Abstract
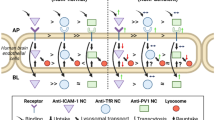
Targeting lysosomal enzymes to receptors involved in transport into and across cells holds promise to enhance peripheral and brain delivery of enzyme replacement therapies (ERTs) for lysosomal storage disorders. Receptors being explored include those associated with clathrin-mediated pathways, yet other pathways seem also viable. Well characterized examples are that of transferrin receptor (TfR) and intercellular adhesion molecule 1 (ICAM-1), involved in iron transport and leukocyte extravasation, respectively. TfR and ICAM-1 support ERT delivery via clathrin- vs. cell adhesion molecule-mediated mechanisms, displaying different valency and size restrictions. To comparatively assess this, we used antibodies vs. larger multivalent antibody-coated carriers and evaluated TfR vs. ICAM-1 binding and endocytosis in endothelial cells, as well as in vivo biodistribution and delivery of a model lysosomal enzyme required in peripheral organs and brain: acid sphingomyelinase (ASM), deficient in types A-B Niemann Pick disease. We found similar binding of antibodies to both receptors under control conditions, with enhanced binding to activated endothelium for ICAM-1, yet only anti-TfR induced endocytosis efficiently. Contrarily, antibody-coated carriers showed enhanced binding, engulfment, and endocytosis for ICAM-1. In mice, anti-TfR enhanced brain targeting over anti-ICAM, with an opposite outcome in the lungs, while carriers enhanced ICAM-1 targeting over TfR in both organs. Both targeted carriers enhanced ASM delivery to the brain and lungs vs. free ASM, with greater enhancement for anti-ICAM carriers. Therefore, targeting TfR or ICAM-1 improves lysosomal enzyme delivery. Yet, TfR targeting may be more efficient for smaller conjugates or fusion proteins, while ICAM-1 targeting seems superior for multivalent carrier formulations.






Similar content being viewed by others
References
Altieri DC, Duperray A, Plescia J, Thornton GB, Languino LR (1995) Structural recognition of a novel fibrinogen gamma chain sequence (117–133) by intercellular adhesion molecule-1 mediates leukocyte-endothelium interaction. J Biol Chem 270(2):696–699
Balyasnikova IV, Metzger R, Visintine DJ et al (2005) Selective rat lung endothelial targeting with a new set of monoclonal antibodies to angiotensin I-converting enzyme. Pulm Pharmacol Ther 18(4):251–267
Banks WA (2009) Blood–brain barrier as a regulatory interface. Forum Nutr 63:102–110
Barreiro O, Zamai M, Yáñez-Mó M et al (2008) Endothelial adhesion receptors are recruited to adherent leukocytes by inclusion in preformed tetraspanin nanoplatforms. J Cell Biol 183(3):527–542
Begley DJ, Pontikis CC, Scarpa M (2008) Lysosomal storage diseases and the blood–brain barrier. Curr Pharm Des 14(16):1566–1580
Boado RJ, Zhang Y, Xia CF, Wang Y, Pardridge WM (2008) Genetic engineering of a lysosomal enzyme fusion protein for targeted delivery across the human blood brain barrier. Biotechnol Bioeng 99(2):475–484
Boado RJ, Zhang Y, Wang Y, Pardridge WM (2009) Engineering and expression of a chimeric transferrin receptor monoclonal antibody for blood–brain barrier delivery in the mouse. Biotechnol Bioeng 102(4):1251–1258
Boado RJ, Hui EK, Lu JZ, Zhou Q, Pardridge WM (2011) Reversal of lysosomal storage in brain of adult MPS-I mice with intravenous Trojan horse-iduronidase fusion protein. Mol Pharm 8(4):1342–1350
Brady RO (2003) Enzyme replacement therapy: conception, chaos and culmination. Philos Trans R Soc Lond B Biol Sci 358(1433):915–919
Calderon AJ, Bhowmick T, Leferovich J et al (2011) Optimizing endothelial targeting by modulating the antibody density and particle concentration of anti-ICAM coated carriers. J Control Release 150(1):37–44
