breast cancer by race, ethnicity: update 2006. CA Cancer J Clin 56(3):168–183
Bray F, McCarron P, Parkin DM (2004) The changing global patterns of female breast cancer incidence and mortality. Breast Cancer Res 6:229–239
Jemal A, Thomas A, Murray T et al (2002) Cancer statistics, 2002. CA Cancer J Clin 52:23–47
Ishii Y, Waxman S, Germain D (2008) Tamoxifen stimulates the growth of cyclin D1–overexpressing breast cancer cells by promoting the activation of signal transducer and activator of transcription 3. Cancer Res 68(3):852–860
Giguere V, Tremblay A, Tremblay GB (1998) Estrogen receptor beta: re-evaluation of estrogen and antiestrogen signaling. Steroids 63(5–6):335–339
Mc Kenna NJ, O’ Malley BW (2002) Combinatorial control of gene expression by nuclear receptors and coregulators. Cell 108:465–474
Cenni B, Picard D (1999) Ligand-independent activation of steroid receptors: new roles for old players. Trends Endocrinol Metab 10:41–46
Weigel NL, Zhang Y (1998) Ligand-independent activation of steroid hormone receptors. J Mol Med 76:469–479
Bunone G, Briand PA, Miksicek RJ et al (1996) Activation of the unliganded estrogen receptor by EGF involves the MAP kinase pathway and direct phosphorylation. EMBO J 15(9):2174–2183
Feng W, Webb P, Nguyen P et al (2001) Potentiation of estrogen receptor activation function 1 (AF-1) by Src/JNK through a serine 118-independent pathway. Mol Endocrinol 15:32–45
Lavinsky RM, Jepsen K, Heinzel T et al (1998) Diverse signaling pathways modulate nuclear receptor recruitment of N-CoR and SMRT complexes. Proc Natl Acad Sci USA 95:2920–2925
Lee H, Jiang F, Wang Q et al (2000) MEKK1 activation of human estrogen receptor alpha and stimulation of the agonistic activity of 4-hydroxytamoxifen in endometrial and ovarian cancer cells. Mol Endocrinol 14:1882–1896
Jaiyesimi IA, Buzdar AU, Decker DA et al (1995) Use of tamoxifen for breast cancer: twenty-eight years later. J Clin Oncol 13:513–529
Massarweh S, Osborne CK, Creighton CJ et al (2008) Tamoxifen resistance in breast tumors is driven by growth factor receptor signaling with repression of classic estrogen receptor genomic function. Cancer Res 68(3):826–833
Lichtner RB (2003) Estrogen/EGF receptor interactions in breast cancer: rationale for new therapeutic combination strategies. Biomed Pharmacother 57(10):447–451
Kim H, Muller WJ (1999) The role of the epidermal growth factor receptor family in mammary tumorigenesis and metastasis. Exp Cell Res 253(1):78–87
Vanhaesebroeck B, Alessi DR (2000) PI3 K-PDK1 connection: more than just a road to PKB. Biochem J 346:561–576
Cantley LC (2002) The phosphoinositide 3-kinase pathway. Science 296:1655–1657
Pearson G, Robinson F, Beers Gibson T et al (2001) Mitogen-activated protein (MAP) kinase pathways: regulation and physiological functions. Endocr Rev 22:153–183
Peyssonnaux C, Eychene A (2001) The Raf/MEK/ERK pathway: new concepts of activation. Biol Cell 93:53–62
Kyriakis JM, Avruch J (2001) Mammalian mitogen-activated protein kinase signal transduction pathways activated by stress and inflammation. Physiol Rev 81:807–869
Schiff R, Massarweh S, Shou J et al (2003) Breast cancer endocrine resistance: how growth factor signaling and estrogen receptor coregulators modulate response. Clin Cancer Res 9:447S–454S
Santen RJ, Song RX, McPherson R et al (2002) The role of mitogen-activated protein (MAP) kinase in breast cancer. J Steroid Biochem Mol Biol 80:239–256
Vignon F, Bouton MM, Rochefort H (1987) Antiestrogens inhibit the mitogenic effect of growth factors on breast cancer cells in the total absence of estrogens. Biochem Biophys Res Commun 146:1502–1508
