Abstract
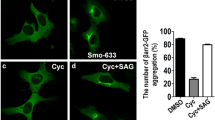
Altered hedgehog signaling is implicated in the development of approximately 20–25% of all cancers, especially those of soft tissues. Genetic evidence in mice as well as immunolocalization studies in human breast cancer specimens suggest that deregulated hedgehog signaling may contribute to breast cancer development. Indeed, two recent studies demonstrated that anchorage-dependent growth of some human breast cancer cell lines is impaired by cyclopamine, a potent hedgehog signaling antagonist targeting the Smoothened (SMO) protein. However, specificity of cyclopamine at the dosage required for growth inhibition (≥10 μM) remained an open question. In this paper we demonstrate that hedgehog signaling antagonists, including cyclopamine, and a second compound, CUR0199691, can inhibit growth of estrogen receptor (ER)-positive and ER-negative tumorigenic breast cancer cells at elevated doses. However, our results indicate that, for most breast cancer cell lines, growth inhibition by these compounds can be independent of detectable Smo gene expression. Rather, our results suggest that cyclopamine and CUR0199691 have unique secondary molecular targets at the dosages required for growth inhibition that are unrelated to hedgehog signaling.











Similar content being viewed by others
References
Lewis MT, Veltmaat JM (2004) Next stop, the twilight zone: hedgehog network regulation of mammary gland development. J Mammary Gland Biol Neoplasia 9:165–181. doi:10.1023/B:JOMG.0000037160.24731.35
Hooper JE, Scott MP (2005) Communicating with hedgehogs. Nat Rev Mol Cell Biol 6:306–317. doi:10.1038/nrm1622
Nusse R (2003) Wnts and hedgehogs: lipid-modified proteins and similarities in signaling mechanisms at the cell surface. Development 130:5297–5305. doi:10.1242/dev.00821
Cohen MM Jr (2003) The hedgehog signaling network. Am J Med Genet 123A:5–28. doi:10.1002/ajmg.a.20495
Liu S, Dontu G, Mantle ID et al (2006) Hedgehog signaling and Bmi-1 regulate self-renewal of normal and malignant human mammary stem cells. Cancer Res 66:6063–6071. doi:10.1158/0008-5472.CAN-06-0054
Briscoe J, Therond P (2005) Hedgehog signaling: from the Drosophila cuticle to anti-cancer drugs. Dev Cell 8:143–151. doi:10.1016/j.devcel.2005.01.008
Chang-Claude J, Dunning A, Schnitzbauer U et al (2003) The patched polymorphism Pro1315Leu (C3944T) may modulate the association between use of oral contraceptives and breast cancer risk. Int J Cancer 103:779–783. doi:10.1002/ijc.10889
Naylor TL, Greshock J, Wang Y et al (2005) High resolution genomic analysis of sporadic breast cancer using array-based comparative genomic hybridization. Breast Cancer Res 7:R1186–R1198. doi:10.1186/bcr1356
Kubo M, Nakamura M, Tasaki A et al (2004) Hedgehog signaling pathway is a new therapeutic target for patients with breast cancer. Cancer Res 64:6071–6074. doi:10.1158/0008-5472.CAN-04-0416
Mukherjee S, Frolova N, Sadlonova A et al (2006) Hedgehog signaling and response to cyclopamine differ in epithelial and stromal cells in benign breast and breast cancer. Cancer Biol Ther 5:674–683
Wolf I, Bose S, Desmond JC et al (2007) Unmasking of epigenetically silenced genes reveals DNA promoter methylation and reduced expression of PTCH in breast cancer. Breast Cancer Res Treat 105:139–155
Moraes RC, Zhang X, Harrington N et al (2007) Constitutive activation of smoothened (Smo) in mammary glands of transgenic mice leads to increased proliferation, altered differentiation and ductal dysplasia. Development 134:1231–1242