Cardone M, Porto C, Tarallo A et al (2008) Abnormal mannose-6-phosphate receptor trafficking impairs recombinant alpha-glucosidase uptake in Pompe disease fibroblasts. Pathogenetics 1(1):6
Carman CV, Springer TA (2004) A transmigratory cup in leukocyte diapedesis both through individual vascular endothelial cells and between them. J Cell Biol 167(2):377–388
Carman CV, Sage PT, Sciuto TE et al (2007) Transcellular diapedesis is initiated by invasive podosomes. Immunity 26(6):784–797
Carpén O, Pallai P, Staunton DE, Springer TA (1992) Association of intercellular adhesion molecule-1 (ICAM-1) with actin-containing cytoskeleton and alpha-actinin. J Cell Biol 118(5):1223–1234
Chen CH, Dellamaggiore KR, Ouellette CP et al (2008) Aptamer-based endocytosis of a lysosomal enzyme. Proc Natl Acad Sci U S A 105(41):15908–15913
Conrad ME, Umbreit JN (2000) Iron absorption and transport—an update. Am J Hematol 64(4):287–298
Dautry-Varsat A (1986) Receptor-mediated endocytosis: the intracellular journey of transferrin and its receptor. Biochimie 68(3):375–381
DeGraba T, Azhar S, Dignat-George F et al (2000) Profile of endothelial and leukocyte activation in Fabry patients. Ann Neurol 47(2):229–233
Desnick RJ, Schuchman EH (2002) Enzyme replacement and enhancement therapies: lessons from lysosomal disorders. Nat Rev Genet 3(12):954–966
Dhami R, Schuchman EH (2004) Mannose 6-phosphate receptor-mediated uptake is defective in acid sphingomyelinase-deficient macrophages: implications for Niemann-Pick disease enzyme replacement therapy. J Biol Chem 279(2):1526–1532
Dvorak AM, Feng D (2001) The vesiculo-vacuolar organelle (VVO). A new endothelial cell permeability organelle. J Histochem Cytochem 49(4):419–432
Fishman JB, Rubin JB, Handrahan JV, Connor JR, Fine RE (1987) Receptor-mediated transcytosis of transferrin across the blood–brain barrier. J Neurosci Res 18(2):299–304
Frey A, Giannasca KT, Weltzin R et al (1996) Role of the glycocalyx in regulating access of microparticles to apical plasma membranes of intestinal epithelial cells: implications for microbial attachment and oral vaccine targeting. J Exp Med 184(3):1045–1059
Fuchs H, Lucken U et al (1998) Structural model of phospholipid-reconstituted human transferrin receptor derived by electron microscopy. Structure 6(10):1235–1243
Futerman AH, van Meer G (2004) The cell biology of lysosomal storage disorders. Nat Rev Mol Cell Biol 5(7):554–565
Garnacho C, Dhami R, Simone E et al (2008a) Delivery of acid sphingomyelinase in normal and niemann-pick disease mice using intercellular adhesion molecule-1-targeted polymer nanocarriers. J Pharmacol Exp Ther 325(2):400–408
Garnacho C, Albelda SM, Muzykantov VR, Muro S (2008b) Differential intra-endothelial delivery of polymer nanocarriers targeted to distinct PECAM-1 epitopes. J Control Release 130(3):226–233
Garnacho C, Serrano D, Muro S (2012) A fibrinogen-derived peptide provides ICAM-1-specific targeting and intra-endothelial transport of polymer nanocarriers in human cell cultures and mice. J Pharmacol Exp Ther 340(3):638–647
Ghaffarian R, Bhowmick T, Muro S (2012) Transport of nanocarriers across gastrointestinal epithelial cells by a new transcellular route induced by targeting ICAM-1. J Control Release June 12 (Epub ahead of print)
Hatakeyama H, Akita H, Maruyama K et al (2004) Factors governing the in vivo tissue uptake of transferrin-coupled polyethylene glycol liposomes in vivo. Int J Pharm 281(1–2):25–33
He X, Miranda SR, Xiong X, Dagan A, Gatt S, Schuchman EH (1999) Characterization of human acid sphingomyelinase purified from the media of overexpressing Chinese hamster ovary cells. Biochim Biophys Acta 1432(2):251–264
Hillebrand U, Hausberg M, Stock C et al (2006) 17beta-estradiol increases volume, apical surface and elasticity of human endothelium mediated by Na+/H+ exchange. Cardiovasc Res 69(4):916–924
