Abstract
Warming may increase the extent and intensity of insect defoliations within Arctic ecosystems. A thorough understanding of the implications of this for litter decomposition is essential to make predictions of soil-atmosphere carbon (C) feedbacks. Soil nitrogen (N) and C cycles naturally are interlinked, but we lack a detailed understanding of how insect herbivores impact these cycles. In a laboratory microcosm study, we investigated the growth responses of heterotrophic soil fungi and bacteria as well as C and N mineralisation to simulated defoliator outbreaks (frass addition), long-term increased insect herbivory (litter addition at higher background N-level) and non-outbreak conditions (litter addition only) in soils from a Subarctic birch forest. Larger amounts of the added organic matter were mineralised in the outbreak simulations compared to a normal year; yet, the fungal and bacterial growth rates and biomass were not significantly different. In the simulation of long-term increased herbivory, less litter C was respired per unit mineralised N (C:N of mineralisation decreased to 20 ± 1 from 38 ± 3 for pure litter), which suggests a directed microbial mining for N-rich substrates. This was accompanied by higher fungal dominance relative to bacteria and lower total microbial biomass. In conclusion, while a higher fraction of foliar C will be respired by insects and microbes during outbreak years, predicted long-term increases in herbivory linked to climate change may facilitate soil C-accumulation, as less foliar C is respired per unit mineralised N. Further work elucidating animal-plant-soil interactions is needed to improve model predictions of C-sink capacity in high latitude forest ecosystems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
High latitude ecosystems contain substantial amounts of terrestrial soil C (Tarnocai et al. 2009), but great uncertainties still remain about how the soil processes governing this C-storage will respond to environmental changes (Conant et al. 2011; Sistla et al. 2013; Crowther et al. 2016). Much of the Arctic is dominated by low productivity ecosystems, but Scandinavian mountain birch forests constitute a relatively productive ecosystem storing substantial amounts of C (Sjögersten and Wookey 2009). Yet, despite their potentially important role in high-latitude C-storage, relatively little research has focused on the biogeochemistry of these systems compared to other Arctic ecosystem types.
Outbreaks by Geometrid moths (primarily Eppirita autumnata and Operophtera brumata), are the largest natural disturbance in the Nordic mountain birch forests (Tenow et al. 2005, 2013; Bjerke et al. 2014; Olsson et al. 2017). During these outbreaks, insect deposits (including excreta/frass, cadavers, moults, pupae, greenfall) constitute considerable nutrient fluxes from the canopy to the soil, with the largest single constituent being frass (Hunter 2001; Arnold et al. 2016). Understanding how these outbreaks influence element cycling and plant growth is essential for improving predictions of C and N cycling (Throop et al. 2004; Metcalfe et al. 2016) and plant community composition (Jepsen et al. 2013) across the Scandinavian Arctic, particularly as the extent and intensity of outbreaks appear to be increasing due to climate warming (Jepsen et al. 2008; Kozlov and Zvereva 2017).
Some recent field studies have examined belowground effects of Geometrid outbreaks on Arctic birch forests with mixed results. For example, studies have reported enhanced resource turnover and decreased fungal:bacterial ratios (Kaukonen et al. 2013), slowed biogeochemical cycling linked to changes in ectomycorrhizal (ECM) associations (Parker et al. 2016), and decreased ECM-abundances and richness benefitting saprotrophic decomposers (Saravesi et al. 2015). Moreover, the substantial short-term N-enrichment following outbreaks in the Subarctic mountain birch system (e.g. Parker et al. 2016) may also increase the likelihood of N-inhibition of microbial activity (Fog 1988; Berg 2000; DeForest et al. 2004). Furthermore, labile organic matter addition may over time shift the organic matter use by decomposers to components richer in N; hence decreasing overall soil organic matter mineralisation (Ehtesham and Bengtson 2017; Rousk et al. 2016). Thus, while it is clear that the outbreaks exert a range of important impacts on vegetation and soil, a detailed picture is still missing of the underlying mechanisms regulating observed changes in soil processes. This is partly due to the difficulty of disentangling the effects of the increased surface input of herbivory transformed labile litter, and the decreased belowground labile C-allocation, due to reduced photosynthesis, which occur simultaneously (Saravesi et al. 2015).
In this study, we investigated how insect herbivore transformation of litter affects its use by the soil microbial community, and its subsequent impact on soil biogeochemistry and bioavailability, in a Subarctic birch forest soil. In a laboratory experiment, we added combinations of frass, senesced litter and inorganic N (Ni) to soil microcosms to simulate a spectrum of insect herbivory scenarios, ranging from no herbivory (senesced litter addition), short-term effects of a moderate (frass and senesced litter addition) and a full outbreak (frass addition). A long-term effect of increased herbivory may be increased inorganic N-availability in the soil (Belovsky and Slade 2000), so we added a treatment simulating a non-outbreak year under such conditions (senesced litter and inorganic N addition). A pure inorganic N treatment (Ni addition) was also included to account for potential N-inhibition effects. We measured growth rates and biomass concentrations of both fungi and bacteria to assess their respective contributions to decomposition, gross C and N mineralisation rates and changes in the microbial community structure. Recent work in Subarctic tundra and boreal forest soils have revealed a decoupling between C and N mineralisation which is emphasized by labile C-input (Rousk et al. 2016; Ehtesham and Bengtson 2017). A stronger decoupling between C- and N-mineralisation has been linked to higher fungal dominance (Rousk et al. 2016) and to higher N-fertilization (Ehtesham and Bengtson 2017). Thus, we hypothesized that (i) insect outbreaks would increase short-term C and N-mineralisation of aboveground litter, due to higher lability of frass compared to senesced litter, while (ii) long-term effects of increased herbivory (higher Ni availability) would result in decreased C and increased N-cycling, due to a shift in substrate use towards organic matter components richer in N. Moreover, in the short-term (iii) addition of frass would favour bacteria over fungi and increase total microbial biomass, while (iv) the consequence of higher background N-availability may be increase dominance of fungal contribution to decomposition of litter and decreased total microbial biomass. Our experimental microcosm design intentionally targeted the direct effect that the herbivore transformation of incoming litter would have on the decomposer microbial community. Consequently, indirect effects likely to add to the responses of a herbivore outbreak in field conditions, including for example changed foliar chemistry, plant community production and composition, changes in soil moisture content, or physical disturbances, have been removed.
