Abstract
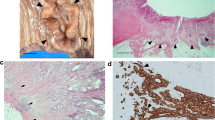
An immunohistochemical study was performed using tissue arrays and specific antibodies against MMPs -1, -2, -7, -9, -11, -13, -14, and TIMPs -1, -2 and -3. More than 5,000 determinations on cancer specimens from 124 patients with invasive breast cancer were performed at the center of the tumor and the invasive front. Immunostaining for MMPs/TIMPs by fibroblasts was evaluated. To identify specific groups of tumors with distinct expression profiles, the data obtained from both fibroblast populations were analyzed by unsupervised hierarchical cluster analysis. Intratumor stromal fibroblasts more frequently showed expression of MMP-2, -7, and -14, and TIMP-3, but less frequently of MMP-9 than fibroblasts at the invasive front. Multivariate analysis showed that a high profile of MMPs and TIMPs staining in both fibroblast populations was the most potent predictor factor of distant metastases, whereas a low staining profile in fibroblasts was associated with a low risk of metastases.





Similar content being viewed by others
References
Bhowmick NA, Moses HL (2005) Tumor–stroma interactions. Curr Opin Genet Dev 15:97–101. doi:10.1016/j.gde.2004.12.003
Kim JB, Stein R, O’Hare MJ (2005) Tumour–stromal interactions in breast cancer: the role of stroma in tumourigenesis. Tumour Biol 26:173–185. doi:10.1159/000086950
Tlsty TD, Coussens LM (2006) Tumor stroma and regulation of cancer development. Annu Rev Pathol 1:119–150. doi:10.1146/annurev.pathol.1.110304.100224
Perou CM, Sorlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA, Pollack JR, Ross DT, Johnsen H, Akslen LA et al (2000) Molecular portraits of human breast tumours. Nature 406:747–752. doi:10.1038/35021093
Sorlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H, Hastie T, Eisen MB, van de Rijn M, Jeffrey SS et al (2001) Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 98:10869–10874. doi:10.1073/pnas.191367098
Finak G, Bertos N, Pepin F, Sadekova S, Souleimanova M, Zhao H, Chen H, Omeroglu G, Meterissian S, Omeroglu A et al (2008) Stromal gene expression predicts clinical outcome in breast cancer. Nat Med 14:518–527. doi:10.1038/nm1764
West RB, Nuyten DS, Subramanian S, Nielsen TO, Corless CL, Rubin BP, Montgomery K, Zhu S, Patel R, Hernandez-Boussard T et al (2005) Determination of stromal signatures in breast carcinoma. PLoS Biol 3:e187. doi:10.1371/journal.pbio.0030187
Chang HY, Nuyten DS, Sneddon JB, Hastie T, Tibshirani R, Sorlie T, Dai H, He YD, van’t Veer LJ, Bartelink H et al (2005) Robustness, scalability, and integration of a wound-response gene expression signature in predicting breast cancer survival. Proc Natl Acad Sci USA 102:3738–3743. doi:10.1073/pnas.0409462102
Bacac M, Provero P, Mayran N, Stehle JC, Fusco C, Stamenkovic I (2006) A mouse stromal response to tumor invasion predicts prostate and breast cancer patient survival. PLoS ONE 1:e32. doi:10.1371/journal.pone.0000032
Patocs A, Zhang L, Xu Y, Weber F, Caldes T, Mutter GL, Platzer P, Eng C (2007) Breast-cancer stromal cells with TP53 mutations and nodal metastases. N Engl J Med 357:2543–2551. doi:10.1056/NEJMoa071825
Fukino K, Shen L, Patocs A, Mutter GL, Eng C (2007) Genomic instability within tumor stroma and clinicopathological characteristics of sporadic primary invasive breast carcinoma. JAMA 297:2103–2111. doi:10.1001/jama.297.19.2103
Bergamaschi A, Tagliabue E, Sorlie T, Naume B, Triulzi T, Orlandi R, Russnes HG, Nesland JM, Tammi R, Auvinen P et al (2008) Extracellular matrix signature identifies breast cancer subgroups with different clinical outcome. J Pathol 214:357–367. doi:10.1002/path.2278
Beck AH, Espinosa I, Gilks CB, van de Rijn M, West RB (2008) The fibromatosis signature defines a robust stromal response in breast carcinoma. Lab Invest 88:591–601. doi:10.1038/labinvest.2008.31
Orimo A, Gupta PB, Sgroi DC, Arenzana-Seisdedos F, Delaunay T, Naeem R, Carey VJ, Richardson AL, Weinberg RA (2005) Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 121:335–348. doi:10.1016/j.cell.2005.02.034
Karnoub AE, Dash AB, Vo AP, Sullivan A, Brooks MW, Bell GW, Richardson AL, Polyak K, Tubo R, Weinberg RA (2007) Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature 449:557–563. doi:10.1038/nature06188
Vizoso FJ, Gonzalez LO, Corte MD, Rodriguez JC, Vazquez J, Lamelas ML, Junquera S, Merino AM, Garcia-Muniz JL (2007) Study of matrix metalloproteinases and their inhibitors in breast cancer. Br J Cancer 96:903–911. doi:10.1038/sj.bjc.6603666
Egeblad M, Werb Z (2002) New functions for the matrix metalloproteinases in cancer progression. Nat Rev Cancer 2:161–174. doi:10.1038/nrc745
