Abstract
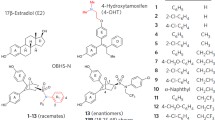
Antiestrogens used for breast cancer therapy can be categorized into two classes that differ in their effect on estrogen receptor (ER) alpha stability. The selective estrogen receptor modulators (SERMs) stabilize ER alpha and the selective estrogen receptor downregulators (SERDs) cause a decrease in cellular ER alpha levels. A clinically relevant antiestrogen, GW7604, appears to work through a SERD-like mechanism, despite sharing the same molecular scaffold as 4-hydroxytamoxifen, a SERM. In order to investigate potential structural features of GW7604 responsible for SERD activity, GW7604 and two analogs were synthesized using a new, improved synthetic route and tested for their effects on ER alpha function and cell proliferation. The two analogs, which have an acrylamide or a methyl vinyl ketone replacing the acrylic acid group of GW7604, display lower binding affinity for ER alpha than GW7604, but show similar antagonism of estradiol-induced activation of ER alpha-mediated transcription as GW7604 and inhibit estradiol-induced proliferation of the MCF-7 cell line with a similar potency as GW7604. Unlike GW7604, neither analog has a significant effect on cellular ER alpha levels, suggesting that the carboxylate is a key determinant in GW7604 action and, for the first time, showing that this group is responsible for inducing ER alpha degradation in breast cancer cells.







Similar content being viewed by others
References
Robertson JF (2004) Selective oestrogen receptor modulators/new antioestrogens: a clinical perspective. Cancer Treat Rev 30:695-706
Osborne CK, Wakeling A, Nicholson RI (2004) Fulvestrant: an oestrogen receptor antagonist with a novel mechanism of action. Br J Cancer 90(Suppl 1):S2–6
Normanno N, Di Maio M, De Maio E et al (2005) Mechanisms of endocrine resistance and novel therapeutic strategies in breast cancer. Endocr Relat Cancer 12:721–747
Willson TM, Norris JD, Wagner BL et al (1997) Dissection of the molecular mechanism of action of GW5638, a novel estrogen receptor ligand, provides insights into the role of estrogen receptor in bone. Endocrinology 138:3901–3911
Wijayaratne AL, Nagel SC, Paige LA et al (1999) Comparative analyses of mechanistic differences among antiestrogens. Endocrinology 140:5828–5840
Fan M, Long X, Bailey JA et al (2002) The activating enzyme of NEDD8 inhibits steroid receptor function. Mol Endocrinol 16:315–330
Wu YL, Yang X, Ren Z et al (2005) Structural basis for an unexpected mode of SERM-mediated ER antagonism. Mol Cell 18:413–424
Willson TM, Henke BR, Momtahen TM et al (1994) 3-[4-(1,2-Diphenylbut-1-enyl)phenyl]acrylic acid: a non-steroidal estrogen with functional selectivity for bone over uterus in rats. J Med Chem 37:1550–1552
Weatherman RV, Clegg NJ, Scanlan TS (2001) Differential SERM activation of the estrogen receptors (ERalpha and ERbeta) at AP-1 sites. Chem Biol 8:427–436
Eaddy III, JF, Heyer D, Katamreddy SR et al (2005) Preparation of acyloxydiphenylbutenylcinnamates as estrogen receptor modulator prodrugs PCT application WO2005033056
Smyth TP, Corby BW (1998) Toward a clean alternative to Friedel-Crafts acylation: In situ formation, observation, and reaction of an acyl bis(trifluoroacetyl)phosphate and related structures. J Org Chem 63:8946–8951
Wijayaratne AL, McDonnell DP (2001) The human estrogen receptor-alpha is a ubiquitinated protein whose stability is affected differentially by agonists, antagonists, and selective estrogen receptor modulators. J Biol Chem 276:35684–35692
Robertson JF, Come SE, Jones SE et al (2005) Endocrine treatment options for advanced breast cancer–the role of fulvestrant. Eur J Cancer 41:346–356
Shao W, Brown M (2004) Advances in estrogen receptor biology: prospects for improvements in targeted breast cancer therapy. Breast Cancer Res 6:39–52
Pike AC, Brzozowski AM, Walton J et al (2001) Structural insights into the mode of action of a pure antiestrogen. Structure (Camb) 9:145–153
Bohley P (1996) Surface hydrophobicity and intracellular degradation of proteins. Biol Chem 377:425–435
Acknowledgements
RVW acknowledges the Purdue Cancer Center, the Indiana Elks Charities and the Army Breast Cancer Research Program (BC030507) for supporting this work. KPN gratefully acknowledges the following agencies for supporting this work: the Walther Cancer Institute, U.S. Army Medical Research Acquisition Activity, Award Numbers DAMD 17-02-1-0418 and DAMD17-02-1-0419; American Cancer Society Research and Alaska Run for Woman Grant TBE-104125
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Fan, M., Rickert, E.L., Chen, L. et al. Characterization of molecular and structural determinants of selective estrogen receptor downregulators. Breast Cancer Res Treat 103, 37–44 (2007). https://doi.org/10.1007/s10549-006-9353-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-006-9353-2