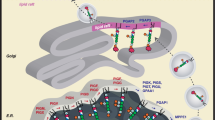
Abstract
Glycosylation - enabling genes are thought to comprise approximately 1–2 % of the human genome, thus, it is not surprising that more than 100 genetic disorders have been identified in this complex multi-pathway cellular process. Recent advances in next generation sequencing technology (NGS) have led to the discovery of genetic causes of many new disorders and importantly highlighted the broad phenotypes that occur. Here we will focus on two glycosylation pathways that involve lipids; glycosylphosphatidylinositol (GPI) anchors and glycosphingolipids (GSL) with emphasis on the specific gene defects, their biochemical properties, and their expanding clinical spectra. These disorders involve the intersection of two pathways: lipids and carbohydrates. Studies of both pathways were founded on structural biochemistry. Those methods and their more refined and sensitive descendants can both identify the specific genes that cause the disorders and validate the importance of the specific mutations.


Similar content being viewed by others
References
Almeida AM, Murakami Y, Layton DM et al (2006) Hypomorphic promoter mutation in PIGM causes inherited glycosylphosphatidylinositol deficiency. Nat Med 12(7):846–851
Almeida AM, Murakami Y, Baker A et al (2007) Targeted therapy for inherited GPI deficiency. N Engl J Med 356(16):1641–1647
Ashida H, Hong Y, Murakami Y et al (2005) Mammalian PIG-X and yeast Pbn1p are the essential components of glycosylphosphatidylinositol-mannosyltransferase I. Mol Biol Cell 16(3):1439–1448
Belet S, Fieremans N, Yuan X et al (2014) Early frameshift mutation in PIGA identified in a large XLID family without neonatal lethality. Hum Mutat 35(3):350–355
Boccuto L, Aoki K, Flanagan-Steet H et al (2014) A mutation in a ganglioside biosynthetic enzyme, ST3GAL5, results in salt & pepper syndrome, a neurocutaneous disorder with altered glycolipid and glycoprotein glycosylation. Hum Mol Genet 23(2):418–433
Boukhris A, Schule R, Loureiro JL et al (2013) Alteration of ganglioside biosynthesis responsible for complex hereditary spastic paraplegia. Am J Hum Genet 93(1):118–123
Brodsky RA, Mukhina GL, Li S et al (2000) Improved detection and characterization of paroxysmal nocturnal hemoglobinuria using fluorescent aerolysin. Am J Clin Pathol 114(3):459–466
Chiyonobu T, Inoue N, Morimoto M, Kinoshita T, Murakami Y (2014) Glycosylphosphatidylinositol (GPI) anchor deficiency caused by mutations in PIGW is associated with West syndrome and hyperphosphatasia with mental retardation syndrome. J Med Genet 51(3):203–207
Edvardson S, Baumann AM, Muhlenhoff M et al (2013) West syndrome caused by ST3Gal-III deficiency. Epilepsia 54(2):e24–e27
Ferguson MAJ, Kinoshita T, Hart GW (2009) Glycosylphosphatidylinositol Anchors. In: A Varki, RD Cummings, JD Esko, HH Freeze, P Stanley, CR Bertozzi, GW Hart, ME Etzler (eds) Essentials of glycobiology. Cold Spring Harbor, New York
Freeze HH, Chong JX, Bamshad MJ, Ng BG (2014) Solving glycosylation disorders: fundamental approaches reveal complicated pathways. Am J Hum Genet 94(2):161–175
Fujita M, Kinoshita T (2012) GPI-anchor remodeling: potential functions of GPI-anchors in intracellular trafficking and membrane dynamics. Biochim Biophys Acta 1821(8):1050–1058
Fujita M, Maeda Y, Ra M, Yamaguchi Y, Taguchi R, Kinoshita T (2009) GPI glycan remodeling by PGAP5 regulates transport of GPI-anchored proteins from the ER to the Golgi. Cell 139(2):352–365
Hansen L, Tawamie H, Murakami Y et al (2013) Hypomorphic mutations in PGAP2, encoding a GPI-anchor-remodeling protein, cause autosomal-recessive intellectual disability. Am J Hum Genet 92(4):575–583