Materials and methods
Study site
The study was conducted in a Subarctic mountain birch forest in the Torneträsk area of Swedish Lapland (68.243°N, 19.507°E). Three sites were laid out along an elevation gradient (150 m. elevation difference) in order to represent the mountainous landscape as well as possible; one close to the valley bottom (360 m above sea-level (a.s.l.), “lower site”), one close to the treeline (510 m.a.s.l., “higher site”) and one in between (430 m.a.s.l., “middle site”), with approximately 750 m horizontal distance between sites (Table 1). Each site was comprised of a c. 20 × 20 m area from which samples were taken. The relative vegetation distribution was estimated as the average coverage within 3 randomly picked squares of 3 × 3 m. Dwarf shrubs were determined to the species level, while mosses, lichens, graminoids and forbs were only recorded as groups. Soil temperature was measured with iButtons (Maxim Integrated, San Jose, CA, USA), and resin capsules (UNIBEST International, Walla Walla, WA, USA) were installed in the topsoil (5–10 cm) to estimate inorganic nutrient content. According to the Swedish Meteorological and Hydrological Institute records for the site (2000–2014), the mean annual air temperature was − 1.1 ± 0.7 °C (mean ± SE) and the mean annual precipitation was 644 ± 88 mm year−1, which is intermediate for the region in general (~ 300–1000 mm year−1). The bedrock geology was dominated by granites and other acid rocks and the subsoil texture was strongly dominated by coarse sand (> 90%) and showed weak to strong podzolisation at all sites.
Sampling
One sample of approximately 3 kg (fresh weight) of the organic horizon (top ~ 5–10 cm) were composited from minimum 3 randomly selected subsamples to represent each site in early September 2015. Only the organic soil horizon was sampled as this is the soil horizon primarily affected by aboveground litter input including insect deposits. Composite sampling was chosen to focus on treatment effects rather than variability within sites. The soil was cut into turfs of approximately 20 × 20 cm with a spade, and the vegetation layer, coarse roots as well as mineral soil were separated from the organic soil material and discarded, as the free living saprotrophs in the bulk soil was the focus of this experiment. Soils were stored at − 20 °C until the beginning of the experiment (~ 50 days) according to standard procedures (ISO/TC IS 1038 1-62009), sieved through a 6 mm mesh prior to analyses, moisture adjustment and establishment of the microcosms, followed by an equilibration period of 8 days at the temperature used in the microcosm experiments (Jenkinson 1988). The homogenisation of the bulk soil was done in order to minimise variation not due to the difference in chemical characteristics of the added substrates.
Approximately 0.5 kg (dry mass, 40 °C, 48 h) of senescent leaves and frass was collected to be used as substrate. Leaves were collected from multiple sites in the same area, including the studied sites, while insect frass was collected in a similar habitat to the study area near Tromsø, Norway, where there was an ongoing Geometrid moth outbreak. Outbreak densities were needed in order to collect feasible amounts of frass for addition experiments, and given the similarity of the Nordic mountain birch forest at the site of frass collection and the study site in terms of plant community, B. pubescens leaf chemistry and moth species, the frass was likely similar. Roughly 200 mesh bags (0.47 × 0.77 mm, Howi insect netting type L; Howitec, Bolsward, NL) were wrapped around attacked branches in late May to let the eggs hatch in the bags. Bags were collected in mid-September and after drying it was sieved through a 1 mm sieve, in order to exclude as much of the non-frass material as possible (green leaves, twigs etc.). Some of the most labile fractions of the frass may have been leached with percolating precipitation, which means that our results from the frass treatments should probably be seen as conservative estimates. Leaves were milled to a grain size similar to the frass (~ 200–500 µm) before addition, thus making the additions more homogenized and standardised than forms occurring in natural field conditions.
Experimental design
The following substrates were added to microcosms of 100 g of moist soil: (a) control (no substrates added), (b) litter, (c) frass + litter, (d) frass, (e) litter + Ni, f) Ni. All substrates were added in equal amounts of N (C:N ratios: litter = 42:1, frass + litter = 29:1, frass = 22:1, litter + Ni = 16:1), corresponding to 0.6 mg N g fresh soil−1 (~ 1 mg g SOM−1), and treatments were replicated 3 times per site, (3 sites × 6 treatments × 3 replicates = 54 microcosms). As Ni was added in solution (3 ml of 2.0% N (weight) NH4NO3-solution), all microcosms also received 3 ml of liquid (N-solution or distilled water). Organic N was only added in the form of frass or litter. Subsamples for laboratory measurements were extracted from microcosms at eight time points with an approximately logarithmic distribution over 64 days, with gentle homogenization before and after sampling to ensure aeration and representative sampling. On average, about 50% (max. 65% in few instances) of the soil was removed over the course of the experiment due to subsampling. Microcosms were weighed before and after each time point to determine soil moisture loss. When necessary, soil moisture levels were adjusted to maintain a water content of ~ 60% (gravimetric, fresh weight) to ensure stable and non-limiting moisture levels through the duration of the experiment. This was only necessary for a few cosms after timepoint 6. Between time points, the microcosms were incubated at 17 °C, which corresponds to a warm summer temperature in the study area.
