Abstract
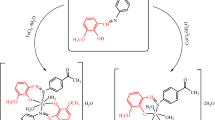
A series of Fe(II), Ni(II), and Pd(II) complexes were prepared with a novel Schiff base ligand containing pyridine moiety. The prepared compounds were characterized using FT-IR, 1H and 13 C NMR, UV–Vis, powder XRD, thermogravimetric analysis, mass spectra, magnetic susceptibility, and elemental analysis. The coordination geometry of Fe(II) and Ni(II) complexes were octahedral, where Fe(II) and Ni(II) metal ions were coordinated by an oxygen atom of the carbonyl group, a nitrogen atom of the azomethine moiety, and a phenolic oxygen atom. The Pd(II) complex had square planar geometry. All of the synthesized compounds were tested for their biochemical properties, including enzyme inhibition and antioxidant activities. According to the in vitro DPPH and FRAP antioxidant methods, the Schiff base ligand and its Fe(II)/Pd(II) complexes showed close antioxidant activities against the standards (BHA, BHT, ascorbic acid, and α-tocopherol). Enzyme inhibitions of the metal complexes were investigated against glutathione S-transferase (GST), acetylcholinesterase (AChE), and butyrylcholinesterase (BChE) enzymes. The best inhibition value (Ki) was observed for the Ni(II) complex against GST (2.63 ± 0.04 µM). Also, the Pd(II) complex showed the best inhibition value (10.17 ± 1.88 µM) against AChE. Molecular docking specified significant interactions at the active pockets of respective target enzymes. The Ni(II) complex exhibited good binding affinity against both BChE (− 9.0 kcal/mol and 9.36 ± 2.03 µM) and GST (− 7.0 kcal/mol and 2.63 ± 0.04 µM) enzymes.
Graphic abstract









Similar content being viewed by others
References
Abdel-Rahman LH, El-Khatib RM, Nassr LA, Abu-Dief AM (2017) DNA binding ability mode, spectroscopic studies, hydrophobicity, and in vitro antibacterial evaluation of some new Fe (II) complexes bearing ONO donors amino acid Schiff bases. Arab J Chem 10:S1835–S1846
Al-Amiery AA, Al-Majedy YK, Kadhum AAH, Mohamad AB (2016) Synthesis of new coumarins complemented by quantum chemical studies. Res Chem Intermed 42:3905–3918
Al-Noaimi M, Abu-Surrah AS, Tahtamouni L (2016) Palladium (II) complexes incorporating phenylazo arylmethine ancillary ligands: synthesis, spectral and antitumor activity. Arab J Chem 9:S1503–S1509
Arabahmadi R (2019) Cobalt (II) complexes derived from azo-azomethine ligands: synthesis, characterization, solvatochromic, fluorescence, thermal, electrochemical and antimicrobial properties. ChemSelect 4:4883–4891
Aras A, Bursal E, Alan Y, Turkan F, Alkan H, Kılıç Ö (2018) Polyphenolic content, antioxidant potential and antimicrobial activity of Satureja boissieri. Iranian J Chem Chem Eng 37:209–219
Aras A, Bursal E, Türkan F, Tohma H, Kılıç Ö, Gülçin İ, Köksal E (2019) Phytochemical content, antidiabetic, anticholinergic, and antioxidant activities of endemic Lecokia cretica extracts. Chem Biodivers 16:e1900341
Biovia DS (2017) Discovery studio visualizer, vol 936. San Diego, CA, USA
Bougherra H, Berradj O, Adkhis A, Amrouche T (2018) Synthesis, characterization, electrochemical and biological activities of mixed ligand copper (II) complexes with dimethylglyoxime and amino acids. J Mol Struct 1173:280–290
Buldurun K, Turan N, Aras A, Mantarcı A, Turkan F, Bursal E (2019) Spectroscopic and structural characterization, enzyme inhibitions, and antioxidant effects of new Ru (II) and Ni (II) complexes of Schiff base. Chem Biodivers 16:e1900243
