Abstract
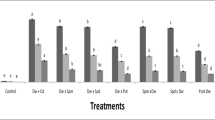
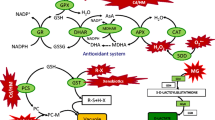
Polyamine metabolism, as well as spermine (Spm) antioxidant properties, were studied in wheat leaves under Cd2+ or Cu2+ stress. The oxidative damage produced by both metals was evidenced by an increased of thiobarbituric acid reactive substances (TBARS) and a significant decrease in glutathione under both metal treatments. Ascorbate peroxidase (APOX) and glutathione reductase (GR) activities were reduced by both metals to values ranging from 30% to 64% of the control values. Conversely, copper produced a raise in superoxide dismutase activity. The high putrescine (Put) content detected under Cd2+ stress (282% over the control) was induced by the increased activity of both enzymes involved in Put biosynthesis, arginine decarboxylase (ADC) and ornithine decarboxylase (ODC). However, only ODC activity was increased in wheat leaves subjected to Cu2+ stress, leading to a lower Put rise (89% over the controls). Spermidine (Spd) content was not affected by metal treatments, while Spm was significantly reduced. Pretreatment with Spm completely reverted the metals-induced TBARS increase whereas metals-dependent H2O2 deposition on leaf segments (revealed using diaminobenzidine), was considerably reduced in Spm pretreated leaf segments. This polyamine failed to reverse the depletion in APOX activity and glutathione (GSH) content produced by Cd2+ and Cu2+, although it showed an efficient antioxidant behavior in the restoration of GR activity to control values. These results suggest that Spm could be exerting a certain antioxidant function by protecting the tissues from the metals-induced oxidative damage, though this effect was not enough to completely avoid Cd2+ and Cu2+ effect on certain antioxidant enzymes, though the precise mechanism of protection still needs to be elucidated.
Similar content being viewed by others
Abbreviations
- ADC:
-
Arginine decarboxylase
- APOX:
-
Ascorbate peroxidase
- DAB:
-
Diaminobenzidine
- DAO:
-
Diamine oxidase
- GPOX:
-
Guaiacol peroxidase
- GR:
-
Glutathione reductase
- ODC:
-
Ornithine decarboxylase
- Pas:
-
Polyamines
- Put:
-
Putrescine
- SOD:
-
Superoxide dismutase
- Spd:
-
Spermidine
- Spm:
-
Spermine
- TBARS:
-
Thiobarbituric acid reactive substances.
References
Anderson ME (1985) Determination of glutathione and glutathione disulfide in biological samples. Methods Enzymol 113:548–554
Aziz A, Martín-Tanguy J, Larher F (1998) Stress-induced changes in polyamine and tyramine levels can regulate proline accumulation in tomato leaf discs treated with sodium chloride. Physiol Plant 104:195–202
Bagni N, Tassoni A (2001) Biosynthesis, oxidation and conjugation of aliphatic polyamines in higher plants. Amino Acids 20:301–317
Baycu G, Tolunay D, Özden H, Günebakan S (2006) Ecophysiological and seasonal variations in Cd, Pb, Zn, and Ni concentrations in the leaves of urban deciduous trees in Istanbul. Environ Pollut In Press
Becana M, Aparicio-Tejo P, Irigoyen JJ, Sánchez-Díaz M (1986)␣Some enzymes of hydrogen peroxide metabolism in leaves and root nodules of Medicago sativa. Plant Physiol 82:1169–1171
Benavides MP, Gallego SM, Comba ME, Tomaro ML (2000) Relationship between polyamines and paraquat toxicity in sunflower leaf discs. Plant Growth Regul 31:215–224
Benavides MP, Gallego SM, Tomaro ML (2005) Cadmium toxicity in plants. Braz J Plant Physiol 17:21–34
Besford RT, Richardson CM, Campos JL, Tiburcio AF (1993) Effect of polyamines on stabilization of molecular complexes of thylakoid membranes of osmotically stressed oat leaves. Planta 189:201–206
Bouchereau A, Aziz A, Larher F, Martin-Tanguy J (1999) Polyamines and environmental challenges: recent development. Plant Sci 140:103–125
Bruns I, Friese K, Market B, Kraus GJ (1997) The use of Fontinalis antipyretica L. as a bioindicator for heavy metals. Heavy metal accumulation and physiological reaction of Fontinalis antipyretica L. ex Hedw. in active biomonitoring in the River Elbe. Sci Tot Environ 204:161–176
Carley E, Wolosiuk RA, Hertig CM (1983) Regulation of the activation of chloroplast fructose-1.6-bis phosphatase (E.C 3.1.11). Inhibition by spermidine and spermine. Biochem Biophys Res Commun 115:707–710
Das S, Bose A, Ghosh B (1995) Effect of salt stress on polyamine metabolism in Brassica campestris. Phytochemistry 39:283–285
Dixit V, Pandey V, Shyam R (2001) Differential oxidative responses to cadmium in roots and leaves of pea (Pisum sativum L cv. Azad). J Exp Bot 52:1101–1109
Drolet G, Dumbroff EB, Legg R, Thompson JE (1986) Radical scavenging properties of polyamines. Phytochemistry 25:367–371
Flores HE, Galston AW (1984) Osmotic stress induced polyamine accumulation in cereal leaves. I. Physiological parameters of the response. Plant Physiol 75:102–109
Gallego SM, Benavides MP, Tomaro ML (1996) Effect of heavy metal ion excess on sunflower leaves: evidence for involvement of oxidative stress. Plant Sci 121:151–159
Gratão PL, Polle A, Lea PJ, Azevedo RA (2005) Making the life of heavy metal-stressed plants a little easier. Funct Plant Biol 32:481–494