Ignar-Trowbridge DM, Nelson KG, Bidwell MC et al (1992) Coupling of dual signaling pathways: epidermal growth factor action involves the estrogen receptor. Proc Natl Acad Sci U S A 89:4658–4662
Hewitt S, Harrell JC, Korach KS (2005) Lessons in estrogen biology from knockout and transgenic animals. Annu Rev Physiol 67:285–308
Takeichi M (1991) Cadherin cell adhesion receptors as a morphogenetic regulator. Science 251:1451–1455
Takeichi M (1995) Morphogenetic roles of classic cadherins. Curr Opin Cell Biol 7:619–627
Perl AK, Wilgenbus P, Dahl U et al (1998) A causal role for E-cadherin in the transition from adenoma to carcinoma. Nature 392:190–193
Behrens J, Mareel MM, Van Roy FM et al (1989) Dissecting tumor cell invasion: epithelial cells acquire invasive properties after the loss of uvomorulin mediated cell–cell adhesion. J Cell Biol 108:2435–2447
Frixen UH, Behrens J, Sachs M et al (1991) E-cadherin mediated cell–cell adhesion prevents invasiveness of human carcinoma cells. J Cell Biol 113:173–185
Vleminckx K, Vakaet L Jr, Mareel M et al (1991) Genetic manipulation of E-cadherin expression by epithelial tumor cells reveals an invasion suppressor role. Cell 66:107–119
Dorudi S, Sheffield JP, Poulsom R et al (1993) E-cadherin expression in colorectal cancer. An immunocytochemical and in situ hybridization study. Am J Pathol 142:981–986
Mbalaviele D, Dunstan CR, Sasaki A et al (1996) E-cadherin expression in human breast cancer cells suppresses the development of osteolytic bone metastases in an experimental metastasis model. Cancer Res 56:4063–4070
Mauro L, Catalano S, Bossi G et al (2007) Evidences that leptin upregulates E-cadherin expression in breast cancer: effects on tumor growth and progression. Cancer Res 67(7):3412–3421
Tsai CN, Tsai CL, Tse KP et al (2002) The Epstein-Barr virus oncogene product, latent membrane protein 1, induces the downregulation of E-cadherin gene expression via activation of DNA methyltransferases. Proc Natl Acad Sci USA 99:10084–10089
Maggiolini M, Donzè O, Picard D (1999) A non-radioactive method for inexpensive quantitative RT-PCR. J Biol Chem 380:695–697
Morelli C, Garofalo C, Sisci D et al (2004) Nuclear insulin receptor substrate 1 interacts with estrogen receptor a at ERE promoters. Oncogene 23:7517–7526
Dangles V, Femenia F, Laine V et al (1997) Two- and three-dimensional cell structures govern epidermal growth factor survival function in human bladder carcinoma cell lines. Cancer Res 57:3360–3364
Kunz-Schughart LA, Kreutz M, Knuechel R (1998) Multicellular spheroids: a three-dimensional in vitro culture system to study tumour biology. Int J Exp Pathol 79:1–23
Santini MT, Rainaldi G (1999) Three-dimensional spheroid model in tumor biology. Pathobiology 67:148–157
Massarweh S, Schiff R (2006) Resistance to endocrine therapy in breast cancer: exploiting estrogen receptor/growth factor signaling crosstalk. Endocr Relat Cancer 13(1):S15–S24
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG) (1998) Tamoxifen for early breast cancer: an overview of the randomised trials. Lancet 351:1451–1467
Arpino G, Wiechmann L, Osborne CK et al (2008) Cross-talk between the estrogen receptor and the HER tyrosine kinase receptor family: molecular mechanism and clinical implications for endocrine therapy resistance. Endocrine Rev 29:217–233
Herynk MH, Fuqua SAW (2004) Estrogen receptor mutations in human disease. Endocrine Rev 25(6):869–898
Long B, McKibben BM, Lynch M et al (1992) Changes in epidermal growth factor receptor expression and response to ligand associated with acquired tamoxifen resistance or oestrogen independence in the ZR-75–1 human breast cancer cell line. Br J Cancer 65(6):865–869
Nicholson RI, McClelland RA, Gee JM et al (1994) Epidermal growth factor receptor expression in breast cancer: association with response to endocrine therapy. Breast Cancer Res Treat 29:117–125
