Abstract
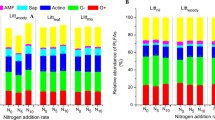
Aboveground litter not only is an important source of nutrients to soil microbes but also regulates the microclimate in topsoil. How the changes in aboveground litter quantity would affect the microbial biogeochemical cycles is still unclear. Here we conducted a litter input manipulation experiment in a temperate mixed forest to investigate how different amounts of litter input affect soil organic carbon (SOC) and soil respiration via their regulation on soil microbes. We found that although neither SOC stock nor soil CO2 efflux was affected by litter manipulation, soil microbial characteristics had responded after four years of litter addition or removal treatments. Microbial biomass carbon (MBC) in the O horizon was higher in litter addition plots than in litter removal plots as a result of the changed availability of labile C under litter treatments. Both double litter and no litter treatments changed microbial compositions, which was probably due to the increased soil pH in no litter treatment and the increased labile C in double litter treatment. The null change in soil respiration could be attributed to the offset between the negative effect of decreased substrate and the positive effect of increased temperature on soil respiration in litter removal plots. Due to the important role of soil microbes in carbon cycling, the altered microbial properties under litter manipulation treatments suggested the inevitable changes in biogeochemical cycling in the long run and call for long-term studies on SOC dynamics in the future.




Similar content being viewed by others
Data availability
Data and materials are available upon request from the corresponding author.
References
Bárcenas-Moreno G, Gómez-Brandón M, Rousk J, Bååth E (2009) Adaptation of soil microbial communities to temperature: comparison of fungi and bacteria in a laboratory experiment. Glob Change Biol 15:2950–2957. https://doi.org/10.1111/j.1365-2486.2009.01882.x
Beck T, Joergensen RG, Kandeler E, Makeschin F, Nuss E, Oberholzer HR, Scheu S (1997) An inter-laboratory comparison of ten different ways of measuring soil microbial biomass C. Soil Biol Biochem 29:1023–1032. https://doi.org/10.1016/s0038-0717(97)00030-8
Bond-Lamberty B, Bailey VL, Chen M, Gough CM, Vargas R (2018) Globally rising soil heterotrophic respiration over recent decades. Nature 560:80–83. https://doi.org/10.1038/s41586-018-0358-x
Bowden RD, Deem L, Plante AF, Peltre C, Nadelhoffer K, Lajtha K (2014) Litter input controls on soil carbon in a temperate deciduous forest. Soil Sci Soc Am J 78:S66–S75. https://doi.org/10.2136/sssaj2013.09.0413nafsc
Bradford MA et al (2008) Thermal adaptation of soil microbial respiration to elevated temperature. Ecol Lett 11:1316–1327. https://doi.org/10.1111/j.1461-0248.2008.01251.x
Bréchet LM, Lopez-Sangil L, George C, Birkett AJ, Baxendale C, Castro Trujillo B, Sayer EJ (2018) Distinct responses of soil respiration to experimental litter manipulation in temperate woodland and tropical forest. Ecol Evol 8:3787–3796. https://doi.org/10.1002/ece3.3945
Brookes PC, Landman A, Pruden G, Jenkinson DS (1985) Chloroform fumigation and the release of soil nitrogen—a rapid direct extraction method to measure microbial biomass nitrogen in soil. Soil Biol Biochem 17:837–842. https://doi.org/10.1016/0038-0717(85)90144-0
Crow SE, Lajtha K, Bowden RD, Yano Y, Brant JB, Caldwell BA, Sulzman EW (2009) Increased coniferous needle inputs accelerate decomposition of soil carbon in an old-growth forest. For Ecol Manage 258:2224–2232. https://doi.org/10.1016/j.foreco.2009.01.014
DeAngelis KM, Chowdhury PR, Pold G, Romero-Olivares A, Frey S (2019) Microbial responses to experimental soil warming: five testable hypotheses. In: Mohan J (ed) Ecosystem consequences of soil warming. Academic Press, San Diego