Hirst J, Robinson MS (1998) Clathrin and adaptors. Biochim Biophys Acta 1404(1–2):173–193
Hsu J, Serrano D, Bhowmick T, Muro S (2011) Enhanced endothelial delivery and biochemical effects of alpha-galactosidase by ICAM-1-targeted nanocarriers for Fabry disease. J Control Release 149(3):323–331
Hsu J, Northrup L, Bhowmick T, Muro S (2012) Enhanced delivery of alpha-glucosidase for Pompe disease by ICAM-1-targeted nanocarriers: comparative performance of a strategy for three distinct lysosomal storage disorders. Nanomedicine 8(5):731–739
Jefferies WA, Brandon MR, Hunt SV, Williams AF, Gatter KS, Mason DY (1984) Transferrin receptor on endothelium of brain capillaries. Nature 312(5990):162–163
Jevnikar AM, Wuthrich RP, Takei F et al (1990) Differing regulation and function of ICAM-1 and class II antigens on renal tubular cells. Kidney Int 38(3):417–425
Jin M, Snider MD (1993) Role of microtubules in transferrin receptor transport from the cell surface to endosomes and the Golgi complex. J Biol Chem 268(24):18390–18397
Jun CD, Carman CV, Redick SD, Shimaoka M, Erickson HP, Springer TA (2001) Ultrastructure and function of dimeric, soluble intercellular adhesion molecule-1 (ICAM-1). J Biol Chem 276(31):29019–29027
Kissel K, Hamm S et al (1998) Immunohistochemical localization of the murine transferrin receptor (TfR) on blood–tissue barriers using a novel anti-TfR monoclonal antibody. Histochem Cell Biol 110(1):63–72
Ko YT, Bhattacharya R, Bickel U (2009) Liposome encapsulated polyethylenimine/ODN polyplexes for brain targeting. J Control Release 133(3):230–237
LeBowitz JH, Grubb JH, Maga JA, Schmiel DH, Vogler C, Sly WS (2004) Glycosylation-independent targeting enhances enzyme delivery to lysosomes and decreases storage in mucopolysaccharidosis type VII mice. Proc Natl Acad Sci U S A 101(9):3083–3088
Lossinsky AS, Mossakowski MJ, Pluta R, Wisniewski HM (1995) Intercellular adhesion molecule-1 (ICAM-1) upregulation in human brain tumors as an expression of increased blood–brain barrier permeability. Brain Pathol 5(4):339–344
Lu JZ, Boado RJ, Hui EK, Zhou QH, Pardridge WM (2011) Expression in CHO cells and pharmacokinetics and brain uptake in the Rhesus monkey of an IgG-iduronate-2-sulfatase fusion protein. Biotechnol Bioeng 108(8):1954–1964
Marlin SD, Springer TA (1987) Purified intercellular adhesion molecule-1 (ICAM-1) is a ligand for lymphocyte function-associated antigen 1 (LFA-1). Cell 51(5):813–819
Matzner U, Matthes F, Weigelt C et al (2008) Non-inhibitory antibodies impede lysosomal storage reduction during enzyme replacement therapy of a lysosomal storage disease. J Mol Med 86(4):433–442
Millán J, Hewlett L, Glyn M, Toomre D, Clark P, Ridley AJ (2006) Lymphocyte transcellular migration occurs through recruitment of endothelial ICAM-1 to caveola- and F-actin-rich domains. Nat Cell Biol 8(2):113–123
Mistry PK, Wraight EP, Cox TM (1996) Therapeutic delivery of proteins to macrophages: implications for treatment of Gaucher’s disease. Lancet 348(9041):1555–1559
Moghimi SM, Hunter AC, Murray JC (2001) Long circulating and target-specific nanoparticles: theory to practice. Pharmacol Rev 53(2):283–318
Murciano JC, Muro S, Koniaris L et al (2003) ICAM-directed vascular immunotargeting of antithrombotic agents to the endothelial luminal surface. Blood 101(10):3977–3984
Muro S (2010) New biotechnological and nanomedicine strategies for treatment of lysosomal storage disorders. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2(2):189–204
Muro S, Wiewrodt R, Thomas A et al (2003) A novel endocytic pathway induced by clustering endothelial ICAM-1 or PECAM-1. J Cell Sci 116(Pt 8):1599–1609
Muro S, Gajewski C, Koval M, Muzykantov VR (2005) ICAM-1 recycling in endothelial cells: a novel pathway for sustained intracellular delivery and prolonged effects of drugs. Blood 105(2):650–658
