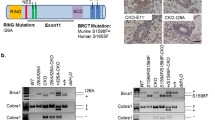
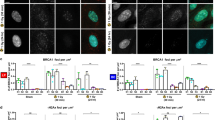
Abstract
Tumours arising in BRCA1 mutation carriers have a characteristic phenotype, the molecular and cellular basis of which is unknown. To address the hypothesis that this phenotype reflects a role for BRCA1 in either in the basal or the stem cell compartments of the mammary epithelia, we have targeted its disruption to K14 and K6a expressing cells of the mouse. Unlike MMTV and WAP driven conditional knockout models of Brca1, these two models did not result in any observable changes in the mammary gland. Our results suggest that BRCA1-associated tumours arise either in K14 and K6a negative basal cells of the mammary gland, or possibly from transdifferentiation of luminal epithelia.


Similar content being viewed by others
References
Marquis ST, Rajan JV, Wynshaw-Boris A, Xu J, Yin GY, Abel KJ, Weber BL, Chodosh LA (1995) The developmental pattern of Brca1 expression implies a role in differentiation of the breast and other tissues. Nat Genet 11:17–26
Brown MA, Nicolai H, Howe K, Katagiri T, Lalani E-N, Simpson KJ, Manning NW, Deans A, Chen P, Khanna KK, Wati MR, Griffiths BL, Xu CF, Stamp GW, Solomon E (2002) Expression of a truncated Brca1 protein delays lactational mammary development in transgenic mice. Transgenic Res 11:467–478
Xu X, Wagner KU, Larson D, Weaver Z, Li C, Ried T, Hennighausen L, Wynshaw-Boris A, Deng CX (1999) Conditional mutation of Brca1 in mammary epithelial cells results in blunted ductal morphogenesis and tumour formation. Nat Genet 22:37–43
Russo J, Lynch H, Russo IH (2001) Mammary gland architecture as a determining factor in the susceptibility of the human breast to cancer. Breast J 7:278–291
Lakhani S, Easton D, Stratton M (1997) Pathology of familial breast cancer: differences between breast cancers in carriers of BRCA1 or BRCA2 mutations and sporadic cases. Breast cancer linkage consortium. Lancet 349:1505–1510
Sorlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H, Hastie T, Eisen MB, van de Rijn M, Jeffrey SS, Thorsen T, Quist H, Matese JC, Brown PO, Botstein D, Eystein Lonning P, Borresen Dale AL (2001) Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 98:10869–10874
Foulkes WD (2004) BRCA1 functions as a breast stem cell regulator. J Med Genet 41:1–5
Silva LD, Clarke C, Lakhani SR (2007) Basal-like breast cancer. J Clin Pathol 60(12):1328–1332
Mikaelian I, Hovick M, Silva KA, Burzenski LM, Shultz LD, Ackert-Bicknell CL, Cox GA, Sundberg JP (2006) Expression of terminal differentiation proteins defines stages of mouse mammary gland development. Vet Pathol 43:36–49
Smith GH, Mehrel T, Roop DR (1990) Differential keratin gene expression in developing, differentiating, preneoplastic, and neoplastic mouse mammary epithelium. Cell Growth Differ 1:161–170
Jonkers J, Meuwissen R, van der Gulden H, Peterse H, van der Valk M, Berns A (2001) Synergistic tumor suppressor activity of BRCA2 and p53 in a conditional mouse model for breast cancer. Nat Genet 29:418–425
Gritli-Linde A, Hallberg K, Harfe BD, Reyahi A, Kannius-Janson M, Nilsson J, Cobourne MT, Sharpe PT, McMahon AP, Linde A (2007) Abnormal hair development and apparent follicular transformation to mammary gland in the absence of hedgehog signaling. Dev Cell 12:99–112
Grimm SL, Bu W, Longley MA, Roop DR, Li Y, Rosen JM (2006) Keratin 6 is not essential for mammary gland development. Breast Cancer Res 8:R29
Stingl J, Eirew P, Ricketson I, Shackleton M, Vaillant F, Choi D, Li HI, Eaves CJ (2006) Purification and unique properties of mammary epithelial stem cells. Nature 439:993–997
Villadsen R, Fridriksdottir AJ, Ronnov-Jessen L, Gudjonsson T, Rank F, LaBarge MA, Bissell MJ, Petersen OW (2007) Evidence for a stem cell hierarchy in the adult human breast. J Cell Biol 177:87–101
Ball SM (1998) The development of the terminal end bud in the prepubertal–pubertal mouse mammary gland. Anat Rec 250:459–464
Wagner KU, McAllister K, Ward T, Davis B, Wiseman R, Hennighausen L (2001) Spatial and temporal expression of the Cre gene under the control of the MMTV-LTR in different lines of transgenic mice. Transgenic Res 10:545–553
Brodie SG, Xu X, Qiao W, Li WM, Cao L, Deng CX (2001) Multiple genetic changes are associated with mammary tumorigenesis in Brca1 conditional knockout mice. Oncogene 20:7514–7523
Berton TR, Matsumoto T, Page A, Conti CJ, Deng CX, Jorcano JL, Johnson DG (2003) Tumor formation in mice with conditional inactivation of Brca1 in epithelial tissues. Oncogene 22:5415–5426
Liu X, Holstege H, van der Gulden H, Treur-Mulder M, Zevenhoven J, Velds A, Kerkhoven RM, van Vliet MH, Wessels LF, Peterse JL, Berns A, Jonkers J (2007) Somatic loss of BRCA1 and p53 in mice induces mammary tumors with features of human BRCA1-mutated basal-like breast cancer. Proc Natl Acad Sci USA 104:12111–12116
Shackleton M, Vaillant F, Simpson KJ, Stingl J, Smyth GK, Asselin-Labat ML, Wu L, Lindeman GJ, Visvader JE (2006) Generation of a functional mammary gland from a single stem cell. Nature 439:84–88
Crook T, Crossland S, Crompton MR, Osin P, Gusterson BA (1997) p53 mutations in BRCA1-associated familial breast cancer. Lancet 350:638–639
Smyth I, Ellis T, Hetherington R, Riley E, Narang M, Mahony D, Wicking C, Rothnagel JA, Wainwright BJ (2004) Krt6a-Cre transgenic mice direct LoxP-mediated recombination to the companion cell layer of the hair follicle and following induction by retinoic acid to the interfollicular epidermis. J Invest Dermatol 122:232–234
Jackson D, Bresnick J, Rosewell I, Crafton T, Poulsom R, Stamp G, Dickson C (1997) Fibroblast growth factor receptor signalling has a role in lobuloalveolar development of the mammary gland. J Cell Sci 110(Pt 11):1261–1268
Durban EM, Barreto PD, Hilgers J, Sonnenberg A (1994) Cell phenotypes and differentiative transitions in mouse submandibular salivary gland defined with monoclonal antibodies to mammary epithelial cells. J Histochem Cytochem 42:185–196
Acknowledgements
The authors are grateful to Will Foulkes for helpful discussion, Paul Addison and Tina Chua for assistance with histology, Craig Dickfos and Kym-Kelly Taylor for animal husbandry, and the National Breast Cancer Foundation of Australia and The University of Queensland for funding.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Smart, C.E., Clarke, C., Brooks, K.M. et al. Targeted disruption of Brca1 in restricted compartments of the mouse mammary epithelia. Breast Cancer Res Treat 112, 237–241 (2008). https://doi.org/10.1007/s10549-007-9859-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-007-9859-2