Evans LR, Pierson D, Lajtha K (2020) Dissolved organic carbon production and flux under long-term litter manipulations in a Pacific Northwest old-growth forest. Biogeochemistry 149:75–86. https://doi.org/10.1007/s10533-020-00667-6
Fekete I et al (2014) Alterations in forest detritus inputs influence soil carbon concentration and soil respiration in a Central-European deciduous forest. Soil Biol Biochem 74:106–114. https://doi.org/10.1016/j.soilbio.2014.03.006
Fekete I et al (2016) The effects of litter production and litter depth on soil microclimate in a central european deciduous forest. Plant Soil 398:291–300. https://doi.org/10.1007/s11104-015-2664-5
Finzi AC, Allen AS, DeLucia EH, Ellsworth DS, Schlesinger WH (2001) Forest litter production, chemistry, and decomposition following two years of free-air CO2 enrichment. Ecology 82(2):470–484. https://doi.org/10.1890/0012-9658(2001)082[0470:flpcad]2.0.co;2
Frostegård Å, Tunlid A, Bååth E (2011) Use and misuse of PLFA measurements in soils. Soil Biol Biochem 43:1621–1625. https://doi.org/10.1016/j.soilbio.2010.11.021
German DP, Weintraub MN, Grandy AS, Lauber CL, Rinkes ZL, Allison SD (2011) Optimization of hydrolytic and oxidative enzyme methods for ecosystem studies. Soil Biol Biochem 43:1387–1397. https://doi.org/10.1016/j.soilbio.2011.03.017
Geyer KM, Dijkstra P, Sinsabaugh R, Frey SD (2019) Clarifying the interpretation of carbon use efficiency in soil through methods comparison. Soil Biol Biochem 128:79–88. https://doi.org/10.1016/j.soilbio.2018.09.036
Hansen MC, Stehman SV, Potapov PV (2010) Quantification of global gross forest cover loss. Proc Natl Acad Sci 107:8650–8655. https://doi.org/10.1073/pnas.0912668107
Huang WJ, Spohn M (2015) Effects of long-term litter manipulation on soil carbon, nitrogen, and phosphorus in a temperate deciduous forest. Soil Biol Biochem 83:12–18. https://doi.org/10.1016/j.soilbio.2015.01.011
WRB IWG (2015) World Reference Base for Soil Resources 2014, Update 2015. International Soil Classification System for Naming Soils and Creating Legends for Soil Maps. World Soil Resources Reports No. 106. FAO, Rome
Jones DL, Willett VB (2006) Experimental evaluation of methods to quantify dissolved organic nitrogen (DON) and dissolved organic carbon (DOC) in soil. Soil Biol Biochem 38:991–999. https://doi.org/10.1016/j.soilbio.2005.08.012
Kaiser C, Frank A, Wild B, Koranda M, Richter A (2010a) Negligible contribution from roots to soil-borne phospholipid fatty acid fungal biomarkers 18:2ω6,9 and 18:1ω9. Soil Biol Biochem 42:1650–1652. https://doi.org/10.1016/j.soilbio.2010.05.019
Kaiser C et al (2010b) Belowground carbon allocation by trees drives seasonal patterns of extracellular enzyme activities by altering microbial community composition in a beech forest soil. New Phytol 187:843–858. https://doi.org/10.1111/j.1469-8137.2010.03321.x
Kalbitz K, Meyer A, Yang R, Gerstberger P (2007) Response of dissolved organic matter in the forest floor to long-term manipulation of litter and throughfall inputs. Biogeochemistry 86:301–318. https://doi.org/10.1007/s10533-007-9161-8
Klotzbucher T, Kaiser K, Stepper C, van Loon E, Gerstberger P, Kalbitz K (2012) Long-term litter input manipulation effects on production and properties of dissolved organic matter in the forest floor of a Norway spruce stand. Plant Soil 355:407–416. https://doi.org/10.1007/s11104-011-1123-1
Lajtha K, Crow SE, Yano Y, Kaushal SS, Sulzman E, Sollins P, Spears JDH (2005) Detrital controls on soil solution N and dissolved organic matter in soils: a field experiment. Biogeochemistry 76:261–281. https://doi.org/10.1007/s10533-005-5071-9