Laboratory analyses
Gravimetric soil moisture fraction of fresh weight (105 °C for 24 h) and soil organic matter content were determined (loss on ignition, 600 °C over night) for soil from all three sites. Total C and N-concentrations of soils and substrates were measured in solid samples (substrates = 2 mg, soil = 15 mg) by Dumas combustion (1020 °C) on an elemental analyser (CE 1110, Thermo Electron, Milan, Italy) after thorough homogenization in a ball-mill. Adsorbed chemicals on the resin capsules were analysed by the capsule provider (UNIBEST International, Walla Walla, WA, USA). Briefly, ammonium and nitrate concentrations were analysed on a flow injection analyser (FIAlab-2500, FIAlab Instruments, Inc., Seattle, WA, USA), while all other elements were analyse using inductively coupled plasma optical emission spectroscopy (ICP-OES, Perkin-Elmer, Waltham, MA, USA) after extraction with 50 ml 2 M HCl. On soil from the microcosms, respiration was measured with gas chromatography as the CO2 developed in the headspace of 20 ml glass vials after incubating subsamples (0.5 g) for 24 h (YL6500 GC; YL Instruments, Gyeonggi-do, Korea). Bacterial growth rates were estimated on 0.5 g of soil by 3H-leucine incorporation into protein (Kirchman et al. 1985) adapted for soil (Bååth et al. 2001; Rousk et al. 2009), after incubation for ~ 2 h at 17 °C. Fungal growth rate was measured by estimating 14C-labelled acetate incorporation in ergosterol on 0.5 g of soil incubated for ~ 4 h at 17 °C, which also yielded fungal biomass estimated from total ergosterol content (Newell and Fallon 1991; Bååth et al. 2001; Rousk et al. 2009). Gross N-mineralisation was measured as the change in 15N/14N ammonium pools over a ~ 20 h incubation period (17 °C) after adding 67 µl 15NH4Cl solution (45 µg N ml−1) to larger subsamples (3 g). The sample size was based on a thorough sensitivity test by Rousk et al. (2016) on similar highly organic soils. N-mineralisation rates were calculated for two replicates per site according to Bengtson et al. (2005). At the end of the experiment, 0.5 g of soil was analysed for phospholipid fatty acids (PLFA) according to Frostegård et al. (1993). This was done on the two replicates used for N-mineralisation (Fig. 1). PLFAs used to indicate bacterial biomass were 10Me16:0a, 10Me16:0b, 10Me17:0, 10Me18:0, i14:0, i15:0, a15:0, i16:0, 16:1ω9, 16:1ω7c, 16:1ω7t, i17:0, a17:0, 17:1ω8, cy17:0, 18:1ω7 and cy19:0, while 18:2ω6,9 was used to estimate fungal biomass (Fig. 2f). 19:0 was used as internal standard to obtain the quantities of the fatty acids (Frostegård and Bååth 1996). Before and after the campaign, pHH2O, SOM and moisture contents were determined in all microcosms.
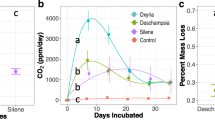
Respiration (a), gross N-mineralisation (b), bacterial growth (c) and fungal growth (d) cumulated over 64 days. The amount of added C in substrate increases from left to right due to increasing substrate C:N (see Table 2). The height of the bars shows mean values and the error bars show 1 standard error around the mean. Different letters indicate significantly different values identified by a Tukey’s HSD post hoc test
PLFA-based estimates of total (a), bacterial (b), and fungal (c) biomasses, as well as fungal:bacterial ratios (d) after 64 days of incubation. Different small letters above the bars indicate significant differences between treatments (one-way ANOVA followed by a Tukey’s HSD post hoc test). The lower panels show the ordinations of community structures (e) and the variable loadings (f) of the PCA analysis. For clarity, the error bars are not shown in the lower left panel, but standard errors were < 0.1
Statistical analyses
Microbial process rates and mineralisation rates over the duration of the experiment were summed over time to estimate cumulative growth and mineralisation. To test for treatment effects, the cumulative values for the duration of the experiments were compared with one-way ANOVAs. Post-hoc (Tukey HSD) tests were applied to identify significantly different treatments. All cumulated data was log-transformed to conform to the assumptions of parametric analysis. We tested for interaction between pH x substrate on growth rates as well as C and N-mineralisation rates, but no significant interactions were found. Test results in Figs. 1 and 2 and Table 2 are for all the data combined (3 sites combined for Figs. 1, 2, Table 2), while test results in the supplementary material are resolved into the sites individually (3 replicates per treatment for Figure S1, S3, S4; 2 replicates per treatment for Figure S2). A principle component analysis (PCA) was applied to analyse variation in the PLFA data on relative abundance data (mol %) after centering and standardising to unit variance. All analyses where made in R version 3.3.3 (R Core Team©, Vienna, Austria).
Results
Site characteristics
The sampling sites were similar with regard to most variables (Table 1). Notable exceptions were the inorganic ammonium content, which was an order of magnitude higher at the higher site compared to the lower (F2,3 = 21.74, p < 0.01). However, neither the total inorganic nitrogen (F2,6 = 0.37, p = 0.57) nor phosphorous (P) (F2,6 = 0.48, p = 0.51) sorbed to the resin capsules showed any differences between sites. All soils were acidic with a pH range of 4.1–4.8, highest at the higher site (F2,6 = 13.50, p < 0.01), where also the C:N was lowest; C:N ranged between 24 and 31 (F2,9 = 9.43, p = 0.01). These differences were also reflected by the ground vegetation, dominated by grasses and forbs at the higher site, compared to the common heath dwarf shrubs at the lower and middle sites (Empetrum sp. and Vaccinium sp.).
C-mineralisation
Cumulative soil respiration was inhibited by Ni-addition (F5,48 = 17.93, p < 0.01, Fig. 1a, Table 2, Figure S1), whereas addition of frass, frass + litter or pure litter yielded significantly higher respiration rates than the control (Fig. 1a, Table 2). Respiration in the litter + Ni treatments were not significantly different than in the control, yet significantly higher than the pure Ni treatment and significantly lower than the frass and frass + litter treatments. Microorganisms respired a significantly higher fraction of the C when added as frass (27 ± 2%, mean ± SE), than when added as frass + litter (14 ± 2) or pure litter (11 ± 2%) (F3,20 = 8.03, p < 0.01, Table 2). The litter + Ni treatment showed an insignificantly lower respired fraction of 5 ± 5% of added C compared to the litter and frass + litter treatments. We used the respiration rate at day 1 per added substrate-C as an operative index for substrate quality (Fierer et al. 2005, 2006), but no significant differences were found (ANOVA, F3,32 = 1.69, p = 0.19).
Gross N-mineralisation
Cumulative gross N mineralisation showed significant differences among treatments (F5,30 = 2.89, p = 0.03, Fig. 1b). The post hoc test was unable to identify pairwise differences, but we note that the mean gross N-mineralisation was lower in the litter and the pure Ni treatments than in the frass and litter + Ni treatments (Fig. 1b). This pattern was consistent at all sites, although the differences were insignificant at the middle site (p = 0.28, Figure S2d-f). All treatments exhibited an initial peak of varying duration and magnitude in N-concentration after substrate addition followed by a roughly exponential decrease in N-concentration (Figure S2a–c). The mineralised fraction of added organic N from litter or frass showed significant variation between treatments (F3,20 = 3.12, p = 0.05, Table 2). The frass treatment showed mineralisation rates of 17 ± 4% of the added organic N, while the litter treatment showed negative values (− 8 ± 7%, less N mineralised than in control) and frass + litter had intermediate mineralisation rates (9 ± 7%). Only the litter + Ni treatment (34 ± 15%) was significantly higher than the pure litter treatment. The C:N of mineralisation varied with the litter + Ni and frass treatments being significantly lower than the pure litter treatment, while only the litter + Ni treatment was also lower than the frass + litter treatment (F5,30 = 16.4, p < 0.01, Table 2).