Hong Y, Maeda Y, Watanabe R et al (1999) Pig-n, a mammalian homologue of yeast Mcd4p, is involved in transferring phosphoethanolamine to the first mannose of the glycosylphosphatidylinositol. J Biol Chem 274(49):35099–35106
Hong Y, Maeda Y, Watanabe R, Inoue N, Ohishi K, Kinoshita T (2000) Requirement of PIG-F and PIG-O for transferring phosphoethanolamine to the third mannose in glycosylphosphatidylinositol. J Biol Chem 275(27):20911–20919
Howard MF, Murakami Y, Pagnamenta AT et al (2014) Mutations in PGAP3 impair GPI-anchor maturation, causing a subtype of hyperphosphatasia with mental retardation. Am J Hum Genet 94(2):278–287
Hu H, Eggers K, Chen W et al (2011) ST3GAL3 mutations impair the development of higher cognitive functions. Am J Hum Genet 89(3):407–414
Johnston JJ, Gropman AL, Sapp JC et al (2012) The phenotype of a germline mutation in PIGA: the gene somatically mutated in paroxysmal nocturnal hemoglobinuria. Am J Hum Genet 90(2):295–300
Kang JY, Hong Y, Ashida H et al (2005) PIG-V involved in transferring the second mannose in glycosylphosphatidylinositol. J Biol Chem 280(10):9489–9497
Kato M, Saitsu H, Murakami Y et al (2014) PIGA mutations cause early-onset epileptic encephalopathies and distinctive features. Neurology 82(18):1587–1596
Krawitz PM, Schweiger MR, Rodelsperger C et al (2010) Identity-by-descent filtering of exome sequence data identifies PIGV mutations in hyperphosphatasia mental retardation syndrome. Nat Genet 42(10):827–829
Krawitz PM, Murakami Y, Hecht J et al (2012) Mutations in PIGO, a member of the GPI-anchor-synthesis pathway, cause hyperphosphatasia with mental retardation. Am J Hum Genet 91(1):146–151
Krawitz PM, Hochsmann B, Murakami Y et al (2013a) A case of paroxysmal nocturnal hemoglobinuria caused by a germline mutation and a somatic mutation in PIGT. Blood 122(7):1312–1315
Krawitz PM, Murakami Y, Riess A et al (2013b) PGAP2 mutations, affecting the GPI-anchor-synthesis pathway, cause hyperphosphatasia with mental retardation syndrome. Am J Hum Genet 92(4):584–589
Kuki I, Takahashi Y, Okazaki S et al (2013) Vitamin B6-responsive epilepsy due to inherited GPI deficiency. Neurology 81(16):1467–1469
Kvarnung M, Nilsson D, Lindstrand A et al (2013) A novel intellectual disability syndrome caused by GPI anchor deficiency due to homozygous mutations in PIGT. J Med Genet 50(8):521–528
Maeda Y, Watanabe R, Harris CL et al (2001) PIG-M transfers the first mannose to glycosylphosphatidylinositol on the lumenal side of the ER. EMBO J 20(1–2):250–261
Maeda Y, Ashida H, Kinoshita T (2006) CHO glycosylation mutants: GPI anchor. Methods Enzymol 416:182–205
Maeda Y, Tashima Y, Houjou T et al (2007) Fatty acid remodeling of GPI-anchored proteins is required for their raft association. Mol Biol Cell 18(4):1497–1506
Martin HC, Kim GE, Pagnamenta AT et al (2014) Clinical whole-genome sequencing in severe early-onset epilepsy reveals new genes and improves molecular diagnosis. Hum Mol Genet. doi:10.1093/hmg/ddu199
Maydan G, Noyman I, Har-Zahav A et al (2011) Multiple congenital anomalies-hypotonia-seizures syndrome is caused by a mutation in PIGN. J Med Genet 48(6):383–389
Murakami Y, Siripanyapinyo U, Hong Y et al (2003) PIG-W is critical for inositol acylation but not for flipping of glycosylphosphatidylinositol-anchor. Mol Biol Cell 14(10):4285–4295
Murakami Y, Kanzawa N, Saito K et al (2012) Mechanism for release of alkaline phosphatase caused by glycosylphosphatidylinositol deficiency in patients with hyperphosphatasia mental retardation syndrome. J Biol Chem 287(9):6318–6825
