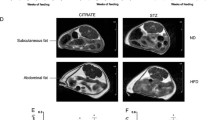
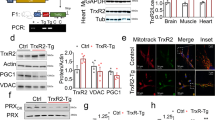
Summary
Diabetes mellitus is occasionally observed in patients with skeletal muscle respiratory chain deficiency, suggesting that skeletal muscle mitochondrial dysfunction might play a pathogenic role in type 2 diabetes (T2D). In support of this hypothesis, decreased muscle mitochondrial activity has been reported in T2D patients and in mouse models of diabetes. However, recent work by several groups suggests that decreased muscle mitochondrial function may be a consequence rather than a cause of diabetes, since decreased mitochondrial function in mice affords protection from diabetes and obesity. We review the data on this controversial but important issue of potential links between mitochondrial dysfunction and diabetes.



Similar content being viewed by others
Abbreviations
- AIF:
-
apoptosis-inducing factor
- HNF-1α:
-
hepatocyte nuclear factor-1α
- IMCL:
-
intramyocellular lipid
- MELAS:
-
mitochondrial encephalopathy, lactic acidosis, and stroke-like episodes
- MIDD:
-
maternally inherited diabetes and deafness syndrome
- MODY:
-
maturity-onset diabetes of the young
- PGC-1α:
-
peroxisome proliferator-activated receptor γ co-activator 1α
- PPAR:
-
peroxisome proliferator-activated receptor
- T2D:
-
type 2 diabetes
- TRMA:
-
thiamine-responsive megaloblastic anaemia
- UCP2:
-
uncoupling protein 2
References
Affourtit C, Brand MD (2008) On the role of uncoupling protein--2 in pancreatic beta cells. Biochim Biophys Acta 1777:973–979
Ashcroft E, Ashcroft S (1992) In: Ashcroft E, Ashcroft S (eds). Insulin: Molecular biology to pathology. OUP, Oxford, pp 97–150
Asmann YW, Stump CS, Short KR et al (2006) Skeletal muscle mitochondrial functions, mitochondrial DNA copy numbers, and gene transcript profiles in type 2 diabetic and nondiabetic subjects at equal levels of low or high insulin and euglycemia. Diabetes 55:3309–3319
Ballinger SW, Shoffner JM, Hedaya EV et al (1992) Maternally transmitted diabetes and deafness associated with a 10.4 kb mitochondrial DNA deletion. Nat Genet 1:11–15
Ballinger SW, Shoffner JM, Gebhart S, Koontz DA, Wallace DC (1994) Mitochondrial diabetes revisited. Nat Genet 7:458–459
Barker DJ, Hales CN, Fall CH, Osmond C, Phipps K, Clark PM (1993) Type 2 (non--insulin--dependent) diabetes mellitus, hypertension and hyperlipidaemia (syndrome X): relation to reduced fetal growth. Diabetologia 36:62–67
Bénit P, Goncalves S, Dassa EP, Brière JJ, Rustin P (2008) The variability of the Harlequin mouse phenotype resembles that of human mitochondrial--complex I--deficiency syndromes. PloS One 3:e3208
Bonnard C, Durand A, Peyrol S et al (2008) Mitochondrial dysfunction results from oxidative stress in the skeletal muscle of diet--induced insulin--resistant mice. J Clin Invest 118:789–800
Boubaker K, Flepp M, Sudre P et al (2001) Hyperlactatemia and antiretroviral therapy: the Swiss HIV Cohort Study. Clin Infect Dis 33:1931–1937
Boushel R, Gnaiger E, Schjerling P, Skovbro M, Kraunsoe R, Dela F (2007) Patients with type 2 diabetes have normal mitochondrial function in skeletal muscle. Diabetologia 50:790–796
Campuzano V, Montermini L, Molto MD et al (1996) Friedreich’s ataxia: autosomal recessive disease caused by an intronic GAA triplet repeat expansion. Science 271:1423–1427