Noe V, Fingleton B, Jacobs K, Crawford HC, Vermeulen S, Steelant W, Bruyneel E, Matrisian LM, Mareel M (2001) Release of an invasion promoter E-cadherin fragment by matrilysin and stromelysin-1. J Cell Sci 114:111–118
Manes S, Llorente M, Lacalle RA, Gomez-Mouton C, Kremer L, Mira E, Martinez AC (1999) The matrix metalloproteinase-9 regulates the insulin-like growth factor-triggered autocrine response in DU-145 carcinoma cells. J Biol Chem 274:6935–6945. doi:10.1074/jbc.274.11.6935
Fingleton B, Vargo-Gogola T, Crawford HC, Matrisian LM (2001) Matrilysin [MMP-7] expression selects for cells with reduced sensitivity to apoptosis. Neoplasia 3:459–468. doi:10.1038/sj.neo.7900190
Stetler-Stevenson WG (1999) Matrix metalloproteinases in angiogenesis: a moving target for therapeutic intervention. J Clin Invest 103:1237–1241. doi:10.1172/JCI6870
Cornelius LA, Nehring LC, Harding E, Bolanowski M, Welgus HG, Kobayashi DK, Pierce RA, Shapiro SD (1998) Matrix metalloproteinases generate angiostatin: effects on neovascularization. J Immunol 161:6845–6852
Jiang Y, Goldberg ID, Shi YE (2002) Complex roles of tissue inhibitors of metalloproteinases in cancer. Oncogene 21:2245–2252. doi:10.1038/sj.onc.1205291
Wurtz SO, Schrohl AS, Sorensen NM, Lademann U, Christensen IJ, Mouridsen H, Brunner N (2005) Tissue inhibitor of metalloproteinases-1 in breast cancer. Endocr Relat Cancer 12:215–227. doi:10.1677/erc.1.00719
Giatromanolaki A, Sivridis E, Koukourakis MI (2004) Tumour angiogenesis: vascular growth and survival. APMIS 112:431–440. doi:10.1111/j.1600-0463.2004.apm11207-0804.x
Parker RL, Huntsman DG, Lesack DW, Cupples JB, Grant DR, Akbari M, Gilks CB (2002) Assessment of interlaboratory variation in the immunohistochemical determination of estrogen receptor status using a breast cancer tissue microarray. Am J Clin Pathol 117:723–728. doi:10.1309/PEF8-GL6F-YWMC-AG56
Gonzalez L, Corte MD, Vazquez J, Junquera S, Sanchez R, Viña A, Rodriguez J, Lamelas ML, Vizoso F (2008) Study of matrix metalloproteinases and their tissular inhibitors in ductal “in situ” carcinomas of the breast. Histophatology 53:403–415
Gonzalez LO, Pidal I, Junquera S, Corte MD, Vazquez J, Rodriguez JC, Lamelas ML, Merino AM, Garcia-Muniz JL, Vizoso FJ (2007) Overexpression of matrix metalloproteinases and their inhibitors in mononuclear inflammatory cells in breast cancer correlates with metastasis-relapse. Br J Cancer 97:957–963. doi:10.1038/sj.bjc.6603935
Eisen MB, Spellman PT, Brown PO, Botstein D (1998) Cluster analysis and display of genome-wide expression patterns. Proc Natl Acad Sci USA 95:14863–14868. doi:10.1073/pnas.95.25.14863
Sato T, Sakai T, Noguchi Y, Takita M, Hirakawa S, Ito A (2004) Tumor–stromal cell contact promotes invasion of human uterine cervical carcinoma cells by augmenting the expression and activation of stromal matrix metalloproteinases. Gynecol Oncol 92:47–56. doi:10.1016/j.ygyno.2003.09.012
Behrens P, Rothe M, Wellmann A, Krischler J, Wernert N (2001) The Ets-1 transcription factor is up-regulated together with MMP 1 and MMP 9 in the stroma of pre-invasive breast cancer. J Pathol 194:43–50. doi:10.1002/path.844
Jones JL, Walker RA (1997) Control of matrix metalloproteinase activity in cancer. J Pathol 183:377–379. doi:10.1002/(SICI)1096-9896(199712)183:4<377::AID-PATH951>3.0.CO;2-R
Liotta LA, Kohn EC (2001) The microenvironment of the tumour–host interface. Nature 411:375–379. doi:10.1038/35077241
Pellikainen JM, Ropponen KM, Kataja VV, Kellokoski JK, Eskelinen MJ, Kosma VM (2004) Expression of matrix metalloproteinase (MMP)-2 and MMP-9 in breast cancer with a special reference to activator protein-2, HER2, and prognosis. Clin Cancer Res 10:7621–7628. doi:10.1158/1078-0432.CCR-04-1061
Jiang WG, Davies G, Martin TA, Parr C, Watkins G, Mason MD, Mokbel K, Mansel RE (2005) Targeting matrilysin and its impact on tumor growth in vivo: the potential implications in breast cancer therapy. Clin Cancer Res 11:6012–6019. doi:10.1158/1078-0432.CCR-05-0275
Acknowledgments
Supported by grants from: FIS-PI040137 and PI070306, Fondo de Inversión Sanitaria del Instituto Carlos III (FIS-Spain), and Obra Social Cajastur.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Del Casar, J.M., González, L.O., Alvarez, E. et al. Comparative analysis and clinical value of the expression of metalloproteases and their inhibitors by intratumor stromal fibroblasts and those at the invasive front of breast carcinomas. Breast Cancer Res Treat 116, 39–52 (2009). https://doi.org/10.1007/s10549-009-0351-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-009-0351-z