Abstract
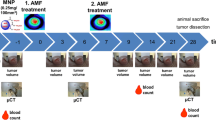
Background We have constructed anti-HER2 immunoliposomes containing magnetite nanoparticles (HML) that generate heat in an alternating magnetic field (AMF). The effective targeting and cytocidal abilities of HML have been achieved using cell culture models. This study aimed to investigate feasibility of this modality for breast cancer treatment using tumor-bearing mouse models. Material and methods The subcutaneous cancer nodules of BT474 (high HER2 expression) or SKOV3 (low HER2 expression) cells in nude mice were employed as models. HMLs were injected into these cancer nodules and were then exposed to an AMF for 30 min twice at 24 h intervals. Accumulation of magnetite and tumor growth rates were examined. Histological findings of the thermal effect were also examined. Results HMLs accumulated in only BT474 tumors. The tumor temperature increased to 45°C whereas the body temperature remained at around 38°C. Tumor regression was observed in the hyperthermic group and was sustained for 10 weeks after hyperthermia. Conclusion These results suggest that hyperthermia using HML is an effective and specific therapy for breast cancer overexpressing HER2. This therapy may provide an alternative way to treat recurrent cancer refractory to other modalities.







Similar content being viewed by others
References
van der Zee J (2002) Heating the patient: a promising approach? Ann Oncol 13:1173–1184
Moroz P, Jones SK, Gray BN (2002) Magnetically mediated hyperthermia: current status and future directions. Int J Hyperthermia 18:267–284
Hand JW, Vernon CC, Prior MV, Gardner RA, Vargas HI, Block JB, Vogel CL, Fenn AJ, Kuehl GV, Doval M (1992) Early experience of a commercial scanned focused ultrasound hyperthermia system. Focused microwave phased array thermotherapy for primary breast cancer. Int J Hyperthermia 8:587–607
Gardner RA, Vargas HI, Block JB, Vogel CL, Fenn AJ, Kuehl GV, Doval M (2002) Focused microwave phased array thermotherapy for primary breast cancer. Ann Surg Oncol 9:326–332
Abe M, Hiraoka M, Takahashi M, Egawa S, Matsuda C, Onoyama Y, Morita K, Kakehi M, Sugahara T (1986) Multi-institutional studies on hyperthermia using an 8-MHz radiofrequency capacitive heating device (Thermotron RF-8) in combination with radiation for cancer therapy. Cancer 58:1589–1595
Oura S, Tamaki T, Hirai I, Yoshimasu T, Ohta F, Nakamura R, Okamura Y (2007) Radiofrequency ablation therapy in patients with breast cancers two centimeters or less in size. Breast Cancer 14:48–54
Wu F, Wang ZB, Cao YD, Chen WZ, Bai J, Zou JZ, Zhu H (2003) A randomised clinical trial of high-intensity focused ultrasound ablation for the treatment of patients with localised breast cancer. Br J Cancer 89:2227–2233
Jordan A, Wust P, Fahling H, John W, Hinz A, Felix R (1993) Inductive heating of ferrimagnetic particles and magnetic fluids: physical evaluation of their potential for hyperthermia. Int J Hyperthermia 9:51–68
Minamimura T, Sato H, Kasaoka S, Saito T, Ishizawa S, Takemori S, Tazawa K, Tsukada K (2000) Tumor regression by inductive hyperthermia combined with hepatic embolization using dextran magnetite-incorporated microspheres in rats. Int J Oncol 16:1153–1158
Ito A, Tanaka K, Honda H, Abe S, Yamaguchi H, Kobayashi T (2003) Complete regression of mouse mammary carcinoma with a size greater than 15 mm by frequent repeated hyperthermia using magnetite nanoparticles. J Biosci Bioeng 96:364–369
Shinkai M, Yanase M, Honda H, Wakabayashi T, Yoshida J, Kobayashi T (1996) Intracellular hyperthermia for cancer using magnetite cationic liposomes: in vitro study. Jpn J Cancer Res 87:1179–1183
Suzuki M, Shinkai M, Honda H, Kobayashi T (2003) Anticancer effect and immune induction by hyperthermia of malignant melanoma using magnetite cationic liposomes. Melanoma Res 13:129–135
Yanase M, Shinkai M, Honda H, Wakabayashi T, Yoshida J, Kobayashi T (1998) Intracellular hyperthermia for cancer using magnetite cationic liposomes: an in vivo study. Jpn J Cancer Res 89:463–469
Hamaguchi S, Tohnai I, Ito A, Mitsudo K, Shigetomi T, Ito M, Honda H, Kobayashi T, Ueda M (2003) Selective hyperthermia using magnetoliposomes to target cervical lymph node metastasis in a rabbit tongue tumor model. Cancer Sci 94:834–839
Goldenberg MM (1999) Trastuzumab, a recombinant DNA-derived humanized monoclonal antibody, a novel agent for the treatment of metastatic breast cancer. Clin Ther 21:309–318
Hortobagyi GN (2001) Overview of treatment results with trastuzumab (Herceptin) in metastatic breast cancer. Semin Oncol 28:43–47
Ito A, Kuga Y, Honda H, Kikkawa H, Horiuchi A, Watanabe Y, Kobayashi T (2004) Magnetite nanoparticle-loaded anti-HER2 immunoliposomes for combination of antibody therapy with hyperthermia. Cancer Lett 212:167–175
Lacroix M, Leclercq G (2004) Relevance of breast cancer cell lines as models for breast tumours: an update. Breast Cancer Res Treat 83:249–289
Shinkai M, Suzuki M, Iijima S, Kobayashi T (1995) Antibody-conjugated magnetoliposomes for targeting cancer cells and their application in hyperthermia. Biotechnol Appl Biochem 21(Pt 2):125–137
Ito A, Honda H, Kobayashi T (2006) Cancer immunotherapy based on intracellular hyperthermia using magnetite nanoparticles: a novel concept of “heat-controlled necrosis” with heat shock protein expression. Cancer Immunol Immunother 55:320–328
Kawai N, Ito A, Nakahara Y, Futakuchi M, Shirai T, Honda H, Kobayashi T, Kohri K (2005) Anticancer effect of hyperthermia on prostate cancer mediated by magnetite cationic liposomes and immune-response induction in transplanted syngeneic rats. Prostate 64:373–381
Kawai N, Ito A, Nakahara Y, Honda H, Kobayashi T, Futakuchi M, Shirai T, Tozawa K, Kohri K (2006) Complete regression of experimental prostate cancer in nude mice by repeated hyperthermia using magnetite cationic liposomes and a newly developed solenoid containing a ferrite core. Prostate 66:718–727
Acknowledgments
We thank Mr. Seisaku Tokunaga, Atsushi Murai, and Yoshinori Morita for their skillful technical assistance. We thank Translational Research Institute (Kobe, Japan) for planning and research support. This work was supported in part by a Gan TR grant, The Charitable Trust Soyu Medical Fundation, and a Grant in Aid from the Ministry of Education, Culture, Sports, Science and Technology, Japan (18591435, 17390348).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kikumori, T., Kobayashi, T., Sawaki, M. et al. Anti-cancer effect of hyperthermia on breast cancer by magnetite nanoparticle-loaded anti-HER2 immunoliposomes. Breast Cancer Res Treat 113, 435–441 (2009). https://doi.org/10.1007/s10549-008-9948-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-008-9948-x