Casteels K, Ong K, Phillips D, Bendall H, Pembrey M (1999) Mitochondrial 16189 variant, thinness at birth, and type-2 diabetes. ALSPAC study team. Avon Longitudinal Study of Pregnancy and Childhood. Lancet 353:1499–1500
Civitarese AE, Carling S, Heilbronn LK et al (2007) Calorie restriction increases muscle mitochondrial biogenesis in healthy humans. PLoS Med 4:e76
Cree LM, Patel SK, Pyle A et al (2008) Age-related decline in mitochondrial DNA copy number in isolated human pancreatic islets. Diabetologia 51:1440–1443
Daneman D (2006) Type 1 diabetes. Lancet 367:847–858
De Feyter HM, van den Broek NM, Praet SF, Nicolay K, van Loon LJ, Prompers JJ (2008a) Early or advanced stage type 2 diabetes is not accompanied by in vivo skeletal muscle mitochondrial dysfunction. Eur J Endocrinol 158:643–653
De Feyter HM, Lenaers E, Houten SM et al (2008b) Increased intramyocellular lipid content but normal skeletal muscle mitochondrial oxidative capacity throughout the pathogenesis of type 2 diabetes. FASEB J 22:3947–3955
Dimauro S, Davidzon G (2005) Mitochondrial DNA and disease. Ann Med 37:222–232
Doria A, Patti ME, Kahn CR (2008) The emerging genetic architecture of type 2 diabetes. Cell Metab 8:186–200
Dunbar DR, Moonie PA, Swingler RJ, Davidson D, Roberts R, Holt IJ (1993) Maternally transmitted partial direct tandem duplication of mitochondrial DNA associated with diabetes mellitus. Hum Mol Genet 2:1619–1624
Fleming JC, Tartaglini E, Steinkamp MP, Schorderet DF, Cohen N, Neufeld EJ (1999) The gene mutated in thiamine-responsive anaemia with diabetes and deafness (TRMA) encodes a functional thiamine transporter. Nat Genet 22:305–308
Freyer C, Larsson NG (2007) Is energy deficiency good in moderation? Cell 131:448–450
Garcia-Roves P, Huss JM, Han DH et al (2007) Raising plasma fatty acid concentration induces increased biogenesis of mitochondria in skeletal muscle. Proc Natl Acad Sci USA 104:10709–10713
Guarente L (2008) Mitochondria—a nexus for aging, calorie restriction, and sirtuins? Cell 132:171–176
Hammer S, Snel M, Lamb HJ et al (2008) Prolonged caloric restriction in obese patients with type 2 diabetes mellitus decreases myocardial triglyceride content and improves myocardial function. J Am Coll Cardiol 52:1006–1012
Hancock CR, Han DH, Chen M et al (2008) High-fat diets cause insulin resistance despite an increase in muscle mitochondria. Proc Natl Acad Sci USA 105:7815–7820
Harding AE (1981) Friedreich’s ataxia: a clinical and genetic study of 90 families with an analysis of early diagnostic criteria and intrafamilial clustering of clinical features. Brain 104:589–620
Herzig S, Long F, Jhala US et al (2001) CREB regulates hepatic gluconeogenesis through the coactivator PGC-1. Nature 413:179–183
Hoeks J, Briede JJ, de Vogel J et al (2008) Mitochondrial function, content and ROS production in rat skeletal muscle: effect of high-fat feeding. FEBS Lett 582:510–516
Holloszy JO (1967) Biochemical adaptations in muscle. Effects of exercise on mitochondrial oxygen uptake and respiratory enzyme activity in skeletal muscle. J Biol Chem 242:2278–2282
Holloszy JO (2009) Skeletal muscle “mitochondrial deficiency” does not mediate insulin resistance. Am J Clin Nutr 89:463S–466S
Hoppeler H, Fluck M (2003) Plasticity of skeletal muscle mitochondria: structure and function. Med Sci Sports Exerc 35:95–104
Kelley DE, He J, Menshikova EV, Ritov VB (2002) Dysfunction of mitochondria in human skeletal muscle in type 2 diabetes. Diabetes 51:2944–2950
