Abstract
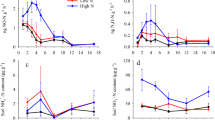
In tropical forests, multi-year studies on the impact of chronic nitrogen (N) enrichment on soil N-oxide fluxes are lacking. Our objectives were to: (1) assess the changes in soil N-oxide (NO+N2O) fluxes from a montane forest in response to 3–4 years of N addition and compare these to the response of a lowland forest with 11–12 years of N addition, (2) quantify the contributions of nitrification and denitrification to nitrous oxide (N2O) emissions, and (3) assess how deep in the soil profile the N2O concentrations are affected by N input. These measurements were conducted in montane and lowland forests in Panama in 2008–2009, which covered the last 2 years of a 4-year investigation that started in 2006. Each forest had a control and N addition treatment (125 kg urea N ha−1 year−1) with four replicate plots (1,600 m2 each) per treatment. N-oxide emissions from the montane forest started to increase within the first 2 years of treatment and continued to increase in the 3rd and 4th years of N addition, during which the emissions were already equivalent to those from the lowland forest with 11–12 years of N addition. The large N-oxide response of the montane forest to N addition were due to the large increases in gross nitrification rates in the organic layer and the high moisture contents of the mineral soil due to the high rainfall (5.5 ± 0.2 m year−1). In the lowland forest (2.7 ± 0.1 m rain year−1), N-oxide response to N addition was more pronounced in wet years (i.e. 2006–2007 with 5–17 % higher rainfall than average) than in dry years (i.e. 2008–2009 with 5–26 % lower rainfall than average). Denitrification was the dominant source of N2O not only for the organic layer and the top 5 cm of mineral soil but possibly also for the entire 2 m depth. Soil–air N2O concentrations were elevated by N addition down to at least 2 m. Our results suggest that the best indicators of these responses were the presence of an organic layer and rainfall quantity and seasonality.


Similar content being viewed by others
References
Adamek M, Corre MD, Holscher D (2009) Early effect of elevated nitrogen input on above-ground net primary production of a lower montane rainforest, Panama. J Trop Ecol 25:635–645
Adamek M, Corre MD, Holscher D (2011) Responses of fine roots to experimental nitrogen addition in a tropical lower montane rain forest, Panama. J Trop Ecol 27:73–81
Arnold J, Corre MD, Veldkamp E (2009) Soil N cycling in old-growth forests across an Andosol toposequence in Ecuador. For Ecol Manag 257:2079–2087
Bouwman AF, Van der Hock KW, Olivier JGJ (1995) Uncertainties in the global source distribution of nitrous oxide. J Geophys Res 100:2785–2800
Breuer L, Kiese R, Butterbach-Bahl (2002) Temperature and moisture effects on nitrification rates in tropical rain-forest soils. Soil Sci Soc Am J 66:834–844
Brookes PC, Landman A, Pruden G, Jenkinson DS (1985) Chloroform fumigation and the release of soil nitrogen: a rapid direct extraction method to measure microbial biomass nitrogen in soil. Soil Biol Biochem 17:837–842
Corre MD, Brumme R, Veldkamp E, Beese FO (2007) Changes in nitrogen cycling and retention processes in soils under spruce forests along a nitrogen enrichment gradient in Germany. Glob Change Biol 13:1509–1527
Corre MD, Veldkamp E, Arnold J, Wright SJ (2010) Impact of elevated N input on soil N cycling and losses in old-growth lowland and montane forests in Panama. Ecology 91:1715–1729
Crawley MJ (2002) The R Book. Wiley, Chichester
Davidson EA (1991) Fluxes of nitrous oxide and nitric oxide from terrestrial ecosystems. In: Rogers GE, Whitman WB (eds) Microbial production and consumption of greenhouse gases: methane, nitrogen oxides and halomethanes. American Society for Microbiology, Washington, pp 219–235
Davidson EA, Keller M, Erickson HE, Verchot LV, Veldkamp E (2000) Testing a conceptual model of soil emissions of nitrous and nitric oxides. Bioscience 50:667–680
Davidson EA, Ishida FY, Nepstad DC (2004) Effects of an experimental drought on soil emissions of carbon dioxide, methane, nitrous oxide, and nitric oxide in a moist tropical forest. Glob Change Biol 10:718–730
Firestone MK, Davidson EA (1989) Microbiological basis of NO and N2O production and consumption in soil. In: Andreae MO, Schimel DS (eds) Exchange of Trace Gases between Terrestrial Ecosystems and the Atmosphere. Wiley, New York, pp 7–21
Galloway JN, Townsend AR, Erisman JW, Bekunda M, Cai Z, Freney JR, Martinelli LA, Seitzinger SP, Sutton MA (2008) Transformation of the nitrogen cycle: recent trends, questions, and potential solutions. Science 320:889–892