Lajtha K, Bowden RD, Nadelhoffer K (2014) Litter and root manipulations provide insights into soil organic matter dynamics and stability. Soil Sci Soc Am J 78:S261–S269. https://doi.org/10.2136/sssaj2013.08.0370nafsc
Lajtha K et al (2018) The detrital input and removal treatment (DIRT) network: insights into soil carbon stabilization. Sci Total Environ 640–641:1112–1120. https://doi.org/10.1016/j.scitotenv.2018.05.388
Liang C, Amelung W, Lehmann J, Kästner M (2019) Quantitative assessment of microbial necromass contribution to soil organic matter. Glob Change Biol 25:3578–3590. https://doi.org/10.1111/gcb.14781
Lichter J, Barron SH, Bevacqua CE, Finzi AC, Irving KE, Stemmler EA, Schlesinger WH (2005) Soil carbon sequestration and turnover in a pine forest after six years of atmospheric CO2 enrichment. Ecology 86:1835–1847. https://doi.org/10.1890/04-1205
Liu J et al (2017) Different fates of deposited NH4+ and NO3− in a temperate forest in northeast China: a N-15 tracer study. Glob Change Biol 23:2441–2449. https://doi.org/10.1111/gcb.13533
Manzoni S, Taylor P, Richter A, Porporato A, Ågren GI (2012) Environmental and stoichiometric controls on microbial carbon-use efficiency in soils. New Phytol 196:79–91. https://doi.org/10.1111/j.1469-8137.2012.04225.x
Manzoni S et al (2018) Reviews and syntheses: carbon use efficiency from organisms to ecosystems—definitions, theories, and empirical evidence. Biogeosciences 15:5929–5949. https://doi.org/10.5194/bg-15-5929-2018
Melillo JM et al (2017) Long-term pattern and magnitude of soil carbon feedback to the climate system in a warming world. Science 358:101–105
Meng C, Tian DS, Zeng H, Li ZL, Chen HYH, Niu SL (2020) Global meta-analysis on the responses of soil extracellular enzyme activities to warming. Sci Total Environ 705:9. https://doi.org/10.1016/j.scitotenv.2019.135992
Nadelhoffer KJ et al (2004) The DIRT experiment: litter and root influences on forest soil organic matter stocks and function. In: Foster D, Aber J (eds) Forests in time: the environmental consequences of 1000 years of change in New England. Yale University Press, New Haven, pp 300–315
Oliver EE et al (2021) Controls on soil microbial carbon use efficiency over long-term ecosystem development. Biogeochemistry 152(2):309–325
Pan YD et al (2011) A large and persistent carbon sink in the world’s forests. Science 333:988–993. https://doi.org/10.1126/science.1201609
Park JH, Matzner E (2003) Controls on the release of dissolved organic carbon and nitrogen from a deciduous forest floor investigated by manipulations of aboveground litter inputs and water flux. Biogeochemistry 66:265–286. https://doi.org/10.1023/B:BIOG.0000005341.19412.7b
Rodtassana C, Tanner EVJ (2018) Litter removal in a tropical rain forest reduces fine root biomass and production but litter addition has few effects. Ecology 99:735–742. https://doi.org/10.1002/ecy.2143
Roels JA (1980) Application of macroscopic principles to microbial metabolism. Biotechnol Bioeng 22(12):2457–2514
Rousk J et al (2010) Soil bacterial and fungal communities across a pH gradient in an arable soil. ISME J 4:1340–1351. https://doi.org/10.1038/ismej.2010.58
Saiya-Cork KR, Sinsabaugh RL, Zak DR (2002) The effects of long term nitrogen deposition on extracellular enzyme activity in an Acer saccharum forest soil. Soil Biol Biochem 34:1309–1315. https://doi.org/10.1016/S0038-0717(02)00074-3
Sayer EJ (2006) Using experimental manipulation to assess the roles of leaf litter in the functioning of forest ecosystems. Biol Rev 81:1–31. https://doi.org/10.1017/s1464793105006846
Sayer EJ, Powers JS, Tanner EVJ (2007) Increased litterfall in tropical forests boosts the transfer of soil CO2 to the atmosphere. PLoS ONE 2:e1299
Silva-Sánchez A, Soares M, Rousk J (2019) Testing the dependence of microbial growth and carbon use efficiency on nitrogen availability, pH, and organic matter quality. Soil Biol Biochem 134:25–35. https://doi.org/10.1016/j.soilbio.2019.03.008