Bacterial and fungal growth rates
Cumulative bacterial growth increased consistently with increasing C-addition (due to different C:N of substrates, Table 2), although treatment differences were not always distinguishable in pair-wise comparisons (F5,48 = 50.7, p < 0.01, Fig. 1c, Figure S3). Yet, the Ni-treatment decreased bacterial growth significantly. Bacterial growth rates roughly followed an exponentially decreasing curve after the initial peak after substrate addition (Figure S3).
Cumulative fungal growth showed a less clear pattern, with only frass and litter + Ni treatments being significantly higher than the control and Ni treatments (F5,48 = 16.79, p < 0.01, Fig. 1d, Figure S4). In contrast with bacterial growth, fungal growth was stimulated with addition of Ni, particularly when Ni was added in combination with litter. The exceptionally high fungal growth rate in the litter + Ni treatment only slowly decreased throughout the experiment (Figure S4).
Microbial biomass and community composition
At the end of the experiment, the total PLFA estimated microbial biomass averaged 934 ± 44 (range 459–1494) nmol g SOM−1, and was significantly lower in the Ni treatment than all other treatments, except litter + Ni (F5,30 = 6.74, p < 0.01, Fig. 2a). Bacterial biomass was also significantly lower in the Ni and litter + Ni treatments compared to the control, while bacterial biomass in the pure Ni treatment was also significantly lower than the frass + litter and pure litter treatments (F5,30 = 6.43, p < 0.01, Fig. 2b). Overall, bacterial biomass averaged 493 ± 27 nmol g SOM−1 (range 200–872), while fungal biomass averaged 53 ± 2 nmol g SOM−1 (range 35–72) and did not vary much across treatments (only the litter treatment was significantly higher than the control and Ni, F5,30 = 3.91, p < 0.01, Fig. 2c). Hence, the observed variation in fungal:bacterial ratio (Fig. 2d) was mainly driven by a decreased bacterial biomass. The fungal:bacterial biomass ratio averaged 0.12 ± 0.01 (range 0.05–0.26), with the lowest ratios in the control treatments. Only the Ni and litter + Ni treatments showed significantly higher ratios than the control (F5,30 = 3.64, p = 0.01, Fig. 2d). In the PCA analysis, the treatments clearly separated along the PC-1 axis explaining 53% of the variation, where addition of Ni in particular shifted the community composition towards negative PC-values (Fig. 2e), which corresponded to the ordination of the fungal marker (18:2w6,9, Fig. 2f). The second component explained 16% of the variation, and varied somewhat according to soil pH (linear regression, p = 0.04).
Discussion
How do insect outbreaks influence litter C and N cycling?
Comparing the outbreak and non-outbreak treatments, we found that a significantly higher fraction of C was respired when aboveground litter was deposited as frass (~ 27%) rather than senesced litter (~ 11%) (Table 2). As an illustration of the potential significance of this finding for ecosystem carbon cycling, we conducted a simple scaling exercise. Assuming typical foliar biomass-C (~ 400 kg ha−1, Kjelvik and Kärenlampi 1975), outbreak defoliation rates (~ 75%, Olsson et al. 2017), and insect respiration (~ 20%, Metcalfe et al. 2014), this means that an additional ~ 100 kg C ha−1 would be released during outbreaks via insect or microbial respiration that would otherwise have remained relatively inert as leaf litter. This would correspond to roughly to a 30% decrease in soil C-accumulation from aboveground litter. More work on the relative importance of seasonal timing and indirect effects, such as altered plant allocation and changes in plant community composition, will be required to more accurately resolve the effects of the outbreaks on total soil C cycling (e.g. Saravesi et al. 2015; Arnold et al. 2016). Nonetheless, we found enhanced decomposition of added substrate-C in frass treatments compared to the pure litter treatments (Table 2), which may suggest that addition of substrates with high C:N (litter) induced severe N-limitation (Kamble and Bååth 2014) due to rapid immobilisation of available N; this is also supported by the consistently low ammonium concentrations in the litter treatments (Figure S2).
N-mineralisation tended to be higher, though not significantly so, when substrate was added as frass compared to litter (Fig. 1b), particularly when observed relative to added substrate N (frass = 17%, litter = − 8% (less than control), Table 2). Interestingly, the time-series of ammonium-N concentrations (Figure S2) suggested quite strong and rapid immobilisation capacities in all treatments (except the control), which is in line with most findings from temperate forests that suggest a temporal and spatial redistribution of N due to insect herbivory rather than a net loss from the system (Lovett and Ruesink 1995; Christenson et al. 2002; Lovett et al. 2002; Frost and Hunter 2007, 2008). This may be explained by lower N mineralisation rates in the frass + litter and litter treatments compared to pure frass treatments. However, if N was the growth limiting element, this would suggest that the growth of heterotrophs should have been lower in litter treatments compared to frass, which was not consistent with our results (Fig. 1c, d). Rather, when N was added in a less labile form (litter), more of it may have been immobilised as amino acids, i.e. never mineralised to ammonium, following the argument of Schimel and Bennett (2004), who suggested that the uptake of organic N-forms can account for a higher fraction of N-turnover in strongly N-limited systems. Several recent studies on high-latitude soils have reported results consistent with this hypothesis (Wild et al. 2013, 2014, 2015).
The lower increase in N-mineralisation relative to C-mineralisation was reflected in the C:N of mineralisation, which dropped significantly from 38 ± 3 when litter was added to 26 ± 1 during outbreaks (frass addition). Thus, in conclusion, our findings provided support for our first hypothesis (i) about higher resource turnover rates during outbreaks, yet with different magnitudes for C and N, with a more pronounced response for N.
How does a long-term increase in insect herbivory influence litter C and N cycling?