Murakami Y, Tawamie H, Maeda Y et al (2014) Null mutation in PGAP1 impairing Gpi-anchor maturation in patients with intellectual disability and encephalopathy. PLoS Genet 10(5):e1004320. doi:10.1371/journal.pgen.1004320
Ng BG, Hackmann K, Jones MA et al (2012) Mutations in the glycosylphosphatidylinositol gene PIGL cause CHIME syndrome. Am J Hum Genet 90(4):685–688
Ohba C, Okamoto N, Murakami Y et al (2014) PIGN mutations cause congenital anomalies, developmental delay, hypotonia, epilepsy, and progressive cerebellar atrophy. Neurogenetics 15(2):85–92
Ohishi K, Inoue N, Kinoshita T (2001) PIG-S and PIG-T, essential for GPI anchor attachment to proteins, form a complex with GAA1 and GPI8. EMBO J 20(15):4088–4098
Schnaar RL, Suzuki A, Stanley P (2009) Glycosphingolipids. In: A Varki, RD Cummings, JD Esko, HH Freeze, P Stanley, CR Bertozzi, GW Hart, ME Etzler (eds) Essentials of glycobiology. Cold Spring Harbor, New York
Schnaar RL, Gerardy-Schahn R, Hildebrandt H (2014) Sialic acids in the brain: gangliosides and polysialic acid in nervous system development, stability, disease, and regeneration. Physiol Rev 94(2):461–518
Simpson MA, Cross H, Proukakis C et al (2004) Infantile-onset symptomatic epilepsy syndrome caused by a homozygous loss-of-function mutation of GM3 synthase. Nat Genet 36(11):1225–1229
Swoboda KJ, Margraf RL, Carey JC et al (2014) A novel germline PIGA mutation in Ferro-Cerebro-Cutaneous syndrome: a neurodegenerative X-linked epileptic encephalopathy with systemic iron-overload. Am J Med Genet A 164A(1):17–28
Takahashi M, Inoue N, Ohishi K et al (1996) PIG-B, a membrane protein of the endoplasmic reticulum with a large lumenal domain, is involved in transferring the third mannose of the GPI anchor. EMBO J 15(16):4254–4261
Takeda J, Miyata T, Kawagoe K et al (1993) Deficiency of the GPI anchor caused by a somatic mutation of the PIG-A gene in paroxysmal nocturnal hemoglobinuria. Cell 73(4):703–711
Tanaka S, Maeda Y, Tashima Y, Kinoshita T (2004) Inositol deacylation of glycosylphosphatidylinositol-anchored proteins is mediated by mammalian PGAP1 and yeast Bst1p. J Biol Chem 279(14):14256–14263
Tettamanti G, Bonali F, Marchesini S, Zambotti V (1973) A new procedure for the extraction, purification and fractionation of brain gangliosides. Biochim Biophys Acta 296(1):160–170
Thompson MD, Killoran A, Percy ME, Nezarati M, Cole DE, Hwang PA (2006) Hyperphosphatasia with neurologic deficit: a pyridoxine-responsive seizure disorder? Pediatr Neurol 34(4):303–307
Wakil SM, Monies DM, Ramzan K et al (2013) Novel B4GALNT1 mutations in a complicated form of hereditary spastic paraplegia. Clin Genet, doi: 10.1111/cge.12312
Watanabe R, Ohishi K, Maeda Y, Nakamura N, Kinoshita T (1999) Mammalian PIG-L and its yeast homologue Gpi12p are N-acetylglucosaminylphosphatidylinositol de-N-acetylases essential in glycosylphosphatidylinositol biosynthesis. Biochem J 339(Pt 1):185–192
Yang HS, Yang M, Li X, Tugulea S, Dong H (2013) Diagnosis of paroxysmal nocturnal hemoglobinuria in peripheral blood and bone marrow with six-color flow cytometry. Biomark Med 7(1):99–111
Compliance with ethics guidelines
ᅟ
Informed consent
This article does not contain any studies with human or animal subjects performed by the any of the authors.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Ron A. Wevers
Rights and permissions
About this article
Cite this article
Ng, B.G., Freeze, H.H. Human genetic disorders involving glycosylphosphatidylinositol (GPI) anchors and glycosphingolipids (GSL). J Inherit Metab Dis 38, 171–178 (2015). https://doi.org/10.1007/s10545-014-9752-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-014-9752-1