Buldurun K, Turan N, Bursal E, Mantarcı A, Turkan F, Taslimi P, Gülçin İ (2020) Synthesis, spectroscopic properties, crystal structures, antioxidant activities and enzyme inhibition determination of Co (II) and Fe (II) complexes of Schiff base. Res Chem Intermed 46:283–297
Bursal E, Aras A, Kılıç Ö (2019) Evaluation of antioxidant capacity of endemic plant Marrubium astracanicum subsp. macrodon: identification of its phenolic contents by using HPLC-MS/MS. Nat Prod Res 33:1975–1979
Celen S, Gungor E, Kara H, Azaz AD (2013) Synthesis, spectroscopic characterization, and antimicrobial activities of Ni (II) and Fe (II) complexes with N-(2-hydroxyethyl)-5-nitrosalicylaldimine. J Coord Chem 66:3170–3181
Che C-M, Siu F-M (2010) Metal complexes in medicine with a focus on enzyme inhibition. Curr Opin Chem Biol 14:255–261
Cheng Z-Q, Zhu K-K, Zhang J, Song J-L, Muehlmann LA, Jiang C-S, Liu C-L, Zhang H (2019) Molecular-docking-guided design and synthesis of new IAA-tacrine hybrids as multifunctional AChE/BChE inhibitors. Bioorg Chem 83:277–288
Cutinho PF, Roy J, Anand A, Cheluvaraj R, Murahari M, Chimatapu HV (2020) Design of metronidazole derivatives and flavonoids as potential non-nucleoside reverse transcriptase inhibitors using combined ligand-and structure-based approaches. J Biomol Struct Dyn 38:1626–1648
Dobritzsch D, Grancharov K, Hermsen C, Krauss G-J, Schaumlöffel D (2020) Inhibitory effect of metals on animal and plant glutathione transferases. J Trace Elem Med Biol 57:48–56
El-Medani SM, Makhlouf AA, Moustafa H, Afifi MA, Haukka M, Ramadan RM (2020) Spectroscopic, crystal structural, theoretical and biological studies of phenylacetohydrazide Schiff base derivatives and their copper complexes. J Mol Struct 1208:127860
Geeta B, Shravankumar K, Reddy PM, Ravikrishna E, Sarangapani M, Reddy KK, Ravinder V (2010) Binuclear cobalt (II), nickel (II), copper (II) and palladium (II) complexes of a new Schiff-base as ligand: synthesis, structural characterization, and antibacterial activity. Spectrochim Acta Part A 77:911–915
Grivani G, Tahmasebi V, Eskandari K, Khalaji AD, Bruno G, Rudbari HA (2013) Synthesis, characterization, crystal structure determination and computational study of the two new bidentate O, N Schiff bases derived from bromosalicylaldehyde and amines containing alkyl halide pendant groups. J Mol Struct 1054:100–106
Gülçin İ, Gören AC, Taslimi P, Alwasel SH, Kılıc O, Bursal E (2020) Anticholinergic, antidiabetic and antioxidant activities of Anatolian pennyroyal (Mentha pulegium)-analysis of its polyphenol contents by LC-MS/MS. Biocatal Agric Biotechnol 23:101441
Hassan LR, Bahron H, Abdullah F, Tajuddin AM (2020) Cytotoxicity, alpha-glucosidase inhibition and molecular docking studies of hydroxamic acid chromium (III) complexes. J Biol Inorg Chem 25:239–252
Houghton DT, Gydesen NW, Arulsamy N, Mehn MP (2010) Synthesis and characterization of iron (II) quinaldate complexes. Inorg Chem 49:879–887
Keypour H, Ansari N, Mahmoudabadi M, Karamian R, Farida SHM, Moghadam ME, Gable RW (2020) Mn (III), Zn (II) and Pt (II) macroacyclic complexes: synthesis, X-ray structures, anticancer and antioxidant activities. Inorganica Chimica Acta. 509:119705
Kiss R, Sandor M, Szalai FA (2012) A public web service for drug discovery. J Cheminform 4:P17
Mermer A, Demirbaş N, Şirin Y, Uslu H, Özdemir Z, Demirbaş A (2018) Conventional and microwave prompted synthesis, antioxidant, anticholinesterase activity screening and molecular docking studies of new quinolone-triazole hybrids. Bioorg Chem 78:236–248
Nithya P, Rajamanikandan R, Simpson J, Ilanchelian M, Govindarajan S (2018) Solvent assisted synthesis, structural characterization and biological evaluation of cobalt (II) and nickel (II) complexes of Schiff bases generated from benzyl carbazate and cyclic ketones. Polyhedron 145:200–217