Khan AH, Pessin JE (2002) Insulin regulation of glucose uptake: a complex interplay of intracellular signalling pathways. Diabetologia 45:1475–1483
Koo SH, Satoh H, Herzig S et al (2004) PGC-1 promotes insulin resistance in liver through PPAR-alpha-dependent induction of TRB-3. Nat Med 10:530–534
Krishnan KJ, Greaves LC, Reeve AK, Turnbull D (2007) The ageing mitochondrial genome. Nucleic Acids Res 35:7399–7405
Kroemer G (2001) Mitochondrial control of apoptosis. Bull Acad Natl Med 185:1135–1142; discussion 1143
Krssak M, Falk Petersen K, Dresner A et al (1999) Intramyocellular lipid concentrations are correlated with insulin sensitivity in humans: a 1H NMR spectroscopy study. Diabetologia 42:113–116
Kuhlmann J, Neumann-Haefelin C, Belz U et al (2003) Intramyocellular lipid and insulin resistance: a longitudinal in vivo 1H-spectroscopic study in Zucker diabetic fatty rats. Diabetes 52:138–144
Labay V, Raz T, Baron D et al (1999) Mutations in SLC19A2 cause thiamine-responsive megaloblastic anaemia associated with diabetes mellitus and deafness. Nat Genet 22:300–304
Lagouge M, Argmann C, Gerhart-Hines Z et al (2006) Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1alpha. Cell 127:1109–1122
Lazar MA (2005) How obesity causes diabetes: not a tall tale. Science 307:373–375
Li X, Monks B, Ge Q, Birnbaum MJ (2007) Akt/PKB regulates hepatic metabolism by directly inhibiting PGC-1alpha transcription coactivator. Nature 447:1012–1016
Lin J, Wu PH, Tarr PT et al (2004) Defects in adaptive energy metabolism with CNS-linked hyperactivity in PGC-1alpha null mice. Cell 119:121–135
Lin J, Handschin C, Spiegelman BM (2005) Metabolic control through the PGC-1 family of transcription coactivators. Cell Metab 1:361–370
Lowell BB, Shulman GI (2005) Mitochondrial dysfunction and type 2 diabetes. Science 307:384–387
Maassen JA, ‘T Hart LM, Van Essen E et al (2004) Mitochondrial diabetes: molecular mechanisms and clinical presentation. Diabetes 53(Suppl 1):103–109
Maassen JA, Janssen GM, t Hart LM (2005) Molecular mechanisms of mitochondrial diabetes (MIDD). Ann Med 37:213–221
Maassen JA, Jahangir Tafrechi RS, Janssen GM, Raap AK, Lemkes HH, t Hart LM (2006a) New insights in the molecular pathogenesis of the maternally inherited diabetes and deafness syndrome. Endocrinol Metab Clin North Am 35:385–396, x–xi
Maassen JA, ‘t Hart LM, Janssen GM, Reiling E, Romijn JA, Lemkes HH (2006b) Mitochondrial diabetes and its lessons for common Type 2 diabetes. Biochem Soc Trans 34:819–823
Maassen JA, t Hart LM, Ouwens DM (2007) Lessons that can be learned from patients with diabetogenic mutations in mitochondrial DNA: implications for common type 2 diabetes. Curr Opin Clin Nutr Metab Care 10:693–697
Maechler P, de Andrade PB (2006) Mitochondrial damages and the regulation of insulin secretion. Biochem Soc Trans 34:824–827
Maechler P, Wollheim CB (2001) Mitochondrial function in normal and diabetic beta-cells. Nature 414:807–812
Majander A, Suomalainen A, Vettenranta K et al (1991) Congenital hypoplastic anemia, diabetes, and severe renal tubular dysfunction associated with a mitochondrial DNA deletion. Pediatr Res 30:327–330
Matschinsky F, Liang Y, Kesavan P et al (1993) Glucokinase as pancreatic beta cell glucose sensor and diabetes gene. J Clin Invest 92:2092–2098