Groppa MD, Tomaro ML, Benavides MP (2001) Polyamines as protectors against cadmium or copper-induced oxidative damage in sunflower leaf discs. Plant Sci 161:481–488
Ha HC, Sirisoma NS, Kuppusamy P, Zweier JL, Woster PM, Casero RA (1998) The natural polyamine spermine functions directly as a free radical scavenger. Proc Natl Acad Sci USA 95:11140–11145
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Hoagland DR, Arnon DI. 1950 The water culture method for growing plants without soil. Agric. Exp. Station, University of California, Berkeley, Circ. 347
Jacobsen S, Hauschild MZ, Rasmussen U (1992) Induction by chromium ion of chitinases and polyamines in barley (Hordeum vulgare L.) and rape (Brassica napus L ssp. oleifera). Plant Sci 84:119–128
Khan AU, Mei YH, Wilson T (1992) A proposed function for spermine and spermidine: protection of replicating DNA against damage by singlet oxygen. Proc Natl Acad Sci USA 89:11426–11427
Kramer GF, Norman HA, Krizek DT, Mirecki RM (1991) Influence of UV-B radiation on polyamines, lipid peroxidation and membrane lipids in cucumber. Phytochemistry 30:2101–2108
Kurepa J, Smalle J, Vanmontagu M, Inze D (1998) Polyamines and paraquat toxicity in Arabidopsis thaliana. Plant Cell Physiol 39:987–992
Lin CC, Kao CH (1999) Excess copper induces an accumulation of putrescine in rice leaves. Bot Bull Acad Sin 40:213–218
Lomozik L, Gasowska A, Bregier-Jarzebowska R, Jastrzab R (2005) Coordination chemistry of polyamines and their interactions in ternary systems including metal ions, nucleosides and nucleotides. Coord Chem Rev 249:2335–2350
Lovaas E (1997) Antioxidant and metal-chelating effects of polyamines. In: Sies H, ed. Advances in Pharmacology, Vol. 38: Antioxidants in Disease Mechanisms and Therapy. Academic Press: 119–149
Maehly AC, Chance B (1954) The assay of catalase and peroxidase. In: Glick D, ed. Methods of Biochemical Analysis, Vol. 1: 357
Martin-Tanguy J (2001) Metabolism and function of polyamines in plants: recent development (new approaches). Plant Growth Regul 34:135–148
Martin-Tanguy J, Aribaud M, Carré M, Gaspar T (1997) ODC-mediated biosynthesis and DAO-mediated catabolism of putrescine involved in rooting of Chrysanthemum explants in vitro. Plant Physiol Biochem 35:595–602
Nagele A, Felix K, Lengfelder E (1994) Induction of oxidative stress and protection against hydrogen-peroxide-mediated cytotoxicity by the superoxide dismutase-mimetic complex copper-putrescine-pyridine. Biochem Pharmacol 47:555–562
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach choroplast. Plant Cell Physiol 22:867–880
Nayyar H, Chander S (2004) Protective effects of polyamines against oxidative stress induced by water and cold stress in chickpea. J Agron Crop Sci 190:355–365
Ouariti O, Boussama N, Zarrouk M, Cherif A, Ghorbal MH (1997) Cadmium- and copper-induced changes in tomato membrane lipids. Phytochemistry 45:1343–1350
Poduslo JF, Curran GL (1996) Increased permeability of superoxide dismutase at the blood-nerve and blood-brain barriers with retained enzymatic activity after modification with naturally occurring polyamine, putrescine. J Neurochem 67:734–741
Quartacci MF, Cosi E, Navari-Izzo F (2001) Lipids and NADPH-dependent superoxide production in plasma membrane vesicles from roots of wheat grown under copper deficiency or excess. J Exp Bot 52:77–84
Sanità di Toppi L, Gabbrielli R (1999) Response to cadmium in higher plants. Environ Exp Bot 41:105–130
Schaedle M, Bassham JA (1977) Chloroplast glutathione reductase. Plant Physiol 59:1011–1012
Shen WY, Nada K, Tachibana S (2000) Involvement of polyamines in the chilling tolerance of cucumber cultivars. Plant Physiol 124:431–439
Smith BN, Meeuse BJD (1966) Production of volatiles amines in some Arum lily species. Plant Physiol 41:343–347
Thordal-Christensen H, Zhang Z, Wei Y, Collinge DB (1997) Subcellular localization of H2O2 in plants. H2O2 accumulation in papillae and hypersensitive response during the barley-powdery mildew interaction. Plant J 11:1187–1194
Tiburcio AF, Altabella T, Borrel A, Masgrau C (1997) Polyamines metabolism and its regulation. Physiol Plant 100:664–674
Van Assche F, Clijsters H (1990) Effects of metals on enzyme activity in plants. Plant Cell Environ 13:195–206
Vitória AP, Lea PJ, Azevedo RA (2001) Antioxidant enzymes responses to cadmium in radish tissues. Phytochemistry 57:701–710
Walters D (2003) Resistance to plant pathogens: possible roles for free polyamines and polyamine catabolism. New Phytol 159:109–115
Weinstein LH, Kaur-Sawhney R, Venkat Rajam M, Wettlaufer SH, Galston AW. 1986 Cadmium-induced accumulation of putrescine in oat and bean leaves. Plant Physiol 82, 641–645
Acknowledgements
This work was supported by the University of Buenos Aires (Grants B01 and B044). Benavides MP and Tomaro ML are researchers of the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Groppa, M.D., Tomaro, M.L. & Benavides, M.P. Polyamines and heavy metal stress: the antioxidant behavior of spermine in cadmium- and copper-treated wheat leaves. Biometals 20, 185–195 (2007). https://doi.org/10.1007/s10534-006-9026-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-006-9026-y