Osborne CK, Shou J, Massarweh S et al (2005) Crosstalk between estrogen receptor and growth factor receptor pathways as a cause for endocrine therapy resistance in breast cancer. Clin Cancer Res 11(865s):865s–870s
Joel P, Smith J, Sturgill T et al (1998) pp90rsk1 regulates estrogen receptor-mediated transcription through phosphorylation of Ser-167. Mol Cell Biol 18:1978–1984
Gee JM, Robertson JF, Gutteridge E et al (2005) Epidermal growth factor receptor/HER2/insulin-like growth factor receptor signalling and oestrogen receptor activity in clinical breast cancer. Endocr Relat Cancer 12:S99–S111
Shou J, Massarweh S, Osborne CK et al (2004) Mechanisms of tamoxifen resistance: increased estrogen receptor-HER2/neu cross-talk in ER/HER2-positive breast cancer. J Natl Cancer Inst 96:926–935
Michalides R, Griekspoor A, Balkenende A et al (2004) Tamoxifen resistance by a conformational arrest of the estrogen receptor a after PKA activation in breast cancer. Cancer Cell Int 5:597–605
Nicholson RI, McClelland RA, Finlay P et al (1993) Relationship between EGF-R, c-erbB2 protein expression and Ki67immunostaining in breast cancer and hormone sensitivity. Eur J Cancer 29A:1018–1023
van Agthoven TT, van Agthoven TL, Portengen H et al (1992) Ectopic expression of epidermal growth factor receptors induces hormone independence in ZR-75–1 human breast cancer cells. Cancer Res 52:5082–5088
Benz CC, Scott GK, Sarup JC et al (2000) Estrogen-dependent, tamoxifen-resistant tumorigenic growth of MCF-7cells transfected with HER2/neu. Breast Cancer Res Treat 24:85–95
Reddy P, Lui L, Ren C et al (2005) Formation of E-cadherin-mediated cell–cell adhesion activates AKT and mitogen activated protein kinase via phosphatidylinositol 3 kinase and ligand-independent activation of epidermal growth factor receptor in ovarian cancer cells. Mol Endocrinol 19:2564–2578
Steinberg MS, McNutt PM (1999) Cadherins and their connections: adhesion junctions have broader functions. Curr Opin Cell Biol 11:554–560
Siitonen SM, Kononen JT, Helin HJ et al (1996) Reduced E-cadherin expression is associated with invasiveness and unfavorable prognosis in breast cancer. Am J Clin Pathol 105:394–402
Charpin C, Garcia S, Bonnier P et al (1998) Reduced E-cadherin immunohistochemical expression in node-negative breast carcinomas correlates with 10-year survival. Am J Clin Pathol 109:431–438
Heimann R, Lan F, McBride R et al (2000) Separating favorable from unfavorable prognostic markers in breast cancer: the role of E-cadherin. Cancer Res 60:298–304
Asgeirsson KS, Jonasson JG, Tryggvadottir L et al (2000) Altered expression of E-cadherin in breast cancer: patterns, mechanisms and clinical significance. Eur J Cancer 36:1098–1106
Lipponen P, Saarelainen E, Aaltomaa S et al (1994) Expression of E-cadherin (E-CD) as related to other prognostic factors and survival in breast cancer. J Pathol 174:101–109
Tan DS, Potts HW, Leong AC et al (1999) The biological and prognostic significance of cell polarity and E-cadherin in grade I infiltrating ductal carcinoma of the breast. J Pathol 189:20–22
Acknowledgments
This work was supported by MURST, Ex 60%, AIRC grants 2007, 2008, MFAG 2008.
Author information
Authors and Affiliations
Corresponding author
Additional information
Loredana Mauro and Michele Pellegrino have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Mauro, L., Pellegrino, M., Lappano, R. et al. E-cadherin mediates the aggregation of breast cancer cells induced by tamoxifen and epidermal growth factor. Breast Cancer Res Treat 121, 79–89 (2010). https://doi.org/10.1007/s10549-009-0456-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-009-0456-4