Mensink M, Hesselink MK, Russell AP, Schaart G, Sels JP, Schrauwen P (2007) Improved skeletal muscle oxidative enzyme activity and restoration of PGC-1 alpha and PPAR beta/delta gene expression upon rosiglitazone treatment in obese patients with type 2 diabetes mellitus. Int J Obes (Lond) 31:1302–1310
Misu H, Takamura T, Matsuzawa N et al (2007) Genes involved in oxidative phosphorylation are coordinately upregulated with fasting hyperglycaemia in livers of patients with type 2 diabetes. Diabetologia 50:268–277
Mogensen M, Sahlin K, Fernstrom M et al (2007) Mitochondrial respiration is decreased in skeletal muscle of patients with type 2 diabetes. Diabetes 56:1592–1599
Mootha VK, Lindgren CM, Eriksson KF et al (2003) PGC-1alpha-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat Genet 34:267–273
Morino K, Petersen KF, Dufour S et al (2005) Reduced mitochondrial density and increased IRS-1 serine phosphorylation in muscle of insulin-resistant offspring of type 2 diabetic parents. J Clin Invest 115:3587–3593
Morten K, Field P, Ashley N et al (2005) Fetal and neonatal exposure to AZT and low-protein diet affects glucose homeostasis: a model with implications for AIDS prevention. Am J Physiol Endocrinol Metab 289:E1115–1118
Mulder H, Ling C (2009) Mitochondrial dysfunction in pancreatic beta-cells in Type 2 diabetes. Mol Cell Endocrinol 297:34–40
Muoio DM, Newgard CB (2008) Mechanisms of disease: molecular and metabolic mechanisms of insulin resistance and beta-cell failure in type 2 diabetes. Nat Rev Mol Cell Biol 9:193–205
Murphy R, Turnbull DM, Walker M, Hattersley AT (2008) Clinical features, diagnosis and management of maternally inherited diabetes and deafness (MIDD) associated with the 3243A>G mitochondrial point mutation. Diabet Med 25:383–399
Nair KS, Bigelow ML, Asmann YW et al (2008) Asian Indians have enhanced skeletal muscle mitochondrial capacity to produce ATP in association with severe insulin resistance. Diabetes 57:1166–1175
Park KS, Chan JC, Chuang LM et al (2008) A mitochondrial DNA variant at position 16189 is associated with type 2 diabetes mellitus in Asians. Diabetologia 51:602–608
Patti ME, Butte AJ, Crunkhorn S et al (2003) Coordinated reduction of genes of oxidative metabolism in humans with insulin resistance and diabetes: Potential role of PGC1 and NRF1. Proc Natl Acad Sci USA 100:8466–8471
Paupe V, Dassa EP, Goncalves S et al (2009) Impaired nuclear Nrf2 translocation undermines the oxidative stress response in Friedreich ataxia. PLoS ONE 4:e4253
Petersen KF, Shulman GI (2006) Etiology of insulin resistance. Am J Med 119:S10–16
Petersen KF, Befroy D, Dufour S et al (2003) Mitochondrial dysfunction in the elderly: possible role in insulin resistance. Science 300:1140–1142
Petersen KF, Dufour S, Befroy D, Garcia R, Shulman GI (2004) Impaired mitochondrial activity in the insulin-resistant offspring of patients with type 2 diabetes. N Engl J Med 350:664–671
Pospisilik JA, Knauf C, Joza N et al (2007) Targeted deletion of AIF decreases mitochondrial oxidative phosphorylation and protects from obesity and diabetes. Cell 131:476–491
Poulton J, Holt IJ (1994) Mitochondrial DNA: does more lead to less? Nat Genet 8:313–315
Poulton J, Deadman ME, Gardiner RM (1989) Duplications of mitochondrial DNA in mitochondrial myopathy. Lancet 1:236–240
Poulton J, O’Rahilly S, Morten KJ, Clark A (1995) Mitochondrial DNA, diabetes and pancreatic pathology in Kearns-Sayre syndrome. Diabetologia 38:868–871