Muro S, Schuchman EH, Muzykantov VR (2006a) Lysosomal enzyme delivery by ICAM-1-targeted nanocarriers bypassing glycosylation- and clathrin-dependent endocytosis. Mol Ther 13(1):135–141
Muro S, Mateescu M, Gajewski C et al (2006b) Control of intracellular trafficking of ICAM-1-targeted nanocarriers by endothelial Na+/H+ exchanger proteins. Am J Physiol Lung Cell Mol Physiol 290(5):L809–L817
Muro S, Garnacho C, Champion JA et al (2008) Control of endothelial targeting and intracellular delivery of therapeutic enzymes by modulating the size and shape of ICAM-1-targeted carriers. Mol Ther 16(8):1450–1458
Nanami M, Ookawara T, Otaki Y et al (2005) Tumor necrosis factor-alpha-induced iron sequestration and oxidative stress in human endothelial cells. Arterioscler Thromb Vasc Biol 25(12):2495–2501
Ohashi T, Iizuka S, Ida H, Eto Y (2008) Reduced alpha-Gal A enzyme activity in Fabry fibroblast cells and Fabry mice tissues induced by serum from antibody positive patients with Fabry disease. Mol Genet Metab 94(3):313–318
Osborn MJ, McElmurry RT, Peacock B, Tolar J, Blazar BR (2008) Targeting of the CNS in MPS-IH using anonviral transferrin-alpha-L-iduronidase fusion gene product. Mol Ther 16(8):1459–1466
Pang Z, Gao H, Yu Y et al (2011) Brain delivery and cellular internalization mechanisms for transferrin conjugated biodegradable polymersomes. Int J Pharm 415(1–2):284–292
Pardridge WM (2010) Biopharmaceutical drug targeting to the brain. J Drug Target 18(3):157–167
Pardridge WM, Boado RJ (2012) Reengineering biopharmaceuticals for targeted delivery across the blood–brain barrier. Methods Enzymol 503:269–292
Prince WS, McCormick LM, Wendt DJ et al (2004) Lipoprotein receptor binding, cellular uptake, and lysosomal delivery of fusions between the receptor-associated protein (RAP) and alpha-L-iduronidase or acid alpha-glucosidase. J Biol Chem 279(33):35037–35046
Rothlein R, Dustin ML, Marlin SD, Springer TA (1986) A human intercellular adhesion molecule (ICAM-1) distinct from LFA-1. J Immunol 137(4):1270–1274
Schnitzer JE (2001) Caveolae: from basic trafficking mechanisms to targeting transcytosis for tissue-specific drug and gene delivery in vivo. Adv Drug Deliv Rev 49(3):265–280
Serrano D, Bhowmick T, Chadha R, Garnacho C, Muro S (2012) Intercellular adhesion molecule 1 engagement modulates sphingomyelinase and ceramide, supporting uptake of drug carriers by the vascular endothelium. Arterioscler Thromb Vasc Biol 32(5):1178–1185
Shen JS, Meng XL, Moore DF, Quirk JM, Shayman JA, Schiffmann R, Kaneski CR (2008) Globotriaosylceramide induces oxidative stress and up-regulates cell adhesion molecule expression in Fabry disease endothelial cells. Mol Genet Metab 95(3):163–168
Shi N, Boado RJ, Pardridge WM (2001) Receptor-mediated gene targeting to tissues in vivo following intravenous administration of pegylated immunoliposomes. Pharm Res 18(8):1091–1095
Steven AC, Hainfeld JF, Wall JS, Steer CJ (1983) Mass distributions of coated vesicles isolated from liver and brain: analysis by scanning transmission electron microscopy. J Cell Biol 97(6):1714–1723
Torchilin VP (2006) Multifunctional nanocarriers. Adv Drug Deliv Rev 58(14):1532–1555
Urayama A, Grubb JH, Sly WS, Banks WA (2004) Developmentally regulated mannose 6-phosphate receptor-mediated transport of a lysosomal enzyme across the blood–brain barrier. Proc Natl Acad Sci U S A 101(34):12658–12663
Vaags AK, Campbell TN, Choy FY (2005) HIV TAT variants differentially influence the production of glucocerebrosidase in Sf9 cells. Genet Mol Res 4(3):491–495
van Rooy I, Mastrobattista E, Storm G, Hennink WE, Schiffelers RM (2011) Comparison of five different targeting ligands to enhance accumulation of liposomes into the brain. J Control Release 150(1):30–36