Pahonţu E, Proks M, Shova S, Lupaşcu G, Ilieş DC, Bărbuceanu ŞF, Socea LI, Badea M, Păunescu V, Istrati D (2019) Synthesis, characterization, molecular docking studies and in vitro screening of new metal complexeswith Schiff base as antimicrobial and antiproliferative agents. Appl Organomet Chem 33:e5185
Ramadan RM, Al-Nasr AKA, Noureldeen AF (2014) Synthesis, spectroscopic studies, antimicrobial activities and antitumor of a new monodentate V-shaped Schiff base and its transition metal complexes. Spectrochim Acta Part A 132:417–422
Saif M, El-Shafiy HF, Mashaly MM, Eid MF, Nabeel A, Fouad R (2016) Synthesis, characterization, and antioxidant/cytotoxic activity of new chromone Schiff base nano-complexes of Zn (II), Cu (II), Ni (II) and Co (II). J Mol Struct 1118:75–82
Schrödinger L (2017) The PyMOL Molecular graphics system, version 2.0. Schrödinger, LLC
Shukla SN, Gaur P, Raidas ML, Chaurasia B (2020) Tailored synthesis of unsymmetrical tetradentate ONNO schiff base complexes of Fe (IIl), Co (II) and Ni (II): Spectroscopic characterization, DFT optimization, oxygen-binding study, antibacterial and anticorrosion activity. J Mol Struct 1202:127362
Sumalatha V, Rambabu A, Vamsikrishna N, Ganji N, Daravath S (2019) Synthesis, characterization, DNA binding propensity, nuclease efficacy, antioxidant and antimicrobial activities of Cu (II), Co (II) and Ni (II) complexes derived from 4-(trifluoromethoxy) aniline Schiff bases. Chem Data Collect 20:100213
Taslimi P, Köksal E, Gören AC, Bursal E, Aras A, Kılıç Ö, Alwasel S, Gülçin İ (2020) Anti-Alzheimer, antidiabetic and antioxidant potential of Satureja cuneifolia and analysis of its phenolic contents by LC-MS/MS. Arab J Chem 13:4528–4537
Trott O, Olson A (2009) Software news and update AutoDock vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comput Chem 31(2):455–461
Turan N, Adıguzel R, Buldurun K, Bursal E (2016) Spectroscopic, thermal and antioxidant properties of novel mixed ligand-metal complexes obtained from saccharinate complexes and azo dye ligand (mnppa). Int J Pharmacol 12:92–100
Turhan K, Pektaş B, Türkan F, Tuğcu FT, Turgut Z, Taslimi P, Karaman HS, Gulcin I (2020) Novel benzo [b] xanthene derivatives: bismuth (III) triflate-catalyzed one-pot synthesis, characterization, and acetylcholinesterase, glutathione S-transferase, and butyrylcholinesterase inhibitory properties. Archiv der Pharmazie. 353(8):e2000030
Türkan F, Huyut Z, Huyut MT, Calimli MH (2019) In vivo biochemical evaluations of some β-lactam group antibiotics on glutathione reductase and glutathione S-transferase enzyme activities. Life Sci 231:116572
Türkan F, Atalar MN, Aras A, Gülçin İ, Bursal E (2020) ICP-MS and HPLC analyses, enzyme inhibition and antioxidant potential of Achillea schischkinii Sosn. Bioorg Chem 94:103333
Zengin G, Senkardes I, Mollica A, Picot-Allain CMN, Bulut G, Dogan A, Mahomoodally MF (2018) New insights into the in vitro biological effects, in silico docking and chemical profile of clary sage–Salvia sclarea L. Comput Biol Chem 75:111–119
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bursal, E., Turkan, F., Buldurun, K. et al. Transition metal complexes of a multidentate Schiff base ligand containing pyridine: synthesis, characterization, enzyme inhibitions, antioxidant properties, and molecular docking studies. Biometals 34, 393–406 (2021). https://doi.org/10.1007/s10534-021-00287-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-021-00287-z