Puigserver P, Rhee J, Donovan J et al (2003) Insulin-regulated hepatic gluconeogenesis through FOXO1-PGC-1alpha interaction. Nature 423:550–555
Reardon W, Ross RJ, Sweeney MG et al (1992) Diabetes mellitus associated with a pathogenic point mutation in mitochondrial DNA. Lancet 340:1376–1379
Richardson DK, Kashyap S, Bajaj M et al (2005) Lipid infusion decreases the expression of nuclear encoded mitochondrial genes and increases the expression of extracellular matrix genes in human skeletal muscle. J Biol Chem 280:10290–10297
Ristow M, Mulder H, Pomplun D et al (2003) Frataxin deficiency in pancreatic islets causes diabetes due to loss of beta cell mass. J Clin Invest 112:527–534
Ritov VB, Menshikova EV, He J, Ferrell RE, Goodpaster BH, Kelley DE (2005) Deficiency of subsarcolemmal mitochondria in obesity and type 2 diabetes. Diabetes 54:8–14
Roden M, Petersen KF, Shulman GI (2001) Nuclear magnetic resonance studies of hepatic glucose metabolism in humans. Recent Prog Horm Res 56:219–237
Rotig A, Bessis JL, Romero N et al (1992) Maternally inherited duplication of the mitochondrial genome in a syndrome of proximal tubulopathy, diabetes mellitus, and cerebellar ataxia. Am J Hum Genet 50:364–370
Rotig A, de Lonlay P, Chretien D et al (1997) Aconitase and mitochondrial iron-sulphur protein deficiency in Friedreich ataxia. Nat Genet 17:215–217
Saltiel AR, Kahn CR (2001) Insulin signalling and the regulation of glucose and lipid metabolism. Nature 414:799–806
Schrauwen-Hinderling VB, Roden M, Kooi ME, Hesselink MK, Schrauwen P (2007) Muscular mitochondrial dysfunction and type 2 diabetes mellitus. Curr Opin Clin Nutr Metab Care 10:698–703
Schrauwen-Hinderling VB, Mensink M, Hesselink MK, Sels JP, Kooi ME, Schrauwen P (2008) The insulin-sensitizing effect of rosiglitazone in type 2 diabetes mellitus patients does not require improved in vivo muscle mitochondrial function. J Clin Endocrinol Metab 93:2917–2921
Semple RK, Chatterjee VK, O’Rahilly S (2006) PPAR gamma and human metabolic disease. J Clin Invest 116:581–589
Silva JP, Kohler M, Graff C et al (2000) Impaired insulin secretion and beta-cell loss in tissue-specific knockout mice with mitochondrial diabetes. Nat Genet 26:336–340
Simmons RA, Suponitsky-Kroyter I, Selak MA (2005) Progressive accumulation of mitochondrial DNA mutations and decline in mitochondrial function lead to beta-cell failure. J Biol Chem 280:28785–28791
Soejima A, Inoue K, Takai D et al (1996) Mitochondrial DNA is required for regulation of glucose-stimulated insulin secretion in a mouse pancreatic beta cell line, MIN6. J Biol Chem 271:26194–26199
Sparks LM, Xie H, Koza RA et al (2005) A high-fat diet coordinately downregulates genes required for mitochondrial oxidative phosphorylation in skeletal muscle. Diabetes 54:1926–1933
Stump CS, Short KR, Bigelow ML, Schimke JM, Nair KS (2003) Effect of insulin on human skeletal muscle mitochondrial ATP production, protein synthesis, and mRNA transcripts. Proc Natl Acad Sci U S A 100:7996–8001
Stumvoll M, Goldstein BJ, van Haeften TW (2005) Type 2 diabetes: principles of pathogenesis and therapy. Lancet 365:1333–1346
Szendroedi J, Schmid AI, Chmelik M et al (2007) Muscle mitochondrial ATP synthesis and glucose transport/phosphorylation in type 2 diabetes. PLoS Med 4:e154
‘t Hart LM, Ruige JB, Dekker JM, Stehouwer CD, Maassen JA, Heine RJ (1999) Altered beta-cell characteristics in impaired glucose tolerant carriers of a GAA trinucleotide repeat polymorphism in the frataxin gene. Diabetes 48:924–926
