Summary
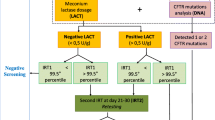
Newborn screening for cystic fibrosis has been carried out for over 25 years, and clinical and cost benefits have been documented. There is still much variation in the methods and strategies adopted. All current screening programmes use a measurement of immunoreactive trypsin as a primary screening test, and in most, a second tier test involves analysing DNA mutations. The choice of DNA mutations depends on the genetic background in the region, and considerations of cost. Using DNA analysis as part of a screening procedure has introduced unwanted carrier detection, and protocols have now been devised in an attempt to avoid this. There are at least seven distinct protocols in use, all of which have different advantages and disadvantages, and no method or strategy will suit every region. Further careful study of performance and costs of various strategies is needed.
Similar content being viewed by others
References
Bombieri C, Pignatti PF (2001) Cystic fibrosis mutation testing in Italy. Genet Test 5: 229–233.
Bowling FG, Watson AR, Rylatt DB, et al (1987) Monoclonal antibody-based enzyme immunoassay for trypsinogen in neonatal screening for cystic fibrosis. Lancet 1(8537): 826–827.
Brock DJ (1990) A consortium approach to molecular genetic services. Scottish Molecular Genetics Consortium. J Med Genet 27: 8–13.
Castellani C, Bonizzato A, Cabrini G, et al (1997) Newborn screening strategy for cystic fibrosis: a field study in an area with high allelic heterogeneity. Acta Paediatr 86: 497–502.
Castellani C, Picci L, Scarpa M, et al (2005) Cystic fibrosis carriers have higher neonatal immunoreactive trypsinogen values than non-carriers. Am J Med Genet A 135(2): 142–144.
Corbetta C, Seia M, Bassotti A, et al (2002) Screening for cystic fibrosis in newborn infants: results of a pilot programme based on a two tier protocol (IRT/DNA/IRT) in the Italian population. J Med Screen 9: 60–63.
Comeau AM, Parad RB, Dorkin HL, et al (2004) Population-based newborn screening for genetic disorders when multiple mutation DNA testing is incorporated: a cystic fibrosis newborn screening model demonstrating increased sensitivity but more carrier detections. Pediatrics 113: 1573–1581.
Crossley JR, Elliott RB, Smith PA (1979) Dried-blood spot screening for cystic fibrosis in the newborn. Lancet 1(8114): 472–474.
Dhondt JL (2005) Implementation of informed consent for a cystic fibrosis newborn screening program in France: low refusal rates for optional testing. J Pediatr 147(Suppl): S106–S108.
Farrell PM (2004) Cystic fibrosis newborn screening: shifting the key question from “should we screen?” to “how should we screen?”. Pediatrics 113(6): 1811–1812.
Gregg RG, Wilfond BS, Farrell PM, et al (1993) Application of DNA analysis in a population-screening program for neonatal diagnosis of cystic fibrosis (CF): comparison of screening protocols. Am J Hum Genet 52(3): 616–626.
Gregg RG, Simantel A, Farrell PM, et al (1997) Newborn screening for cystic fibrosis in Wisconsin: comparison of biochemical and molecular methods. Pediatrics 99: 819–824.
Henry RL, Hettiarachchi LC, Colley P, et al (1996) Genotype of the cystic fibrosis population of the Hunter Region of New South Wales. J Paediatr Child Health 32: 416–418.
Lee DS, Rosenberg MA, Peterson A, et al (2003) Analysis of the costs of diagnosing cystic fibrosis with a newborn screening program. J Pediatr 142: 617–623.
Littlewood JM, Littlewood AE, McLaughlin S, Shapiro L, Connolly S (1995) Twenty years’ continuous neonatal screening in one hospital: progress of the 37 patients and their families. Pediatr Pulmonol 19(Suppl): 213.
McKay K (2007) Cystic fibrosis: benefits and clinical outcome. J Inherit Metab Dis 30 (in press).
Merelle ME, Scheffer H, De Jong D, et al (2006) Extended gene analysis can increase specificity of neonatal screening for cystic fibrosis. Acta Paediatr 95: 1424–1428.