On average, only 5% of the added C was respired from the litter + Ni treatments compared to 11% from pure litter treatments (Table 2). If representative of field conditions across Subarctic birch forest, this suggested that Geometrid outbreaks may in the long run decrease decomposition rates of incoming organic matter, which may in turn lead to accumulation of organic matter in the soil (Knorr et al. 2005; Berg 2014). Therefore, despite strongly negative short-term effects of severe herbivore outbreaks on soil C-storage, the long-term effect, if it increases Ni-availability, may increase soil C storage and thereby mitigate climate change. The long-term herbivory simulation significantly increased the mineralised fraction of added N from values lower than in the control (− 8%) to increases of 34%. Combined with the decreased respiration rate (Table 2), this represented a substantial shift towards higher respiration:ammonification efficiency, i.e. the shift in C:N of mineralisation from 38 ± 3 to 20 ± 1 (Table 2). This provided support for hypothesis (ii) that a long-term increase in insect herbivory would decrease C-turnover and increase N-mineralisation of incoming aboveground litter, and corroborated recent findings close to our study area, where addition of labile organic matter over three consecutive years showed a similar shift towards lower SOM-turnover and higher N-mining (Rousk et al. 2016). The mineralised fractions of added substrates (Table 2) are based on the naïve assumption that all additional C or N mineralised compared to the control was from the added substrate, and ignored potential priming effects. Yet, the decoupling of C and N turnover presented in two recent studies (Rousk et al. 2016; Ehtesham and Bengtson 2017) suggest that this assumption is likely conservative in terms of C, as they both found negative C-priming when labile organic matter was added. Similarly, the higher N-mineralisation rate under higher background N-availability followed the predictions made by Schimel and Bennett (2004) that increasing N-availability would shift N-limited systems away from being dominated by uptake of organic N towards more inorganic N-forms. Overall, increasing insect herbivory could over time result in consequences for C and N cycling of aboveground litter input (Fig. 3). Following this conceptual model, the C-mineralisation of the added litter decreased, while the N-mineralisation increased, yielding a substantial decrease in the C:N of mineralisation. This in turn implied that a smaller amount of the litter added to the soil was decomposed thereby promoting soil C-accumulation of aboveground litter.
Conceptual model summarizing the hypothetical development in key soil microbial ecology and biogeochemical variables with the projected climate warming driven increase in long-term insect herbivory rates based on the present study. Increased herbivory will increase N-availability in the soil, which decreases the overall microbial biomass and the respired fraction of aboveground litter-C undertaken by microbes. In contrast, as the gross N-mineralisation increases, so does the importance of fungi relative to bacteria (fungi:bacteria), both in terms of biomass and growth rates. This drives a substantial decrease of the C:N of mineralisation (the ratio of respiration to gross N mineralisation), which, in turn, increases the fraction of foliar-C stored in the soil. Thus, everything else being equal, a climate change driven increase in insect herbivory, may shift the soil biogeochemistry towards higher mitigation potential
The influence of insect herbivory on soil microbial dynamics
Both bacteria (Fig. 1c) and fungi (Fig. 1d) showed similar growth results for severe (frass), moderate (frass + litter) and non-outbreak (litter) scenarios. This suggested that, in terms of microbial growth rates, the form in which organic matter from the canopy entered the soil was not very important, as long as the background N-concentration was unchanged. The PLFA data also showed insignificant differences in fungal:bacterial ratios and total microbial biomass between these treatments (Fig. 2d), so hypothesis (iii) predicting increased bacterial dominance over fungi when substrate was added as frass rather than litter, was not supported. This was surprising, as other studies suggest that bacteria was the primary coloniser of labile organic matter (e.g. Rousk and Bååth 2007), while a field study of faeces addition in the tundra found increases in the microbial biomass (van der Wal et al. 2004). In contrast with most expectations in the field (e.g. Wardle et al. 2004), but similar to other laboratory studies, inorganic N apparently stimulated fungal growth (Fig. 1d), which may have been a consequence of the reduced competition from bacteria (Rousk and Bååth 2007; Rousk et al. 2008), as all significant patterns were driven by reduced bacterial biomass rather than fungal increase. This contrasts the findings of Malik et al. (2016), who attributed the increased fungal:bacterial biomass and decreased substrate-C respiration following litter addition to increased fungal biomass. These shifts could be linked to the presumed more flexible resource-use efficiency of fungi compared to bacteria (Sterner and Elser 2002; Six et al. 2006). Inorganic N addition was the primary cause of variation in the PLFA-results, driving increased fungi:bacteria ratios due to decreased bacterial biomass (Fig. 2e, f). This was in line with hypothesis (iv) predicting that long-term herbivory would increase fungal dominance relative to bacteria, and other laboratory studies (Rousk and Bååth 2007), although not all differences were significant. It was, however, in disagreement with the field study by Kaukonen et al. (2013) finding decreasing soil fungi:bacteria after repeated defoliations, suggesting that in the field, plant + symbiont feedbacks (Saravesi et al. 2015; Parker et al. 2016) may be more important than the direct effects of litter degradation studied here. Similarly, our findings contradict the general trends for field studies in temperate and boreal forests (Frey et al. 2004; Treseder 2008), where chronic inorganic N fertilisation had a negative impact on fungal biomass, while bacteria showed no response. This could be due to decreased mycorrhizal biomass, hence may not reflect saprotroph dynamics, which further emphasises the challenge of disentangling direct and indirect consequences in field studies. It has also been suggested that P could be a limiting or co-limiting element in Subarctic systems (Hartley et al. 2010; Vincent et al. 2014). However, the consistently high resin-P concentrations found in our sites (Table 1) offered no evidence for any limitation by P, making it an unlikely explanation. Overall, our results suggested that the form of nitrogen (organic/inorganic) may be more important for microbial competition for substrates than the amount of N, even in a N-limited system. The main conceptual implications of long-term herbivory on soil ecology of the present study, i.e. decreased microbial biomass and increased fungi:bacteria, were summarized in Fig. 3. Further work with long-term addition studies with herbivore deposits should be conducted to investigate these differences. Combining different types of isotope-tracking pool dilution methods (e.g. Wild et al. 2015) with growth rate tools (Rousk and Bååth 2011) would form a powerful experimental platform to strengthen our understanding of the microbial underpinnings of C and N process rates. To test the validity of these predictions under future climate conditions, and to better inform ecosystem models, a promising way forward is the combination of laboratory experiments like the present with field approaches, to resolve direct and indirect consequences of herbivory and the significance of temporal variation.
Ecosystem impacts of insect litter transformation
It has been predicted that herbivory increases resource turnover in productive systems and decreases it in low productivity systems (Bardgett and Wardle 2003; Wardle et al. 2004; van der Putten et al. 2014). This is, however, based on two major assumptions; first, it assumes a simple positive relationship between C and N cycling (no decoupling); and second, that it is governed by trophic interaction only, i.e. all herbivore types have similar ecosystem effects. The complex relationship between C and N mineralisation we found in this study (and others before) challenges the first assumption. The recent review of belowground consequences of vertebrate herbivory by Andriuzzi and Wall (2017) challenges the second assumption, as herbivore types were found to be equally or more important than trophic effects for predicting soil process rates and ecological responses, particularly due to physical soil disturbance. Insect herbivores do not physically influence their environment to the same extent as vertebrates, but they tend to show larger variation in abundances (i.e. outbreak cycles). Thus, we argue that if insect herbivory is to be included into the conceptual framework for understanding ecosystem consequences of herbivores, additional modifications are needed, particularly in terms of temporal variability. Finally, Andriuzzi and Wall (2017) highlight the need for herbivory studies outside grassland ecosystems, which is also strongly relevant based on our results from a N poor forest ecosystem.
