Abstract
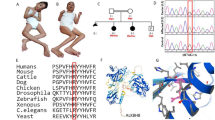
Intellectual disability is genetically heterogeneous, and it is likely that many of the responsible genes have not yet been identified. We describe three siblings with isolated, severe developmental encephalopathy. After extensive uninformative genetic and metabolic testing, whole exome sequencing identified a homozygous novel variant in glutamic pyruvate transaminase 2 (GPT2) or alanine transaminase 2 (ALT2), c.459 C > G p.Ser153Arg that segregated with developmental encephalopathy in the family. This variant was predicted to be damaging by all in silico prediction algorithms. GPT2 is the gene encoding ALT2 which is responsible for the reversible transamination of alanine and 2-oxoglutarate to form pyruvate and glutamate. GPT2 is expressed in brain and is in the pathway to generate glutamate, an excitatory neurotransmitter. Functional assays of recombinant wild-type and mutant ALT2 proteins demonstrated the p.Ser153Arg mutation resulted in a severe loss of enzymatic function. We suggest that recessively inherited loss of function GPT2 mutations are a novel cause of intellectual disability.




Similar content being viewed by others
References
Daily DK et al (2000) Identification and evaluation of mental retardation. Am Fam Physician 61(4):1059–1067, 1070
DePristo M, Banks E, Poplin R et al (2011) A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat Genet 43:491–498
Ellison JW et al (2013) Genetic Basis of Intellectual Disability. Annu Rev Med 64:441–450
Kaufman L et al (2010) The genetic basis of non-syndromic intellectual disability: a review. J Neuro Dev Disord 2:182–209
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25(14):1754–1760
Li H, Handsaker B, Wysoker A et al (2009) The Sequence alignment/map (SAM) format and SAMtools. Bioinformatics 25:2078–2079
Lindblom P, Rafter I, Glinghammar B (2007) Isoforms of alanine aminotransferases in human tissues and serum—differential tissue expression using novel antibodies. Arch Biochem Biophys 466:66–77
Liu L, Zhong S, Yang R et al (2008) Expression, purification, and initial characterization of human alanine aminotransferase (GPT2) isoenzyme 1 and 2 in high-five insect cells. Protein Expr Purif 60:225–231
Masson J, Darmon M, Hen R, Rayport S (2006) Mice lacking brain/kidney phosphate-activated glutaminase have impaired glutamatergic synaptic transmission, altered breathing, disorganized goal-directed behavior and die shortly after birth. J Neurosci 26(17):4660–4671, 4241-05
Najmabadi H, Hu H, Garshasbi M et al (2011) Deep sequencing reveals 50 novel genes for recessive cognitive disorders. Nature 478:57–63
Snell K, Duff DA (1985) Branched-chain amino acid metabolism and alanine formation in rat muscles in vitro. Mitochondrial-cytosolic interrelationships. Biochem J 225(3):737–743
Topper S et al (2011) Exome sequencing and the genetics of intellectual disability. Clin Genet 2011(80):117–126
Utine GE, Haliloğlu G, Volkan-Salancı B et al (2014) Etiological yield of SNP microarrays in idiopathic intellectual disability. Eur J Pediatr Neurol 18(3):327–337
Yang RZ et al (2002) cDNA cloning, genomic structure, chromosomal mapping, and functional expression of a novel human alanine aminotransferase. Genomics 79:445–450
Yang RZ, Park S, Reagan WJ et al (2009) Alanine aminotransferase isoenzymes: molecular cloning and quantitative analysis of tissue expression in rats and serum elevation in liver toxicity. Hepatology 49:598–607
Acknowledgments
We thank the patients and their family for their contribution. Research reported in this publication was supported by the National Institutes of Health under award number: T32GM082771, Maryland Clinical Nutrition Research Unit (DK072488), and The Baltimore Diabetes Research and Training Center (P60-DK-079637). Authors acknowledge Alan Shuldiner for helpful discussions and critical review of the manuscript.
Conflicts of Interest
Eden Haverfield is an employee of GeneDx, Inc. Wendy K. Chung is a consultant to BioReference Laboratories.
Human and animal rights and informed consent
This article does not contain any studies with human or animal subjects performed by the any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Gajja Salomons
Katrina Celis, Scott Shuldiner, Da-Wei Gong and Wendy K. Chung contributed equally to this work.
Rights and permissions
About this article
Cite this article
Celis, K., Shuldiner, S., Haverfield, E.V. et al. Loss of function mutation in glutamic pyruvate transaminase 2 (GPT2) causes developmental encephalopathy. J Inherit Metab Dis 38, 941–948 (2015). https://doi.org/10.1007/s10545-015-9824-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-015-9824-x