‘t Hart LM, Hansen T, Rietveld I et al (2005) Evidence that the mitochondrial leucyl tRNA synthetase (LARS2) gene represents a novel type 2 diabetes susceptibility gene. Diabetes 54:1892–1895
Toledo FG, Menshikova EV, Ritov VB et al (2007) Effects of physical activity and weight loss on skeletal muscle mitochondria and relationship with glucose control in type 2 diabetes. Diabetes 56:2142–2147
Tontonoz P, Spiegelman BM (2008) Fat and beyond: the diverse biology of PPARgamma. Annu Rev Biochem 77:289–312
Trifunovic A, Larsson NG (2008) Mitochondrial dysfunction as a cause of ageing. J Intern Med 263:167–178
Trifunovic A, Wredenberg A, Falkenberg M et al (2004) Premature ageing in mice expressing defective mitochondrial DNA polymerase. Nature 429:417–423
Turner N, Bruce CR, Beale SM et al (2007) Excess lipid availability increases mitochondrial fatty acid oxidative capacity in muscle: evidence against a role for reduced fatty acid oxidation in lipid-induced insulin resistance in rodents. Diabetes 56:2085–2092
Vahsen N, Cande C, Briere JJ et al (2004) AIF deficiency compromises oxidative phosphorylation. Embo J 23:4679–4689
van den Ouweland JM, Lemkes HH, Ruitenbeek W et al (1992) Mutation in mitochondrial tRNA(Leu)(UUR) gene in a large pedigree with maternally transmitted type II diabetes mellitus and deafness. Nat Genet 1:368–371
Wang H, Antinozzi PA, Hagenfeldt KA, Maechler P, Wollheim CB (2000) Molecular targets of a human HNF1 alpha mutation responsible for pancreatic beta-cell dysfunction. Embo J 19:4257–4264
Whittaker RG, Schaefer AM, McFarland R, Taylor RW, Walker M, Turnbull DM (2007) Prevalence and progression of diabetes in mitochondrial disease. Diabetologia 50:2085–2089
Williams RS (1986) Mitochondrial gene expression in mammalian striated muscle. Evidence that variation in gene dosage is the major regulatory event. J Biol Chem 261:12390–12394
Wobser H, Dussmann H, Kogel D et al (2002) Dominant-negative suppression of HNF-1 alpha results in mitochondrial dysfunction, INS-1 cell apoptosis, and increased sensitivity to ceramide-, but not to high glucose-induced cell death. J Biol Chem 277:6413–6421
Wredenberg A, Freyer C, Sandstrom ME et al (2006) Respiratory chain dysfunction in skeletal muscle does not cause insulin resistance. Biochem Biophys Res Commun 350:202–207
Yoon JC, Puigserver P, Chen G et al (2001) Control of hepatic gluconeogenesis through the transcriptional coactivator PGC-1. Nature 413:131–138
Zhang D, Liu ZX, Choi CS et al (2007) Mitochondrial dysfunction due to long-chain Acyl-CoA dehydrogenase deficiency causes hepatic steatosis and hepatic insulin resistance. Proc Natl Acad Sci USA 104:17075–17080
Acknowledgement
This work was supported by grants from the AFM (Association Française contre les Myopathies), AMMi (Association contre les Maladies Mitochondriales), AFAF (Association française de l’ataxie de Friedreich), and Leducq foundation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicating editor: Garry Brown
Competing interests: None declared
Rights and permissions
About this article
Cite this article
Schiff, M., Loublier, S., Coulibaly, A. et al. Mitochondria and diabetes mellitus: untangling a conflictive relationship?. J Inherit Metab Dis 32, 684–698 (2009). https://doi.org/10.1007/s10545-009-1263-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-009-1263-0