In conclusion, we showed that the transformation of foliage by Geometrid moths almost tripled the respired fraction of aboveground C added to the soil from 11% in a non-outbreak year to 27% during outbreaks. When accounting for the fraction respired by the insects themselves, this means that ~ 30% less foliar C was stored in the soil after a simulated outbreak. In parallel with this, the mineralised fraction of organic N in the added substrates increased during outbreaks, with a stronger response than for C. In contrast, the long-term consequence of increased herbivory, simulated by adding litter to soils with artificially raised inorganic N content, decreased the respired fraction of added C, but caused an increase in the mineralised fraction of added litter-N. Counterintuitively, this significant negative shift in the C:N of mineralisation from 38 to 20 suggests that the microbes targeted more N-rich components of the organic matter when N-availability was already high. These biogeochemical shifts coincided with an increased dominance of saprotrophic fungi and reduced bacteria. Other factors being equal, these herbivory induced changes to the soil biogeochemical fate of aboveground litter would result in higher litter-C storage in the soil in the longer term. We emphasize the need to develop a more sophisticated representation of the relationship between C and N cycling and to include the effects of different herbivore types to improve predictive models of herbivore impacts on ecosystem processes.
Change history
22 May 2018
Table 1 contained a typesetting error in the initial online publication. The original article has been corrected.
References
Andriuzzi WS, Wall DH (2017) Responses of belowground communities to large aboveground herbivores: meta-analysis reveals biome-dependent patterns and critical research gaps. Glob Change Biol 23(9):3857–3868. https://doi.org/10.1111/gcb.13675
Arnold AIM, Grüning M, Simon J et al (2016) Forest defoliator pests alter carbon and nitrogen cycles. R Soc Open Sci 3(10):1–7. https://doi.org/10.1098/rsos.160361
Bååth E, Pettersson M, Söderberg KH (2001) Adaptation of a rapid and economical microcentrifugation method to measure thymidine and leucine incorporation by soil bacteria. Soil Biol Biochem 33(11):1571–1574. https://doi.org/10.1016/S0038-0717(01)00073-6
Bardgett RD, Wardle DA (2003) Herbivore-mediated linkages between aboveground and belowground communities. Ecology 84(9):2258–2268. https://doi.org/10.1890/02-0274
Belovsky GE, Slade JB (2000) Insect herbivory accelerates nutrient cycling and increases plant production. Proc Natl Acad Sci USA 97(26):14412–14417
Bengtson P, Falkengren-Grerup U, Bengtsson G (2005) Relieving substrate limitation-soil moisture and temperature determine gross N transformation rates. Oikos 111(1):81–90. https://doi.org/10.1111/j.0030-1299.2005.13800.x
Berg B (2000) Litter decomposition and organic matter turnover in northern forest soils. For Ecol Manage 133:13–22. https://doi.org/10.1016/S0378-1127(99)00294-7
Berg B (2014) Decomposition patterns for foliar litter—A theory for influencing factors. Soil Biol Biochem 78:222–232. https://doi.org/10.1016/j.soilbio.2014.08.005
Bjerke JW, Karlsen SR, Høgda KA et al (2014) Record-low primary productivity and high plant damage in the Nordic Arctic Region in 2012 caused by multiple weather events and pest outbreaks. Environ Res Lett 9(8):084006. https://doi.org/10.1088/1748-9326/9/8/084006
Christenson LM, Lovett GM, Mitchell MJ et al (2002) The fate of nitrogen in gypsy moth frass deposited to an oak forest floor. Oecologia 131(3):444–452. https://doi.org/10.1007/s00442-002-0887-7
Conant RT, Ryan MG, Ågren GI et al (2011) Temperature and soil organic matter decomposition rates–synthesis of current knowledge and a way forward. Glob Change Biol 17(11):3392–3404. https://doi.org/10.1111/j.1365-2486.2011.02496.x
Crowther T, Todd-Brown K, Rowe C et al (2016) Quantifying global soil C losses in response to warming. Nature 104(540):104–108. https://doi.org/10.1038/nature20150
DeForest JL, Zak DR, Pregitzer KS et al (2004) Atmospheric nitrate deposition, microbial community composition, and enzyme activity in northern hardwood forests. Soil Sci Soc Am J 68(1):132–138. https://doi.org/10.2136/sssaj2004.1320
Ehtesham E, Bengtson P (2017) Decoupling of soil carbon and nitrogen turnover partly explains increased net ecosystem production in response to nitrogen fertilization. Sci Rep 7:46286. https://doi.org/10.1038/srep46286
Fierer N, Craine JM, McLauchlan K et al (2005) Litter quality and the temperature sensitivity of decomposition. Ecology 86(2):320–326
Fierer N, Colman BP, Schimel JP et al (2006) Predicting the temperature dependence of microbial respiration in soil: a continental-scale analysis Glob Biogeochem Cycles 20(3):GB3016. https://doi.org/10.1029/2005GB002644
Fog K (1988) The effect of added nitrogen on the rate of decomposition of organic matter. Biol Rev 63(3):433–462. https://doi.org/10.1111/j.1469-185X.1988.tb00725.x
Frey SD, Knorr M, Parrent JL et al (2004) Chronic nitrogen enrichment affects the structure and function of the soil microbial community in temperate hardwood and pine forests. For Ecol Manage 196(1):159–171. https://doi.org/10.1016/j.foreco.2004.03.018
Frost CJ, Hunter MD (2007) Recycling of nitrogen in herbivore feces: plant recovery, herbivore assimilation, soil retention, and leaching losses. Oecologia 151(1):42–53. https://doi.org/10.1007/s00442-006-0579-9
Frost CJ, Hunter MD (2008) Insect herbivores and their frass affect Quercus rubra leaf quality and initial stages of subsequent litter decomposition. Oikos 117:13–22. https://doi.org/10.1111/j.2007.0030-1299.16165.x