Sinsabaugh RL et al (2008) Stoichiometry of soil enzyme activity at global scale. Ecol Lett 11:1252–1264. https://doi.org/10.1111/j.1461-0248.2008.01245.x
Sinsabaugh RL et al (2016) Stoichiometry of microbial carbon use efficiency in soils. Ecol Monogr 86:172–189. https://doi.org/10.1890/15-2110.1
Soares M, Rousk J (2019) Microbial growth and carbon use efficiency in soil: links to fungal-bacterial dominance, SOC-quality and stoichiometry. Soil Biol Biochem 131:195–205. https://doi.org/10.1016/j.soilbio.2019.01.010
Soong JL, Fuchslueger L, Marañon-Jimenez S, Torn MS, Janssens IA, Penuelas J, Richter A (2020) Microbial carbon limitation: the need for integrating microorganisms into our understanding of ecosystem carbon cycling. Glob Change Biol 26:1953–1961. https://doi.org/10.1111/gcb.14962
Sulzman EW, Brant JB, Bowden RD, Lajtha K (2005) Contribution of aboveground litter, belowground litter, and rhizosphere respiration to total soil CO2 efflux in an old growth coniferous forest. Biogeochemistry 73:231–256. https://doi.org/10.1007/s10533-004-7314-6
Wang D, Heckathorn SA, Wang XZ, Philpott SM (2012) A meta-analysis of plant physiological and growth responses to temperature and elevated CO2. Oecologia 169:1–13. https://doi.org/10.1007/s00442-011-2172-0
Wang QK, He TX, Wang SL, Liu L (2013) Carbon input manipulation affects soil respiration and microbial community composition in a subtropical coniferous forest. Agric For Meteorol 178:152–160. https://doi.org/10.1016/j.agrformet.2013.04.021
Wang Q, Wang S, He T, Liu L, Wu J (2014) Response of organic carbon mineralization and microbial community to leaf litter and nutrient additions in subtropical forest soils. Soil Biol Biochem 71:13–20. https://doi.org/10.1016/j.soilbio.2014.01.004
Wang C et al (2021) Large-scale importance of microbial carbon use efficiency and necromass to soil organic carbon. Glob Change Biol. https://doi.org/10.1111/gcb.15550
Weintraub SR, Wieder WR, Cleveland CC, Townsend AR (2013) Organic matter inputs shift soil enzyme activity and allocation patterns in a wet tropical forest. Biogeochemistry 114:313–326. https://doi.org/10.1007/s10533-012-9812-2
Xu X, Thornton PE, Post WM (2013) A global analysis of soil microbial biomass carbon, nitrogen and phosphorus in terrestrial ecosystems. Glob Ecol Biogeogr 22:737–749. https://doi.org/10.1111/geb.12029
Yano Y, Lajtha K, Sollins P, Caldwell BA (2005) Chemistry and dynamics of dissolved organic matter in a temperate coniferous forest on Andic soils: effects of litter quality. Ecosystems 8:286–300. https://doi.org/10.1007/s10021-005-0022-9
Acknowledgements
We would like to thank the staff in the National Research Station of Changbai Mountain Forest Ecosystems for their assistance in sample collection and storage. This study was supported by the National Natural Science Foundation of China (41971058, 41401289), and the Key Research Program of Frontier Sciences, CAS (QYZDB-SSW-DQC006).
Funding
This work was supported by the National Natural Science Foundation of China (41971058, 41401289), and the Key Research Program of Frontier Sciences, CAS (QYZDB-SSW-DQC006).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Sujay Kaushal.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This paper is an invited contribution to the 35th Anniversary Special Issue, edited by Sujay Kaushal, Robert Howarth, and Kate Lajtha.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dai, W., Peng, B., Liu, J. et al. Four years of litter input manipulation changes soil microbial characteristics in a temperate mixed forest. Biogeochemistry 154, 371–383 (2021). https://doi.org/10.1007/s10533-021-00792-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-021-00792-w