Summary
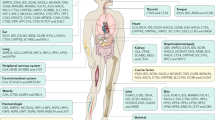
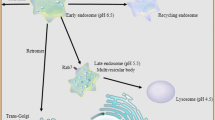
Lysosomal disease represents a large group of more than 50 clinically recognized conditions resulting from inborn errors of metabolism affecting the organelle known as the lysosome. The lysosome is an integral part of the larger endosomal/lysosomal system, and is closely allied with the ubiquitin–proteosomal and autophagosomal systems, which together comprise essential cell machinery for substrate degradation and recycling, homeostatic control, and signalling. More than two-thirds of lysosomal diseases affect the brain, with neurons appearing particularly vulnerable to lysosomal compromise and showing diverse consequences ranging from specific axonal and dendritic abnormalities to neuron death. While failure of lysosomal function characteristically leads to lysosomal storage, new studies argue that lysosomal diseases may also be appropriately viewed as ‘states of deficiency’ rather than simply overabundance (storage). Interference with signalling events and salvage processing normally controlled by the endosomal/lysosomal system may represent key mechanisms accounting for the inherent complexity of lysosomal disorders. Analysis of lysosomal disease pathogenesis provides a unique window through which to observe the importance of the greater lysosomal system for normal cell health.

Similar content being viewed by others
Abbreviations
- AMPA:
-
α-amino-3-hydroxy-5-methylisoxazole-4-proprionic acid
- CMA:
-
chaperone-mediated autophagy
- E/L:
-
endosomal/lysosomal
- ER:
-
endoplasmic reticulum
- GAG:
-
glycosaminoglycan
- GSL:
-
glycosphingolipid
- ML:
-
mucolipidosis
- MPS:
-
mucopolysaccharidosis
- NMDA:
-
N-methyl-d-aspartate
- TGN:
-
trans-Golgi network
- UPS:
-
ubiquitin–proteosomal system
References
Barry MF, Ziff EB (2002) Receptor trafficking and the plasticity of excitatory synapses. Curr Opin Neurobiol 12: 279–286. doi:10.1016/S0959-4388(02)00329-X.
Beutler E, Grabowski G (2001) Gaucher disease. In: Scriver CR, Beaudet AL, Sly WS, Valle D, eds; Childs B, Kinzler KW, Vogelstein B, assoc. eds. The Metabolic and Molecular Bases of Inherited Disease, 8th edn. New York: McGraw-Hill, 3635–3668.
Bredt DS, Nicoll RA (2003) AMPA receptor trafficking at excitatory synapses. Neuron 40: 361–379. doi:10.1016/S0896-6273(03)00640-8.
Bronfman FC, Escudero CA, Weis J, Kruttgen A (2007) Endosomal transport of neurotrophins: Roles in signaling and neurodegenerative disease. Dev Neurobiol 67(9): 1183–1203. doi:10.1002/dneu.20513.
Cao Y, Espinola JA, Fossale E, et al (2006) Autophagy is disrupted in a knock-in mouse model of juvenile neuronal ceroid lipofuscinosis. J. Biol Chem 281: 20483–20493. doi:10.1074/jbc.M602180200.
Cuervo AM (2004) Autophagy: in sickness and in health. Trends Cell Biol 14: 70–77. doi:10.1016/j.tcb.2003.12.002.
d’Azzo A, Tessitore A, Sano R (2006) Gangliosides as apoptotic signals in ER stress response. Cell Death Differ 13 : 404–413.
deVeber GA, Schwarting GA, Kolodny EH, Kowall NW (1992) Fabry disease: immunocytochemical characterization of neuronal involvement. Ann Neurol. 31: 409–415.
Desnick RJ, Thorpe SR, Fiddler M (1976) Toward enzyme therapy for lysosomal storage diseases. Physiol Rev 56: 57–99.
Garrod AE (1909) Inborn Errors of Metabolism. London: Oxford University Press.
Ginzburg L, Kacher Y, Futerman AH (2004) The pathogenesis of glycosphingolipid storage disorders. Semi Cell Dev Biol 15: 417–431.
Gondré-Lewis M, Walkley SU (2003) Cholesterol accumulation in neurons with NPC1 dysfunction is ganglioside-dependent. Curr Biol 13: 1324–1329.
Griffin LD, Gong W, Verot L, Mellon SH (2004) Niemann–Pick type C disease involves disrupted neurosteroidogenesis and responds to allopregnanolone. Nat Med 10: 704–711.
Hara T, Nakamura K, Matsui M, et al (2006) Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature 441: 885–889.
Hers HG (1965) Progress in gastroenterology: inborn lysosomal diseases. Gastroenterology 48: 625–633.
Howe CL, Mobley WC (2005) Long-distance retrograde neurotrophic signaling. Curr Opin Neurobiol 15: 40–48.
Isaac JTR, Ashby MC, McBain CJ (2007) The role of the GluR2 subunit in AMPA receptor function and synaptic plasticity. Neuron 54: 859–871.
Jou I, Lee JH, Park SY, Yoon HJ, Joe E-H, Park EJ (2006) Gangliosides trigger inflammatory responses via TLR4 in brain glia. Am J Pathol 168: 1619–1630.
Kolter T, Sandhoff K (2005) Principles of lysosomal membrane digestion: Stimulation of sphingolipid degradation by sphingolipid activator proteins and anionic lysosomal lipids. Annu Rev Cell Dev Biol 21: 81–103.
Komatsu M, Waguri S, Chiba T, et al (2006) Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature 441: 880–884.
Kyttälä A, Lahtinen U, Braulke T, Hofmann SL (2006) Functional biology of the neuronal ceroid lipofuscinoses (NCL) proteins. Biochim Biophys Acta 1762: 920–933.