Narzi L, Lucarelli M, Lelli A, et al (2002) Comparison of two different protocols of neonatal screening for cystic fibrosis. Clin Genet 62: 245–249.
Nazer HM (1992) Early diagnosis of cystic fibrosis in Jordanian children. J Trop Pediatr 38(3): 113–115.
O’Sullivan BP, Zwerdling RG, Dorkin HL, et al (2006) Early pulmonary manifestation of cystic fibrosis in children with the DeltaF508/R117H-7T genotype. Pediatrics 118: 1260–1265.
Parsons EP, Clarke AJ, Bradley DM (2003) Implications of carrier identification in newborn screening for cystic fibrosis. Arch Dis Child Fetal Neonatal Ed 88(6): F467–F471.
Pollitt RJ, Dalton A, Evans S, et al (1997) Neonatal screening for cystic fibrosis in the Trent region (UK): two-stage immunoreactive trypsin screening compared with a three-stage protocol with DNA analysis as an intermediate step. J Med Screen 4: 23–28.
Rock MJ, Mischler EH, Farrell PM, et al (1990) Newborn screening for cystic fibrosis is complicated by age-related decline in immunoreactive trypsinogen levels. Pediatrics 85: 1001–1007.
Rosenberg MA, Farrell PM (2005) Assessing the cost of cystic fibrosis diagnosis and treatment. J Pediatr 147(Suppl): S101–S105.
Soini E, Kojola H (1983) Time-resolved fluorometer for lanthanide chelates—a new generation of nonisotopic immunoassays. Clin Chem 29: 65–68.
Sarles J, Berthezene P, Le Louarn C, et al (2005) Combining immunoreactive trypsinogen and pancreatitis-associated protein assays, a method of newborn screening for cystic fibrosis that avoids DNA analysis. J Pediatr 147: 302–305.
Schwartz M, Brandt NJ, Skovby F (1993) Screening for carriers of cystic fibrosis. Results of a pilot study among pregnant women. Eur J Hum Genet 1(3): 239–244.
Scotet V, de Braekeleer M, Roussey M, et al (2000) Neonatal screening for cystic fibrosis in Brittany, France: assessment of 10 years’ experience and impact on prenatal diagnosis. Lancet 356(9232): 789–794.
Seltzer WK, Accurso F, Fall MZ, et al (1991) Screening for cystic fibrosis; feasibility of molecular genetic analysis of dried blood specimens. Biochem Med Metal Biol 1: 105–109.
Southern KW, Littlewood JM (2003) Newborn screening programmes for cystic fibrosis. Paediatr Respir Rev 4(4): 299–305.
Southern KW, Munck A, Pollitt R, et al (2007) A survey of newborn screening for cystic fibrosis in Europe. J Cyst Fibros 6(1): 57–65.
Spence WC, Paulus-Thomas J, Orenstein DM, et al (1993) Neonatal screening for cystic fibrosis: addition of molecular diagnostics to increase specificity. Biochem Med Metab Biol 49: 200–211.
Stephan U, Busch EW, Kollberg H, et al (1975) Cystic fibrosis detection by means of a test-strip. Pediatrics 55(1): 35–38.
Travert G (1988) Analysis of worldwide experience of neonatal screening for cystic fibrosis by measurement of blood immunoreactive trypsin. In: Travert G, ed. Mucoviscidose: depistage neonatal et prise en charge precoce. Caen: CHRU de Caen, 1–23.
van den Akker-van Marle ME, Dankert HM, Verkerk PH, et al (2006) Cost-effectiveness of 4 neonatal screening strategies for cystic fibrosis. Pediatrics 118: 896–905.
Wilcken B, Wiley V (2003) Newborn screening methods for cystic fibrosis. Paediatr Respir Rev 4(4): 272–277.
Wilcken B, Wiley V, Sherry G, et al (1995) Neonatal screening for cystic fibrosis: a comparison of two strategies for case detection in 1.2 million babies. J Pediatr 127: 965–970.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicating editor: Rodney Pollitt
Competing interests: None declared
Rights and permissions
About this article
Cite this article
Wilcken, B. Newborn screening for cystic fibrosis: Techniques and strategies. J Inherit Metab Dis 30, 537–543 (2007). https://doi.org/10.1007/s10545-007-0584-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-007-0584-0