Abstract
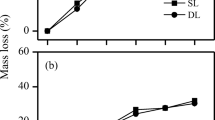
Carbon dynamics during litter decomposition have been described in a variety of forest ecosystems and provided insights into carbon flow in soils. To quantitatively assess how decomposition processes vary between litter types, solid-state 13C cross-polarization and magic-angle spinning nuclear magnetic resonance (CPMAS NMR) technique was applied to analyze conifer (cedar, cypress) and hardwood (chinquapin, beech, oak, birch) litter which had degraded during a 3 year litterbag experiment throughout Japan. The results were used to identify compositional changes and estimate decomposition constants (k values) in exponential equations. Total litter and carbon type mass losses during decomposition varied significantly between litter types, being affected by the initial physicochemical litter quality. Concomitant increases and decreases in carbonyl and O/N-alkyl C compositions, respectively, were observed for all litter types, but aromatic and aliphatic C dynamics were less consistent. In hardwoods, [aromatic/aliphatic C ratio] was generally stable during decomposition, suggesting that, in hardwoods, the decomposabilities of aromatic and aliphatic C were similar. In the conifers, an increasing [aromatic/aliphatic C ratio] during decomposition suggested that aromatic C was more recalcitrant than aliphatic C. These results suggest that different decomposition processes between litter types might be related to different aromatic and aliphatic C behaviors, as affected by lignin stability and lipid leachability and biosynthesis. Variations in the k values for total litter and carbon types were not obvious between litter types, although the mass loss patterns differed significantly. The k values estimated in this study may contribute to predictions of soil carbon dynamics and the validation of carbon compartment models in forest ecosystems.






Similar content being viewed by others
References
Baldock JA, Preston CM (1995) Chemistry of carbon decomposition processes in forests as revealed by solid-state carbon-13 nuclear magnetic resonance. In: McFee WW, Kelly JM (eds) Carbon forms and functions in forest soils. SSSA Inc, Wisconsin, pp 89–117
Baldock JA, Oades JM, Nelson PN, Skene TM, Golchin A, Clarke P (1997) Assessing the extent of decomposition of natural organic materials using solid-state 13C NMR spectroscopy. Aust J Soil Res 35:1061–1083
Berg B, McClaugherty C (2003) Plant litter, decomposition, humus formation, carbon sequestration. Springer-Verlag, Germany
Chapin FS, Matson PA, Mooney HA (2002) Principles of terrestrial ecoststem ecology. Springer-Verlag, USA
Coleman K, Jenkinson DS (2008) RothC 26.3. A model for the turnover of carbon in soil. Model description and windows user guide. www.rothamsted.bbsrc.ac.uk/aen/carbon/mod26_3_win.pdf
Crossley DAJ, Hoglund MP (1962) A litter bag method for the study of microarthropods inhabiting leaf litter. Ecology 43:571–573
Cwynaw LC (1982) A lake-quaternary vegetation history from Hanging lake, northern Yukon. Ecol Monogr 52:1–24
Davidson EA, Janssens IA (2006) Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature 440:165–173
Feagri K, Iversen J (1989) Textbook of pollen analysis, 4th edn. Wiley, Chichester, p 328
FFPRI (1998a) Operational forest planning in Tsukuba experimental site. FFPRI, Tsukuba
FFPRI (1998b) Operational forest planning in Tengakura experimental site. FFPRI, Tsukuba
Fründ R, Lüdeman H-D (1991) Quantitative characterization of soil organic matter and its fractionation products by solid state high resolution C-13 (CPMAS) spectroscopy. Z Naturforsch 46c:982–988
Government Forest Experiment Station (1958) Handbook of wood industry. Maruzen Ltd, Tokyo, Japan
Hiradate S, Yonezawa T, Takesako H (2006) Isolation and purification of hydrophilic fulvic acids by precipitation. Geoderma 132:196–205
Hopkins DW, Chudek JA, Webster EA, Barraclough D (1997) Following the decomposition of ryegrass labeled with 13C and 15N in soil by solid-state nuclear magnetic resonance spectroscopy. Eur J Soil Sci 48:623–631
IUSS Working Group (2006) World Reference Base for Soil Resource 2006: A Framework for International Classification, Correlation and Communication: World Soil Resource Reports, vol 103
Jenkinson DS, Rayner JH (2006) The turnover of soil organic matter in some of the Rothamsted classical experiments. Soil Sci 171:S130–S137
Jenny H (1941) Factors of soil formation. McGraw-Hill, New York
Kögel-Knabner I (1997) 13C and 15N NMR spectroscopy as a tool in soil organic matter studies. Geoderma 80:243–270
Kögel-Knabner I, Zech W, Hatcher PG (1988) Chemical composition of the organic matter in forest soils: the humus layer. Z Pflanzenernähr Bodenk 151:331–340
Lemma B, Nilsson I, Kleja DB, Slsson M, Knicher H (2007) Decomposition and substrate quality of leaf litter and fine roots from three exotic plantations and a native forest in the southwestern highlands of Ethiopia. Soil Bio Biochem 39:2317–2328