Frostegård Å, Bååth E (1996) The use of phospholipid fatty acid analysis to estimate bacterial and fungal biomass in soil. Biol Fertil Soils 22(1–2):59–65. https://doi.org/10.1007/BF00384433
Frostegård Å, Tunlid A, Bååth E (1993) Phospholipid fatty acid composition, biomass, and activity of microbial communities from two soil types experimentally exposed to different heavy metals. Appl Environ Microbiol 59(11):3605–3617
Hartley IP, Hopkins DW, Sommerkorn M, Wookey PA (2010) The response of organic matter mineralisation to nutrient and substrate additions in sub-arctic soils. Soil Biol Biochem 42(1):92–100. https://doi.org/10.1016/j.soilbio.2009.10.004
Hunter MD (2001) Insect population dynamics meets ecosystem ecology: effects of herbivory on soil nutrient dynamics. Agric For Entomol 3(2):77–84. https://doi.org/10.1046/j.1461-9563.2001.00100.x
ISO/TC IS 10381-6 (2009) Soil quality - Sampling - Part 6: Guidance on the collection, handling and storage of soil under aerobic conditions for the assessment of microbiological processes, biomass and diversity in the laboratory. International Organization for Standardization, Geneva, Switzerland. https://www.iso.org/standard/43691.html. Accessed 12 May 2015
Jenkinson DS (1988) The determination of microbial carbon and nitrogen in soil. In: Wilson JR (ed) Advances in nitrogen cycling in agricultural ecosystems. CAB International, Wallingford, pp 368–386
Jepsen JU, Hagen SB, Ims RA et al (2008) Climate change and outbreaks of the geometrids Operophtera brumata and Epirrita autumnata in Subarctic birch forest: evidence of a recent outbreak range expansion. J Anim Ecol 77(2):257–264. https://doi.org/10.1111/j.1365-2656.2007.0
Jepsen JU, Biuw M, Ims RA et al (2013) Ecosystem impacts of a range expanding forest defoliator at the forest-tundra ecotone. Ecosystems 16(4):561–575. https://doi.org/10.1007/s10021-012-9629-9
Kamble PN, Bååth E (2014) Induced N-limitation of bacterial growth in soil: effect of carbon loading and N status in soil. Soil Biol Biochem 74:11–20. https://doi.org/10.1016/j.soilbio.2014.02.015
Kaukonen M, Routsalainen AL, Wäli PR et al (2013) Moth herbivory enhances resource turnover in subarctic mountain birch forests? Ecology 94(2):267–272
Kirchman D, K’nees E, Hodson R (1985) Leucine incorporation and its potential as a measure of protein synthesis by bacteria in natural aquatic systems. Appl Environ Microbiol 49(3):599–607. https://doi.org/10.1128/AEM.67.4.1775-1782.2001
Kjelvik S, Kärenlampi L (1975) Plant biomass and primary production of fennoscandian Subarctic and Subalpine forests and of Alpine willow and heath ecosystems. In: Wielgolaski FE (ed) Fennoscandian tundra ecosystems. Springer, Berlin, pp 111–120
Knorr M, Frey SD, Curtis PS (2005) Nitrogen additions and litter decomposition: a meta-analysis. Ecology 86(12):3252–3257. https://doi.org/10.1890/05-0150
Kozlov MV, Zvereva EL (2017) Background insect herbivory: impacts, patterns and methodology. In: Lüttge U, Beyschlag W, Büdel B, Francis D (eds) Progress in botany. Springer, Berlin, pp 1–43
Lovett GM, Ruesink AE (1995) Carbon and nitrogen mineralization from decomposing gypsy moth frass. Oecologia 104:133–138. https://doi.org/10.1007/BF00328577
Lovett GM, Christenson LM, Groffman PM et al (2002) Insect defoliation and nitrogen cycling in forests. Bioscience 52(4):335–341. https://doi.org/10.1641/0006-3568(2002)052[0335:IDANCI]2.0.CO;2
Malik AA, Chowdhury S, Schlager V et al (2016) Soil fungal: bacterial ratios are linked to altered carbon cycling. Front Microbiol 7:1247. https://doi.org/10.3389/fmicb.2016.01247
Metcalfe DB, Asner GP, Martin RE et al (2014) Herbivory makes major contributions to ecosystem carbon and nutrient cycling in tropical forests. Ecol Lett 17(3):324–332. https://doi.org/10.1111/ele.12233
Metcalfe DB, Crutsinger GM, Kumordzi BB et al (2016) Nutrient fluxes from insect herbivory increase during ecosystem retrogression in boreal forest. Ecology 97(1):124–132. https://doi.org/10.1890/15-0302.1
Newell SY, Fallon RD (1991) Toward a method for measuring instantaneous fungal growth rates in field samples. Ecol Soc Am 72(5):1547–1559
Olsson P-O, Heliasz M, Jin H et al (2017) Mapping the reduction in gross primary productivity in subarctic birch forests due to insect outbreaks. Biogeosciences 14(6):1703–1719. https://doi.org/10.5194/bg-14-1703-2017
Parker TC, Sadowsky J, Dunleavy H et al (2016) Slowed biogeochemical cycling in Sub-arctic Birch Forest Linked to Reduced Mycorrhizal Growth and Community Change after a Defoliation Event. Ecosystems 20(2):316–330. https://doi.org/10.1007/s10021-016-0026-7
Rousk J, Bååth E (2007) Fungal and bacterial growth in soil with plant materials of different C/N ratios. FEMS Microbiol Ecol 62(3):258–267. https://doi.org/10.1111/j.1574-6941.2007.00398.x
Rousk J, Bååth E (2011) Growth of saprotrophic fungi and bacteria in soil. FEMS Microbiol Ecol 78(1):17–30. https://doi.org/10.1111/j.1574-6941.2011.01106.x
Rousk J, Demoling LA, Bahr A et al (2008) Examining the fungal and bacterial niche overlap using selective inhibitors in soil. FEMS Microbiol Ecol 63(3):350–358. https://doi.org/10.1111/j.1574-6941.2008.00440.x
Rousk J, Brookes PC, Bååth E (2009) Contrasting soil pH effects on fungal and bacterial growth suggest functional redundancy in carbon mineralization. Appl Environ Microbiol 75(6):1589–1596. https://doi.org/10.1128/AEM.02775-08
Rousk K, Michelsen A, Rousk J (2016) Microbial control of soil organic matter mineralization responses to labile carbon in subarctic climate change treatments. Glob Change Biol 22(12):4150–4161. https://doi.org/10.1111/gcb.13296
Saravesi K, Aikio S, Wäli PR et al (2015) Moth outbreaks alter root-associated fungal communities in subarctic mountain birch forests. Microb Ecol 69(4):788–797. https://doi.org/10.1007/s00248-015-0577-8