Xie J, Johnson RL, Zhang X et al (1997) Mutations of the PATCHED gene in several types of sporadic extracutaneous tumors. Cancer Res 57:2369–2372
Evangelista M, Tian H, de Sauvage FJ (2006) The hedgehog signaling pathway in cancer. Clin Cancer Res 12:5924–5928. doi:10.1158/1078-0432.CCR-06-1736
Riobo NA, Saucy B, Dilizio C, Manning DR (2006) Activation of heterotrimeric G proteins by Smoothened. Proc Natl Acad Sci USA 103:12607–12612. doi:10.1073/pnas.0600880103
Barnes EA, Kong M, Ollendorff V, Donoghue DJ (2001) Patched1 interacts with cyclin B1 to regulate cell cycle progression. EMBO J 20:2214–2223. doi:10.1093/emboj/20.9.2214
Meloni AR, Fralish GB, Kelly P et al (2006) Smoothened signal transduction is promoted by g protein-coupled receptor kinase 2. Mol Cell Biol 26:7550–7560. doi:10.1128/MCB.00546-06
Chen W, Ren XR, Nelson CD et al (2004) Activity-dependent internalization of smoothened mediated by beta-arrestin 2 and GRK2. Science 306:2257–2260. doi:10.1126/science.1104135
Frank-Kamenetsky M, Zhang XM, Bottega S et al (2002) Small-molecule modulators of hedgehog signaling: identification and characterization of Smoothened agonists and antagonists. J Biol 1:10. doi:10.1186/1475-4924-1-10
Williams JA, Guicherit OM, Zaharian BI et al (2003) Identification of a small molecule inhibitor of the hedgehog signaling pathway: effects on basal cell carcinoma-like lesions. Proc Natl Acad Sci USA 100:4616–4621. doi:10.1073/pnas.0732813100
Chen JK, Taipale J, Young KE, Maiti T, Beachy PA (2002) Small molecule modulation of Smoothened activity. Proc Natl Acad Sci USA 99:14071–14076. doi:10.1073/pnas.182542899
Incardona JP, Gaffield W, Kapur RP, Roelink H (1998) The teratogenic Veratrum alkaloid cyclopamine inhibits sonic hedgehog signal transduction. Development 125:3553–3562
Gabay L, Lowell S, Rubin LL, Anderson DJ (2003) Deregulation of dorsoventral patterning by FGF confers trilineage differentiation capacity on CNS stem cells in vitro. Neuron 40:485–499. doi:10.1016/S0896-6273(03)00637-8
Levitt RJ, Zhao Y, Blouin MJ, Pollak M (2007) The hedgehog pathway inhibitor cyclopamine increases levels of p27, and decreases both expression of IGF-II and activation of Akt in PC-3 prostate cancer cells. Cancer Lett 255:300–306
Masdeu C, Faure H, Coulombe J et al (2006) Identification and characterization of hedgehog modulator properties after functional coupling of Smoothened to G15. Biochem Biophys Res Commun 349:471–479. doi:10.1016/j.bbrc.2006.07.216
Mimeault M, Moore E, Moniaux N et al (2005) Cytotoxic effects induced by a combination of cyclopamine and gefitinib, the selective hedgehog and epidermal growth factor receptor signaling inhibitors, in prostate cancer cells. Int J Cancer 118:1022–1031
Chen JK, Taipale J, Cooper MK, Beachy PA (2002) Inhibition of hedgehog signaling by direct binding of cyclopamine to Smoothened. Genes Dev 16:2743–2748. doi:10.1101/gad.1025302
Berman DM, Karhadkar SS, Hallahan AR et al (2002) Medulloblastoma growth inhibition by hedgehog pathway blockade. Science 297:1559–1561. doi:10.1126/science.1073733
Tabs S, Avci O (2004) Induction of the differentiation and apoptosis of tumor cells in vivo with efficiency and selectivity. Eur J Dermatol 14:96–102
Sanchez P, Ruiz i Altaba A (2005) In vivo inhibition of endogenous brain tumors through systemic interference of Hedgehog signaling in mice. Mech Dev 122:223–230. doi:10.1016/j.mod.2004.10.002
Athar M, Li C, Tang X et al (2004) Inhibition of smoothened signaling prevents ultraviolet B-induced basal cell carcinomas through regulation of Fas expression and apoptosis. Cancer Res 64:7545–7552. doi:10.1158/0008-5472.CAN-04-1393
