Abstract
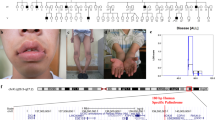
Alu-mediated tandem duplication of exons 4 and 5 (g.15815_22218dup6404) is a novel mutation that has been detected in the LAMP2 gene (Xq24). This exon copy number variation was found in two brothers with the typical phenotype of Danon disease, including characteristic myocardial changes on magnetic resonance imaging. The 6.4 kb duplication was identified in both boys by a combination of exon dosage qPCR analyses and duplication breakpoint/junction mapping. The rearrangement results in a plethora of abnormal LAMP2 splicing variants and also in use of likely cryptic splice sites in the 3′ terminus of LAMP2 gene. Although we found minute amounts of normal LAMP2B and LAMP2A mRNAs, no protein was detectable in peripheral blood leukocytes by flow cytometry in both brothers. Uniquely, the fraction of LAMP2-deficient granulocytes (0.06 %) assessed by flow cytometry in the patients’ asymptomatic mother substantially differed from the random distribution of X-chromosome inactivation in her leukocytes. This discrepancy was later explained by molecular genetic methods as a consequence of mosaic distribution of the mutation in her somatic tissues. Altogether, we report a novel and mosaically distributed exon copy number rearrangement in the LAMP2 gene and comment on obstacles this genetic setup presents to the overall cellular and molecular diagnostic algorithm of Danon disease. Our observations of the mosaicism in the asymptomatic mother suggest that similarly affected females could be a potentially under-diagnosed Danon disease carrier group and that LAMP2 flow cytometry, because of its supreme sensitivity, can be an efficient method for pedigree screening.


Similar content being viewed by others
Abbreviations
- bp:
-
base pair
- CDS:
-
coding sequence
- CMR:
-
cardiac magnetic resonance
- CNV:
-
copy number variation
- CP:
-
crossing point value
- DD:
-
Danon disease
- ECG:
-
electrocardiography
- LGE:
-
late gadolinium enhancement
- LAMP:
-
lysosomal-associated membrane protein
- LV:
-
left ventricle
- WBC:
-
white blood cells
- qPCR:
-
quantitative polymerase chain reaction
- wt:
-
wild-type
- XCI:
-
X-chromosome inactivation
References
Boucek D, Jirikowic J, Taylor M (2011) Natural history of Danon disease. Genet Med 13:563–568
Chen JW, Murphy TL, Willingham MC, Pastan I, August JT (1985) Identification of two lysosomal membrane glycoproteins. J Cell Biol 101:85–95
Chen XL, Zhao Y, Ke HP, Liu WT, Du ZF, Zhang XN (2012) Detection of somatic and germline mosaicism for the LAMP2 gene mutation c.808dupG in a Chinese family with Danon disease. Gene 507:174–176
Cuervo AM, Dice JF (2000) Unique properties of lamp2a compared to other lamp2 isoforms. J Cell Sci 113(Pt 24):4441–4450
Danon MJ, Oh SJ, DiMauro S et al (1981) Lysosomal glycogen storage disease with normal acid maltase. Neurology 31:51–57
den Dunnen JT, Antonarakis SE (2001) Nomenclature for the description of human sequence variations. Hum Genet 109:121–124
Eskelinen EL (2006) Roles of LAMP-1 and LAMP-2 in lysosome biogenesis and autophagy. Mol Asp Med 27:495–502
Fanin M, Nascimbeni AC, Fulizio L, Spinazzi M, Melacini P, Angelini C (2006) Generalized lysosome-associated membrane protein-2 defect explains multisystem clinical involvement and allows leukocyte diagnostic screening in Danon disease. Am J Pathol 168:1309–1320
Fujiwara Y, Furuta A, Kikuchi H, et al (2013) Discovery of a novel type of autophagy targeting RNA. Autophagy 9(3):403–409
Huynh KK, Eskelinen EL, Scott CC, Malevanets A, Saftig P, Grinstein S (2007) LAMP proteins are required for fusion of lysosomes with phagosomes. EMBO J 26:313–324
Konecki DS, Foetisch K, Zimmer KP, Schlotter M, Lichter-Konecki U (1995) An alternatively spliced form of the human lysosome-associated membrane protein-2 gene is expressed in a tissue-specific manner. Biochem Biophys Res Commun 215:757–767
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods 25:402–408
Majer F, Vlaskova H, Krol L et al (2012) Danon disease: a focus on processing of the novel LAMP2 mutation and comments on the beneficial use of peripheral white blood cells in the diagnosis of LAMP2 deficiency. Gene 498:183–195
Nishino I, Fu J, Tanji K et al (2000) Primary LAMP-2 deficiency causes X-linked vacuolar cardiomyopathy and myopathy (Danon disease). Nature 406:906–910
Nucifora G, Miani D, Piccoli G, Proclemer A (2012) Cardiac magnetic resonance imaging in Danon disease. Cardiology 121:27–30
Regelsberger G, Hoftberger R, Pickl WF et al (2009) Danon disease: case report and detection of new mutation. J Inherited Metab Dis doi:10.1007/s10545-009-1097-9
Sugie K, Yamamoto A, Murayama K et al (2002) Clinicopathological features of genetically confirmed Danon disease. Neurology 58:1773–1778
Takahashi M, Yamamoto A, Takano K et al (2002) Germline mosaicism of a novel mutation in lysosome-associated membrane protein-2 deficiency (Danon disease). Ann Neurol 52:122–125
Tanaka Y, Guhde G, Suter A et al (2000) Accumulation of autophagic vacuoles and cardiomyopathy in LAMP-2- deficient mice. Nature 406:902–906
Wolach B, Scharf Y, Gavrieli R, de Boer M, Roos D (2005) Unusual late presentation of X-linked chronic granulomatous disease in an adult female with a somatic mosaic for a novel mutation in CYBB. Blood 105:61–66
Yang Z, Funke BH, Cripe LH et al (2010) LAMP2 microdeletions in patients with Danon disease. Circulation 3:129–137
Acknowledgements
Milan Elleder, sadly deceased in September 2011 fundamentally contributed to the LAMP2 protein studies and thus to DD diagnosis in the family described in this manuscript. This work was primarily supported by Research Project no. 0021620806 and the PRVOUK-P24/LF1/3 and MH CZ–DRO VFN64165 projects. OP and TK are supported by MH CR, DRO 00064203 and OPPK CZ.2.16/3.1.00/24022 (University Hospital Motol, Prague, Czech Republic) projects. TP and PK are supported by the PRVOUK-P35/LF1/5 project.
We would like to acknowledge Josef Zamecnik M.D., Ph.D. (Department of Pathology and Molecular Medicine, 2nd Faculty of Medicine, Charles University in Prague, University Hospital Motol in Prague, Czech Republic).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: John Christodoulou
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1
(XLS 38 kb)
Supplementary Table 2
(XLS 36 kb)
Supplementary Fig. 1
(JPEG 131 kb)
Supplementary Fig. 2
(JPEG 143 kb)
Supplementary Fig. 3
(JPEG 93 kb)
Supplementary Fig. 4
(JPEG 17 kb)
Rights and permissions
About this article
Cite this article
Majer, F., Pelak, O., Kalina, T. et al. Mosaic tissue distribution of the tandem duplication of LAMP2 exons 4 and 5 demonstrates the limits of Danon disease cellular and molecular diagnostics. J Inherit Metab Dis 37, 117–124 (2014). https://doi.org/10.1007/s10545-013-9617-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-013-9617-z