McAllister KA (2001) Cellular and molecular mechanisms of dendrite growth. Cerebral Cortex 10: 963–973.
McGlynn R, Dobrenis K, Walkley SU (2004) Differential subcellular localization of cholesterol, gangliosides and glycosaminoglycans in murine models of mucopolysaccharide storage disorders. J Comp Neurol 480: 415–426.
Neufeld E, Muenzer J (2001) The mucopolysaccharidoses. In: Scriver CR, Beaudet AL, Sly WS, Valle D, eds; Childs B, Kinzler KW, Vogelstein B, assoc. eds. The Metabolic and Molecular Bases of Inherited Disease, 8th edn. New York: McGraw-Hill, 3421–3452.
Pacheco CD, Lieberman AP (2007) Lipid trafficking defects increase beclin-1 and activate autophagy in Niemann–Pick type C disease. Autophagy 3: 487–489.
Pacheco CD, Kunkel R, Lieberman AP (2007) Autophagy in Niemann–Pick C disease is dependent upon Beclin-1 and responsive to lipid trafficking defects. Hum Mol Genet 16: 1495–1503.
Passafaro M, Nakagawa T, Sala C, Sheng M (2003) Induction of dendritic spines by an extracellular domain of AMPA receptor subunit GluR2. Nature 424: 677–681.
Pearce DA, Nosel SA, Herman F (1999) Studies of pH regulation by Btn1p, the yeast homolog of human Cln3p. Mol Genet Metab 66: 320–323.
Pentchev PG, Blanchette-Mackie E, Dawidowicz EA (1994) The NP-C gene: a key to pathways of intracellular cholesterol transport. Trends Cell Biol 4: 365–369.
Platt FM, Walkley SU (2004) Lysosomal defects and storage, In: Platt FM, Walkley SU, eds. Lysosomal Disorders of the Brain. Oxford: Oxford University Press, Chapter 2, 32–49.
Pol S, Di Fiore PP (2006) Endocytosis conducts the cell signaling orchestra. Cell 124: 897–900.
Rakheja D, Narayan SB, Bennett MJ (2008) The function of CLN3p, the Batten disease protein. Mol Gen Metab 93: 269–274.
Ravikumar BR, Rubinsztein DC (2006) Role of autophagy in the clearance of mutant huntingtin: a step toward therapy? Mol Aspects Med 27: 520–527.
Schwarzmann G, Sandhoff K (1990) Metabolism and intracellular transport of glycosphingolipids. Biochemistry 29: 10865–10871.
Settembre C, Fraldi A, Jahreiss L, et al (2008) A block of autophagy in lysosomal storage disorders. Hum Mol Genet 17: 119–129.
Tayebi N, Walker J, Stubblefield B, et al (2003) Gaucher disease with parkinsonian manifestations: does glucocerebrosidase deficiency contribute to a vulnerability to parkinsonism? Mol Genet Metab 79: 104–109.
Tettamanti G, Bassi R, Viani P, Riboni L (2003) Salvage pathways in glycosphingolipid metabolism. Biochemie 85: 432–437.
Walkley SU (2003) Neurobiology and cellular pathogenesis of glycolipid storage diseases. Trans R Soc B 358: 893–904.
Walkley SU (2004a) Pathogenic cascades and brain dysfunction, In: Platt FM, Walkley SU, eds. Lysosomal Disorders of the Brain. Oxford: Oxford University Press, Chapter 12, 290–324.
Walkley SU (2004b) Secondary accumulation of gangliosides in lysosomal storage disorders. Semin Cell Biol Dev 15: 433–444.
Walkley SU (2007) Pathogenic mechanisms in lysosomal disease: a reappraisal of the role of the lysosome. Acta Paediatr 96(s455): 26–32.
Walkley SU, Suzuki K (2004) Consequences of NPC1 and NPC2 loss of function in neurons. Biochim Biophys Acta 1685: 48–62.
Walkley SU, Baker HJ, Rattazzi MC, Haskins ME, Wu J-Y (1991) Neuroaxonal dystrophy in neuronal storage disorders: evidence for major GABAergic neuron involvement. J Neurol Sci 104: 1–8.
Walkley SU, Zervas M, Wiseman S (2000) Gangliosides as modulators of dendritogenesis in storage disease-affected and normal pyramidal neurons. Cerebral Cortex 10: 1028–1037.
Wenger DA, Suzuki K, Suzuki Y, Suzuki K (2001) Galactosylceramide lipidosis: Globoid cell leukodystrophy (Krabbe disease). In: Scriver CR, Beaudet AL, Sly WS, Valle D, eds; Childs B, Kinzler KW, Vogelstein B, assoc. eds. The Metabolic and Molecular Bases of Inherited Disease, 8th edn. New York: McGraw-Hill, 3669–3694.
Woloszynek JC, Coleman T, Semenkovich CF, Sands MS (2007) Lysosomal dysfunction results in altered energy balance. J. Biol Chem 282: 35765–35771.
Zeevi DA, Frumkin A, Bach G (2007) TRPML and lysosomal function. Biochim Biophys Acta 1772: 851–858.
Acknowledgements
This work was supported by grants from the NIH (HD045561 and NS053677), the Ara Parseghian Medical Research Foundation and Dana’s Angel’s Research Trust.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicating editor: Gregory McCarthy Pastores
Competing interests: None declared
Rights and permissions
About this article
Cite this article
Walkley, S.U. Pathogenic cascades in lysosomal disease—Why so complex?. J Inherit Metab Dis 32, 181–189 (2009). https://doi.org/10.1007/s10545-008-1040-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-008-1040-5