Abstract
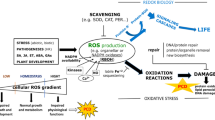
Although crosstalk between cytosolic and plastidic terpenoid pathways has been validated in many plant species, we report here for the first time a striking elevation of the nucleus-encoded artemisinin biosynthesis relevant DBR2 mRNA following the incubation of plants with fosmidomycin (FM). FM decreased singlet oxygen (1O2) scavengers such as β-carotene and α-tocopherol and subsequently invoked 1O2 burst. The treatment of plants with fluridone (FD) neither decreased α-tocopherol content nor triggered 1O2 emission. In conclusion, FM can up-regulate 1O2-sensitive nuclear genes responsible for artemisinin biogenesis by mitigating the accumulation of plastidic scavenging terpenoids, thereby eliciting 1O2 generation and initiating 1O2 retrograde signaling.
Similar content being viewed by others
Abbreviations
- ADS:
-
amorpha-4,11-dienesynthase
- CPR:
-
cytochrome P450 reductase
- CYP71AV1:
-
cytochrome P450 monooxygenase
- DBR2:
-
Δ11(13)-double-bond reductase 2
- DHAA:
-
dihydroartemisinic acid
- DXP:
-
1-deoxy-O-xylulose-5-phosphate
- DXR:
-
DXP reductoisomerase
- DXS:
-
DXP synthase
- FD:
-
fluridone
- FM:
-
fosmidomycin
- FPP:
-
farnesyl pyrophosphate
- MVA:
-
mevalonic acid
References
Bartels, P.G., Watson, C.W.: Inhibition of carotenoid synthesis by fluridone and norflurazon. — Weed Sci. 26: 198–203, 1978.
Benderliev, K.M., Ivanova, N.I., Pilarski, P.S.: Singlet oxygen and other reactive oxygen species are involved in regulation of release of iron-binding chelators from Scenedesmus cells. — Biol. Plant. 47: 523–526, 2003.
Bertea, C.M., Freije, J.R., Van der Woude, H., Verstappen, F.W., Perk, L., Marquez, V., De Kraker, J.W., Posthumus, M.A., Jansen, B.J., De Groot, A., Franssen, M.C., Bouwmeester, H.J.: Identification of intermediates and enzymes involved in the early steps of artemisinin biosynthesis in Artemisia annua. — Planta med. 71: 40–47, 2005.
Bertea, C.M., Voster, A., Verstappen, F.W., Maffei, M., Beekwilder, J., Bouwmeester, H.J.: Isoprenoid biosynthesis in Artemisia annua: cloning and heterologous expression of a germacrene A synthase from a glandular trichome cDNA library. — Arch. Biochem. Biophys. 448: 3–12, 2006.
Chappell, J.: The genetics and molecular genetics of terpene and sterol origami. — Curr. Opin. Plant Biol. 5: 151–157, 2002.
Covello, P. S.: Making artemisinin. — Phytochemistry 69: 2881–2885, 2008.
Covello, P.S., Teoh, K.H., Polichuk, D.R., Reed, D.W., Nowak, G.: Functional genomics and the biosynthesis of artemisinin. — Phytochemistry 68: 1864–1871, 2007.
Feng, L.L., Yang, R.Y., Yang, X.Q., Zeng, X.M., Lu, W.J., Zeng, Q.P.: Synergistic re-channeling of mevalonate pathway for artemisinin overproduction in transgenic Artemisia annua. — Plant Sci. 177: 57–67, 2009.
Guo, X.X., Yang, X.Q., Yang, R.Y., Zeng, Q.P.: Salicylic acid and methyl jasmonate but not Rose Bengal up-regulate artemisinin biosynthetic genes through invoking burst of endogenous singlet oxygen. — Plant Sci. 178: 390–397, 2010.
Hirayama, O., Nakamura, K., Hamada, S., Kobayashi, Y.: Singlet oxygen quenching ability of naturally occurring carotenoids. — Lipids 29: 149–150, 1994.
Kim, S.H., Chang, Y.J., Kim, S.U.: Tissue specificity and developmental pattern of amorpha-4,11-diene synthase (ADS) proved by ADS promoter-driven GUS expression in the heterologous plant, Arabidopsis thaliana. — Planta med. 74: 188–193, 2008.
Laloi, C., Przybyla, D., Apel, K.: A genetic approach towards elucidating the biological activity of different reactive oxygen species in Arabidopsis thaliana.— J. exp. Bot. 57: 1719–1724, 2006.
Laloi, C., Stachowiak, M., Pers-Kamczyc, E., Warzych, I.M., Apel, K.: Cross-talk between singlet oxygen- and hydrogen peroxide-dependent signalling of stress responses in Arabidopsis thaliana. — Proc. nat. Acad. Sci. USA 104: 672–677, 2007.
Laule, O., Furholz, A., Chang, H.S., Zhu, T., Wang, X., Heifetz, P.B., Gruissem, W., Lange, M.: Crosstalk between cytosolic and plastidial pathways of isoprenoid biosynthesis in Arabidopsis thaliana. — Proc. nat. Acad. Sci. USA 100: 6866–6871, 2003.
Lee, K.P., Kim, C., Landgraf, F., Apel, K.: EXECUTER1- and EXECUTER2-dependent transfer of stress-related signals from the plastid to the nucleus of Arabidopsis thaliana. —Proc. nat. Acad. Sci. USA 104: 10270–10275, 2007.
Maeda, H., Sakuragi, Y., Bryant, D.A., Della Penna, D.: Tocopherols protect Synechocystis sp. strain PCC 6803 from lipid peroxidation. — Plant Physiol. 138: 1422–1435, 2005.