Liski J, Palosou T, Peltoniemi M, Sievänen R (2005) Carbon and decomposition model Yasso for forest soils. Ecol Model 189:168–182
Lorenz K, Preston CM, Raspe S, Morrison IK, Feger KH (2000) Litter decomposition and humus characteristics in Canadian and German spruce ecosystems: information from tannin analysis and 13C CPMAS NMR. Soil Biol Biochem 32:779–792
Nakano J, Higuchi T, Sumimoto M, Ishizu A (1983) Wood chemistry. Yuni Shuppan Ltd., Tokyo
Nakashizuka T, Matsumoto Y (2002) Diversity and interaction in a temperate forest community. Ogawa Forest Reserve of Japan. Springer-Verlag, Japan
Nakazawa F, Suzuki K (2008) The alternation in the pollen concentration peak in a melting snow cover. Bull Glaciol Res 25:1–7
Ohnuki Y, Ikuzawa H, Terazono R (1998) Relationship between soil erosion and physical properties of soils at zero-order basins in Okinawa Japan. Jpn J For Environ 40:17–25
Olson J (1963) Energy storage and the balance of producers and decomposers in ecological systems. Ecology 44:322–331
Ono K, Hirai K, Morita S, Ohse K, Hiradate S (2009) Organic carbon accumulation processes on a forest floor during an early humification stage in a temperate deciduous forest in Japan: Evaluations of chemical compositional changes by 13C NMR and their decomposition rates from litterbag experiment. Geoderma 151:351–356
Ono K, Hiradate S, Morita S, Ohse K, Hirai K (2011) Humification processes of needle litter on forest floors in Japanese cedar (Cryptomeria japonica) and Hinoki cypress (Chamaecyparis obtusa) plantations in Japan. Plant Soil 338:171–181
Osono T, Takeda H, Azuma J (2008) Carbon isotope dynamics during leaf litter decomposition with reference to lignin fractions. Ecol Res 23:51–55
Parton WJ, Schimel DS, Cole CV, Ojima DS (1987) Analysis of factors controlling soil organic matter levels in Great Plains grasslands. Soil Sci Soc Am J 51:1173–1179
Preston CM, Nault JR, Trofymow JA (2010) Chemical changes during 6 years of decomposition of 11 litter in some Canadian forest sites. Part 2. 13C abundance, solid-state 13C NMR spectroscopy and thee meaning of “lignin”. Ecosystems 12:1078–1102
Quideau SA, Graham RC, Oh S-W, Hendrix PF, Wasylishen RE (2005) Leaf litter decomposition in a chaparral ecosystem, Southern California. Soil Biol Biochem 37:1988–1998
SAS Institute (2010) JMP statistics and graphic guide, version 9.0.0. SAS Institute Inc, Cary
Skene TM, Skjemstad JO, Oades JM, Clarke PJ (1996) The influence of inorganic matrices on the decomposition of straw. Aust J Soil Res 34:413–426
Skene TM, Skjemstad JO, Oades JM, Clarke PJ (1997) The influence of inorganic matrices on the decomposition of Eucalyptus litter. Aust J Soil Res 35:73–87
Skjemstad JO, Clarke P, Golchin A, Oades JM (1997) Characterization of soil organic matter by solid-state 13C NMR spectroscopy. In: Cadisch G, Giller KE (eds) Driven by nature. Plant litter quality and decomposition. CABI Publishing, Oxon, pp 253–271
Suzuki S, Ishizuka S, Kitamura K, Yamanoi K, Nakai Y (2006) Continuous estimation of winter carbon dioxide efflux from the snow surface in a deciduous broadleaf forest. J Geophys Res 111:D17101
Syafii W, Yoshimoto T, Samejima M (1988) The effect of lignin structure on decay resistance of some tropical woods. Bull Tokyo Univ For 80:69–77
Trofymow JA, CIDET Working Group (1998) CIDET–The Canadian Intersite Decomposition Experiment: Project and Site Establishment Report. Information Report BC-X-378. Pacific Forestry Centre, Victoria, British Columbia
Acknowledgments
We are grateful to Drs. Shinji Kaneko, Masamichi Takahashi, Yojiro Matsuura, Makoto Araki, Shigehiro Ishizuka for their valuable advice and comments, and we thank Ms. Yumiko Okazaki, Teru Notsukidaira, and Keiko Sawai for their help with sample preparation and laboratory analysis. We wish to express our appreciation to the staff of the Department of Forest Site Environment, the Tohoku Research Center, and the Arboretum and Nursery Office in the Forestry and Forest Products Research Institute, as their advice and assistance were extremely helpful during our fieldwork and experiments. This study was supported in part by a program of the Japanese Ministry of the Environment, entitled “Evaluation, Adaptation and Mitigation of Global Warming in Agriculture, Forestry, and Fisheries: Research and Development (A1120)” and the program of the Japanese Ministry of Education, Culture, Sports, Science, and Technology for Young Scientists, entitled “Study on the production process of humic substances in forest soil by noting recalcitrant lignin compounds (No. 20780122)”.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ono, K., Hiradate, S., Morita, S. et al. Fate of organic carbon during decomposition of different litter types in Japan. Biogeochemistry 112, 7–21 (2013). https://doi.org/10.1007/s10533-011-9682-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-011-9682-z