Schimel JP, Bennett J (2004) Nitrogen mineralization: challenges of a changing paradigm. Ecology 85(3):591–602. https://doi.org/10.1890/03-8024
Sistla SA, Moore JC, Simpson RT et al (2013) Long-term warming restructures Arctic tundra without changing net soil carbon storage. Nature 497(7451):615–618. https://doi.org/10.1038/nature12129
Six J, Frey SD, Thiet RK, Batten KM (2006) Bacterial and fungal contributions to carbon sequestration in agroecosystems. Soil Sci Soc Am J 70(2):555–569. https://doi.org/10.2136/sssaj2004.0347
Sjögersten S, Wookey PA (2009) The impact of climate change on ecosystem carbon dynamics at the Scandinavian mountain birch forest-tundra heath ecotone. Ambio 38(1):2–10. https://doi.org/10.1579/0044-7447-38.1.2
Sterner RW, Elser JJ (2002) Ecological Stoichiometry - The Biology of Elements from Molecules to the Biosphere. Princeton University Press, Princeton
Tarnocai C, Canadell JG, Schuur EAG et al (2009) Soil organic carbon pools in the northern circumpolar permafrost region. Glob Biogeochem Cycles 23(2):1–11. https://doi.org/10.1029/2008GB003327
Tenow O, Bylund H, Nilssen AC et al (2005) Long-term influence of herbivores on northern birch forests. Plant Ecol 180:165–181
Tenow O, Nilssen AC, Bylund H et al (2013) Geometrid outbreak waves travel across Europe. J Anim Ecol 82(1):84–95. https://doi.org/10.1111/j.1365-2656.2012.02023.x
Throop HL, Holland EA, Parton WJ et al (2004) Effects of nitrogen deposition and insect herbivory on patterns of ecosystem-level carbon and nitrogen dynamics: results from the CENTURY model. Glob Change Biol 10(7):1092–1105. https://doi.org/10.1111/j.1529-8817.2003.00791.x
Treseder KK (2008) Nitrogen additions and microbial biomass: a meta-analysis of ecosystem studies. Ecol Lett 11(10):1111–1120. https://doi.org/10.1111/j.1461-0248.2008.01230.x
van der Putten WH, Bardgett RD, Bever JD et al (2014) Plant-soil feedbacks: the past, the present and future challenges. J Ecol 101(2):265–276. https://doi.org/10.1111/1365-2745.12054
van der Wal R, Bardgett RD, Harrison KA et al (2004) Vertebrate herbivores control: cascading effects of faeces on tundra ecosystems. Ecography 27(2):242–252
Vincent AG, Sundqvist MK, Wardle DA, Giesler R (2014) ’Bioavailable soil phosphorus decreases with increasing elevation in a subarctic tundra landscape’. PLoS ONE 9(3):e92942. https://doi.org/10.1371/journal.pone.0092942
Wardle DA, Bardgett RD, Klironomos JN et al (2004) Ecological linkages between aboveground and belowground biota. Science 304(5677):1629–1633. https://doi.org/10.1126/science.1094875
Wild B, Schnecker J, Bárta J et al (2013) Nitrogen dynamics in Turbic Cryosols from Siberia and Greenland. Soil Biol Biochem 67:85–93. https://doi.org/10.1016/j.soilbio.2013.08.004
Wild B, Schnecker J, Alves RJE et al (2014) Input of easily available organic C and N stimulates microbial decomposition of soil organic matter in arctic permafrost soil. Soil Biol Biochem 75:143–151. https://doi.org/10.1016/j.soilbio.2014.04.014
Wild B, Schnecker J, Knoltsch A et al (2015) Microbial nitrogen dynamics in organic and mineral soil horizons along a latitudinal transect in western Siberia. Glob Biogeochem Cycles 29:567–582. https://doi.org/10.1002/2015GB005084
Acknowledgements
We thank the Abisko Scientific Research Station (ANS) for logistic support, and Jane U. Jepsen’s group at the Norwegian Institute for Nature Research (NINA)/University of Tromsø, Norway, for frass collection. We also thank Jens Arne Antoft and Thomas Heister for field assistance. This work was supported by Grants from the Swedish Research Council (Grant Nos. 2015-04942 and 2014-5177), the Swedish research council Formas (Grant No 941-2015-270), the Royal Physiographic Society of Lund (Kungliga fysiografen) and from Helge Ax:son Johnson’s Foundation (Helge Ax:son Johnsons Stiftelse).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Melany Fisk
Electronic supplementary material
Below is the link to the electronic supplementary material.
10533_2018_448_MOESM1_ESM.pdf
Supplementary material 1 (PDF 283 kb). Figure S1: Mean respiration rates (upper panels) and cumulative respiration (lower panels) over the course of the experiment at the lower (a,d), middle (b,e) and higher (c,f) sites. Error bars show standard errors. Different letters indicate significant differences, one-way ANOVA followed by a Tukey HSD test. Figure S2: Mean N-concentrations (upper panels) and cumulative N-mineralisation (lower panels) over the course of the experiment at the lower (a,d), middle (b,e) and higher (c,f) sites. Error bars show standard errors. Note that the primary Y-axis in panel c is different than in panel a and b. Filled symbols are plotted on the secondary Y-axis. Different letters indicate significant differences, one-way ANOVA followed by a Tukey’s HSD test. Figure S3: Mean bacterial growth rates (upper panels) and cumulative growth (lower panels) over the course of the experiment at the lower (a,d), middle (b,e) and higher (c,f) sites. Error bars show standard errors. Different letters indicate significant differences, one-way ANOVA followed by a Tukey’s HSD test. Figure S4: Mean fungal growth rates (upper panels) and cumulative growth (lower panels) over the course of the experiment at the lower (a,d), middle (b,e) and higher (c,f) sites. Error bars show standard errors. Different letters indicate significant differences, one-way ANOVA followed by a Tukey’s HSD test
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Kristensen, J.A., Metcalfe, D.B. & Rousk, J. The biogeochemical consequences of litter transformation by insect herbivory in the Subarctic: a microcosm simulation experiment. Biogeochemistry 138, 323–336 (2018). https://doi.org/10.1007/s10533-018-0448-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-018-0448-8