Romer JT, Kimura H, Magdaleno S et al (2004) Suppression of the Shh pathway using a small molecule inhibitor eliminates medulloblastoma in Ptc1(+/−)p53(−/−) mice. Cancer Cell 6:229–240. doi:10.1016/j.ccr.2004.08.019
Sasai K, Romer JT, Lee Y et al (2006) Shh pathway activity is down-regulated in cultured medulloblastoma cells: implications for preclinical studies. Cancer Res 66:4215–4222. doi:10.1158/0008-5472.CAN-05-4505
Lewis DE, Ng Tang DS, Wang X, Kozinetz C (1999) Costimulatory pathways mediate monocyte-dependent lymphocyte apoptosis in HIV. Clin Immunol 90:302–312. doi:10.1006/clim.1998.4663
Incardona JP, Roelink H (2000) The role of cholesterol in Shh signaling and teratogen-induced holoprosencephaly. Cell Mol Life Sci 57:1709–1719. doi:10.1007/PL00000653
Incardona JP, Gaffield W, Lange Y et al (2000) Cyclopamine inhibition of Sonic hedgehog signal transduction is not mediated through effects on cholesterol transport. Dev Biol 224:440–452. doi:10.1006/dbio.2000.9775
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)). Methods 25:402–408
Chiang C, Swan RZ, Grachtchouk M et al (1999) Essential role for Sonic hedgehog during hair follicle morphogenesis. Dev Biol 205:1–9. doi:10.1006/dbio.1998.9103
Detmer K, Walker AN, Jenkins TM, Steele TA, Dannawi H (2000) Erythroid differentiation in vitro is blocked by cyclopamine, an inhibitor of hedgehog signaling. Blood Cells Mol Dis 26:360–372. doi:10.1006/bcmd.2000.0318
Taipale J, Chen JK, Cooper MK et al (2000) Effects of oncogenic mutations in Smoothened and Patched can be reversed by cyclopamine. Nature 406:1005–1009. doi:10.1038/35023008
Qualtrough D, Buda A, Gaffield W, Williams AC, Paraskeva C (2004) Hedgehog signalling in colorectal tumour cells: induction of apoptosis with cyclopamine treatment. Int J Cancer 110:831–837. doi:10.1002/ijc.20227
Zhang J, Lipinski R, Shaw A, Gipp J, Bushman W (2007) Lack of demonstrable autocrine hedgehog signaling in human prostate cancer cell lines. J Urol 177:1179–1185. doi:10.1016/j.juro.2006.10.032
Mimeault M, Moore E, Moniaux N et al (2006) Cytotoxic effects induced by a combination of cyclopamine and gefitinib, the selective hedgehog and epidermal growth factor receptor signaling inhibitors, in prostate cancer cells. Int J Cancer 118:1022–1031. doi:10.1002/ijc.21440
Lewis MT, Ross S, Strickland PA et al (1999) Defects in mouse mammary gland development caused by conditional haploinsufficiency of Patched-1. Development 126:5181–5193
Lewis MT, Ross S, Strickland PA et al (2001) The Gli2 transcription factor is required for normal mouse mammary gland development. Dev Biol 238:133–144. doi:10.1006/dbio.2001.0410
Acknowledgements
This work was supported, in part, by a Department of Defense IDEA Award (DAMD17-00-1-0477), a SPORE Developmental Project Grant (P50 CA50183), and a National Institutes of Health Grant (P01- CA30195). The authors thank Drs. Frederick de Sauvage (Genentech Inc. South San Francisco, CA) and Stephen Gould (Curis Inc. Cambridge MA) for providing CUR0199691 and recombinant SHH-N ligand for this study, as well as for critical evaluation of the data. We thank Dr. Matthew Herynk for advice regarding cell culture, and Dr. Andra Frost for helpful discussions, provision of a Smo primer/probe set used previously in her laboratory, and for critical evaluation of the data.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Zhang, X., Harrington, N., Moraes, R.C. et al. Cyclopamine inhibition of human breast cancer cell growth independent of Smoothened (Smo) . Breast Cancer Res Treat 115, 505–521 (2009). https://doi.org/10.1007/s10549-008-0093-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-008-0093-3