Visser CC, Voorwinden LH, Crommelin DJ, Danhof M, de Boer AG (2004) Characterization and modulation of the transferrin receptor on brain capillary endothelial cells. Pharm Res 21(5):761–769
Xia H, Anderson B, Mao Q, Davidson BL (2000) Recombinant human adenovirus: targeting to the human transferrin receptor improves gene transfer to brain microcapillary endothelium. J Virol 74(23):11359–11366
Xia H, Mao Q, Davidson BL (2001) The HIV Tat protein transduction domain improves the biodistribution of beta-glucuronidase expressed from recombinant viral vectors. Nat Biotechnol 19(7):640–644
Zhang XY, Dinh A, Cronin J, Li SC, Reiser J (2008) Cellular uptake and lysosomal delivery of galactocerebrosidase tagged with the HIV Tat protein transduction domain. J Neurochem 104(4):1055–1064
Zhou QH, Boado RJ, Lu JZ, Hui EK, Pardridge WM (2012) Brain-penetrating IgG-iduronate 2-sulfatase fusion protein for the mouse. Drug Metab Dispos 40(2):329–335
Acknowledgments
This study was funded by grants from the American Heart Association (09BGIA2450014) and National Institutes of Health (R01 HL098416) to SM.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Frits Wijburg
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
Fluorescence microscopy of antibody binding to ICAM-1 vs. TfR on endothelial cells. Control or TNFα-activated HUVECs were incubated with anti-ICAM or anti-TfR for 15 min at 37 °C. Cells were washed to remove unbound antibody, fixed, and stained with FITC-conjugated goat anti-mouse IgG. Phase-contrast images were used to delimit cell borders (dashed lines). Scale bar = 10 μm. (JPEG 43 kb)
Supplementary Fig. 2
Fluorescence microscopy of carrier binding to ICAM-1 vs. TfR on endothelial cells. Binding of ∼250 nm FITC-labeled anti-ICAM vs. anti-TfR carriers to control or TNFα-activated HUVECs after 1 h incubation. Cells were subsequently washed to remove unbound carriers. Phase-contrast images were used to delimit cell borders (dashed lines). Scale bar = 10 μm. (JPEG 36 kb)
Supplementary Fig. 3
Fluorescence microscopy of endocytosis of antibodies targeted to endothelial ICAM-1 vs. TfR. Uptake of anti-ICAM vs. anti-TfR by TNFα-activated HUVECs, assessed after 1 h incubation at 37 °C. Unbound antibodies were washed off and surface-bound antibodies were stained with a Texas-Red secondary IgG. Cells were then permeabilized and antibodies located both at the surface + internalized were stained with FITC-labeled secondary IgG. Internalized antibodies appear as FITC single-labeled in green, while surface-bound antibodies display FITC + Texas-Red double-labeled yellow color. Phase-contrast images were used to delimit cell borders (dashed lines). Scale bar = 10 μm. (JPEG 23 kb)
Supplementary Fig. 4
Fluorescence microscopy of endocytosis of carriers targeted to endothelial ICAM-1 vs. TfR. Uptake of ∼250 nm FITC-labeled anti-ICAM vs. anti-TfR carriers, assessed after 1 h incubation at 37 °C with control or TNFα-activated HUVECs. Unbound carriers were removed and surface-bound carriers were stained with a Texas-Red secondary IgG. Internalized carriers appear as FITC single-labeled in green, while surface-bound carriers display FITC + Texas-Red double-labeled yellow color. Phase-contrast images were used to delimit cell borders (dashed lines). Scale bar = 10 μm. (JPEG 53 kb)
Rights and permissions
About this article
Cite this article
Papademetriou, J., Garnacho, C., Serrano, D. et al. Comparative binding, endocytosis, and biodistribution of antibodies and antibody-coated carriers for targeted delivery of lysosomal enzymes to ICAM-1 versus transferrin receptor. J Inherit Metab Dis 36, 467–477 (2013). https://doi.org/10.1007/s10545-012-9534-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-012-9534-6