Ro, D.K., Paradise, E.M., Ouelle, t M., Fisher, K.J., Newman, K.L., Ndungu, J.M., Ho, K.A., Eachus, R.A., Ham, T.S., Kirby, J., Chang, M.C., Withers, S.T., Shiba, Y., Sarpong, R., Keasling, J.D.: Production of the antimalarial drug precursor artemisinic acid in engineered yeast. — Nature 440: 940–943, 2006.
Rodriguez-Concepcion, M., Fores, O., Martinez-Garcia, J.F., Gonzalez, V., Phillips, M.A., Ferrer, A., Boronat, A.: Distinct light-mediated pathways regulate the biosynthesis and exchange of isoprenoid precursors during Arabidopsis seedling development. — Plant Cell 16: 144–156, 2004.
Sy, L.K., Brown, G.D.: The mechanism of the spontaneous autoxidation of dihydroartemisinic acid. — Tetrahedron 58: 897–908, 2002.
Teoh, K.H., Polichuk, D.R., Reed, D.W., Nowak, G., Covello, P. S.: Artemisia annua L. (Asteraceae) trichome-specific cDNAs reveal CYP71AV1, a cytochrome P450 with a key role in the biosynthesis of the antimalarial sesquiterpene lactone artemisinin. — FEBS Lett. 580: 1411–1416, 2006.
Towler, M.J., Weathers, P.J.: Evidence of artemisinin production from IPP stemming from both the mevalonate and the nonmevalonate pathways. — Plant Cell Rep. 26: 2129–2136, 2007.
Triantaphylides, C., Havaux, M.: Singlet oxygen in plants: production, detoxification and signaling. — Trends Plant Sci. 14: 219–228, 2009.
Wallaart, T.E., Van Uden, W., Lubberink, H.G.M., Woerdenbag, H.J., Pras, N., Quax, W.J.: Isolation and identification of dihydroartemisinic acid from Artemisia annua and its possible role in the biosynthesis of artemisinin. — J. natur. Prod. 62: 430–433, 1999.
Wu, S., Schalk, M., Clark, A., Miles, R.B., Coates, R., Chappell, J.: Redirection of cytosolic or plastidic isoprenoid precursors elevates terpene production in plants. — Nat. Biotechnol. 24: 1441–1447, 2006.
Xu, X., Hu, X., Neill, S.J., Fang, J., Cai, W.: Fungal elicitor induces singlet oxygen generation, ethylene release and saponin synthesis in cultured cells of Panax ginseng C.A. Meyer. — Plant Cell Physiol. 46: 947–954, 2005.
Yang, R.Y., Feng, L.L., Yang, X.Q., Yin, L.L., Xu, X.L., Zeng, Q.P.: Quantitative transcript profiling reveals downregulation of a steroid pathway relevant gene and overexpression of artemisinin biogenetic genes in transgenic Artemisia annua plants. — Planta med. 74: 1510–1516, 2008.
Yang, R.Y., Zeng, X.M., Lu, Y.Y., Lu, W.J., Feng, L.L., Yang, X.Q., Zeng, Q.P.: Senescent leaves of Artemisia annua are one of the most active organs for overexpression of artemisinin biosynthesis responsible genes upon burst of singlet oxygen. — Planta med. 76: 734–742, 2010.
Yin, L.L., Zhao, C., Huang, Y., Yang, R.Y.: Abiotic stressinduced expression of artemisinin biosynthesis genes in Artemisia annua L. — Chin. appl. environ. Biol. 14: 1–5, 2008.
Zeng, Q.P., Qiu, F., Yuan, L.: Production of artemisinin by genetically modified microbes. — Biotechnol. Lett. 30: 581–592, 2008a.
Zeng, Q.P., Yang, R.Y., Feng, L.L., Yang, X.Q.: Genetic and environmental regulation and artificial metabolic manipulation for artemisinin biosynthesis. — In: Richter, F.W. (ed.): Biotechnology: Research, Technology and Applications. Pp. 159–210. Nova Science Publishers, New York 2008b.
Zeng, Q.P., Zeng, X.M., Feng, L.L., Yin, L.L., Yang, X.Q., Yang, R.Y.: Quantification of three key enzymes involved in artemisinin biosynthesis in Artemisia annua by polyclonal antisera-based ELISA. — Plant mol. Biol. Rep. 27: 50–57, 2009.
Zeng, Q.P., Zhao, C., Yin, L.L., Yang, R.Y., Zeng, X.M., Huang, Y., Feng, L.L., Yang, X.Q.: Cloning of artemisinin biosynthetic cDNAs and novel ESTs and quantification of low temperature-induced gene overexpression. — Sci. China Ser. C 51: 232–244, 2008c.
Zhang, Y.S., Teoh, K.H., Reed, D.W., Maes, L., Goossens, A., Olson, D.J.H., Ross, A.R.S., Covello, P.S.: The molecular cloning of artemisinic aldehyde Δ11 (13) reductase and its role in glandular trichome-dependent biosynthesis of artemisinin in Artemisia annua. — J. biol. Chem. 283: 21501–21508, 2008.
Acknowledgements
We thank Li-Xiang Zeng, Xiao-Xia Guo and Ping-Zu Zhang for their helpful assistance. This work is financially supported by the National Natural Science Foundation of China (NSFC) (No. 30271591) and Guangdong Provincial Scientific Development Project of China (No. 2007B031404008).
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors contributing equally to this work.
Rights and permissions
About this article
Cite this article
Zeng, Q.P., Zeng, X.M., Yang, R.Y. et al. Singlet oxygen as a signaling transducer for modulating artemisinin biosynthetic genes in Artemisia annua . Biol Plant 55, 669–674 (2011). https://doi.org/10.1007/s10535-011-0166-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10535-011-0166-8