Hall SJ, Matson PA (2003) Nutrient status of tropical rainforests influences soil N dynamics after N additions. Ecol Monogr 73:107–129
Hall SJ, Asner GP, Kitayama K (2004) Substrate, climate and land use controls over soil N dynamics and N-oxide emissions in Borneo. Biogeochemistry 70:27–58
Hietz P, Turner BL, Wanek W, Richter A, Nock CA, Wright SJ (2011) Long-term change in the nitrogen cycle of tropical forests. Science 334:664–666
Kiese R, Hewett B, Graham A, Butterbach-Bahl K (2003) Seasonal variability of N2O emissions and CH4 uptake by tropical rainforest soils of Queensland, Australia. Glob Biogeochem Cycles 17:1043. doi:10.1029/2002GB002014
Knowles R (1982) Denitrification. Microbiol Rev 46:43–70
Koba K, Osaka K, Tobari Y, Toyoda S, Ohte N, Katsuyama M, Suzuki N (2009) Biogeochemistry of nitrous oxide in groundwater in a forested ecosystem elucidated by nitrous oxide isotopomer measurements. Geochim Cosmochim Acta 73(11):3115–3133
Koehler B, Corre MD, Veldkamp E, Wullaert H, Wright SJ (2009) Immediate and long-term nitrogen oxide emissions from tropical forest soils exposed to elevated nitrogen input. Glob Change Biol 15:2049–2066
Koehler B, Zehe E, Corre MD, Veldkamp E (2010) An inverse analysis reveals limitations of the soil-CO2 profile method to calculate CO2 production and efflux for well-structured soils. Biogeosciences 7:2311–2325
Koehler B, Corre MD, Steger K, Well R, Zehe E, Sueta JP, Veldkamp E (2012) An in-depth look into a tropical lowland forest soil: nitrogen-addition effects on the contents of N2O, CO2 and CH4 and N2O isotopic signatures down to 2 m depth. Biogeochem 111: 695–713. Erratum 111:715–717
Loftfield N, Flessa H, Augustin J, Beese F (1997) Automated gas chromatographic system for rapid analysis of the atmospheric trace gases methane, carbon dioxide, and nitrous oxide. J Environ Qual 26:560–564
Martinson G, Corre MD, Veldkamp E (2013) Responses of nitrous oxide fluxes and soil nitrogen cycling to nutrient additions in montane forests along an elevation gradient in southern Ecuador. Biogeochemistry 112:625–636
Ostrom NE, Pitt A, Sutka RL, Ostrom PH, Grandy AS, Huizinga KM, Robertson GP (2007) Isotopologue effects during N2O reduction in soils and in pure cultures of denitrifiers. J Geophys Res 112:G02005. doi:10.1029/2006JG000287
Panek JA, Matson PA, Ortíz-Monasterio I, Brooks P (2000) Distinguishing sources of N2O in a Mexican wheat system using 15N. Ecol Appl 10:506–514
R Development Core Team (2010) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. ISBN 3-900051-07-0, URL http://www.R-project.org
Schuur EAG, Matson PA (2001) Net primary productivity and nutrient cycling across a mesic to wet precipitation gradient in Hawaiian montane forest. Oecologia 128: 431–442
Snider DM, Venkiteswaran JJ, Schiff SL, Spoelstra J (2012) Deciphering the oxygen isotope composition of nitrous oxide produced by nitrification. Glob Change Biol 18:356–370
Sotta ED, Corre MD, Veldkamp E (2008) Differing N status and N retention processes of soils under old-growth lowland forest in Eastern Amazonia, Caxiuanã, Brazil. Soil Biol Biochem 40:740–750
Tanner EVJ, Vitousek PM, Cuevas E (1998) Experimental investigation of nutrient limitation of forest growth on wet tropical mountains. Ecology 79:10–22
Wright SJ, Yavitt JB, Wurzburger N, Turner BL, Tanner EVJ, Sayer EJ, Santiago LS, Kaspari M, Hedin LO, Harms KE, Garcia MN, Corre MD (2011) Potassium, phosphorus or nitrogen limit root allocation, tree growth and litter production in a lowland tropical forest. Ecology 92:1616–1625
Yienger JJ, Levy H (1995) Empirical model of global soil-biogenic NOx emissions. J Geophys Res 100:11447–11464
Acknowledgments
This study was funded by the Robert Bosch Foundation (Germany) for M.D. Corre’s independent research group, NITROF, and by the Deutsche Forschungsgemeinschaft (German Research Foundation, Co 749/1-1). The Smithsonian Tropical Research Institute and ANAM, Panama provided invaluable administrative and technical support. S. Joseph Wright hosted our study in the Gigante nutrient manipulation experiment. The help of the NITROF assistants (Rodolfo Rojas, Erick Diaz and Ignacio Del Cid), and the SSTSE laboratory technicians (especially Kerstin Langs) are highly appreciated.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Jan Mulder
Rights and permissions
About this article
Cite this article
Corre, M.D., Sueta, J.P. & Veldkamp, E. Nitrogen-oxide emissions from tropical forest soils exposed to elevated nitrogen input strongly interact with rainfall quantity and seasonality. Biogeochemistry 118, 103–120 (2014). https://doi.org/10.1007/s10533-013-9908-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-013-9908-3