Abstract
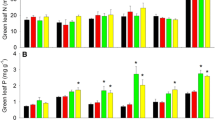
It is commonly assumed that nitrogen (N) is the primary mineral resource limiting the productivity of temperate forests. Sustained inputs of N via atmospheric deposition are altering the N status of temperate forests raising the possibility that nutrients such as phosphorus (P) are increasingly limiting productivity. The objective of this study was to determine whether P availability limits tree growth alone or in combination with N. This study was conducted in two forest types common throughout the New England landscape of the northeastern United States; in sugar maple and white ash dominated stands growing on base rich parent material characterized by rapid rates of N cycling and high N availability, and in red oak–beech–hemlock dominated stands growing on base-poor parent material characterized by slow rates of N cycling and low N availability. Starting in 2004, N and P were added to replicate plots in each forest type in factorial combination at a rate of 150 and 50 kg ha−1 year−1, respectively. Diameter growth rates of all trees >10 cm DBH were measured in 2005 and 2006 using dendrometer bands and converted into units of basal area increment (BAI) and wood production. Following 2 years of fertilization, basal area increment in the sugar maple–white ash forests remained strongly N limited. Fertilization with P did not significantly increase BAI alone, although both N and P fertilization tended (P < 0.10) to increase diameter growth in white ash. Wood production in the N-fertilized plots increased by 100 g C m−2 year−1, roughly doubling production in the non-fertilized plots. In the red oak–beech–hemlock stands, there was no overall effect of N or P fertilization on BAI or wood production because BAI in some species was stimulated by fertilization with N alone (e.g., black cherry, red oak), while in other species BAI was unaffected (e.g., red maple, beech) or negatively affected by fertilization with N or P (e.g., eastern hemlock). Given that BAI in several tree species responded to fertilization with N alone and that only one species responded to P fertilization once N was added, this study suggests that decades of atmospheric N deposition have not (yet) resulted in widespread P limitation or saturation of tree demand for N.


Similar content being viewed by others
References
Aber JD, Goodale CL, Smith ML, Magill AH, Martin ME, Hallet RA, Stoddard JL (2003) Is nitrogen deposition altering the nitrogen status of northeastern forests? Bioscience 53:375–389. doi:10.1641/0006-3568(2003)053[0375:INDATN]2.0.CO;2
Auchmoody L (1982) Response of young black cherry stands to fertilization. Can J For Res 12:319–325
Auchmoody L, Filip S (1973) Forest fertilization in the eastern united states: hardwoods. USDA Forest Service General Technica l Report NE-3. pp 211–225
Bernier B, Brazeau M (1988) Nutrient deficiency symptoms associated associated with sugar maple dieback and decline in the Quebec Appalachians. Can J For Res 18:762–767. doi:10.1139/x88-116
Binkley D, Hart S (1989) The components of nitrogen availability in forest soils. Adv Soil Sci 10:57–112
Crews TE, Kitayama K, Fownes JH, Riley RH, Herbert DA, Mullerdombois D, Vitousek PM (1995) Changes in soil-phosphorus fractions and ecosystem dynamics across a long chronosequence in Hawaii. Ecology 76:1407–1424. doi:10.2307/1938144
Cross AF, Schlesinger WH (1995) A literature-review and evaluation of the Hedley fractionation—applications to the biogeochemical cycle of soil-phosphorus in natural ecosystems. Geoderma 64:197–214. doi:10.1016/0016-7061(94)00023-4
Davidson EA, Chorover J, Dail DB (2003) A mechanism of abiotic immobilization of nitrate in forest ecosystems: the ferrous wheel hypothesis. Glob Chang Biol 9:228–236. doi:10.1046/j.1365-2486.2003.00592.x
De Vries W, Reinds GJ, Gundersen P, Sterba H (2006) The impact of nitrogen deposition on carbon sequestration in European forests and forest soils. Glob Chang Biol 12:1151–1173. doi:10.1111/j.1365-2486.2006.01151.x
de Vries W, Solberg S, Dobbertin M, Sterba H, Laubhahn D, Reinds GJ, Nabuurs GJ, Gundersen P, Sutton MA (2008) Ecologically implausible carbon response? Nature 451:E1–E3. doi:10.1038/nature06579
Elser JJ, Bracken MES, Cleland EE, Gruner DS, Harpole WS, Hillebrand H, Ngai JT, Seabloom EW, Shurin JB, Smith JE (2007) Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol Lett 10:1135–1142. doi:10.1111/j.1461-0248.2007.01113.x
Fahey TJ, Battles JJ, Wilson GF (1998) Responses of early successional northern hardwood forests to changes in nutrient availability. Ecol Monogr 68:183–212
Finzi AC, Berthrong ST (2005) The uptake of amino acids by microbes and trees in three cold-temperate forests. Ecology 86:3345–3353. doi:10.1890/04-1460
Finzi AC, van Breemen N, Canham CD (1998a) Canopy tree soil interactions within temperate forests: species effects on soil carbon and nitrogen. Ecol Appl 8:440–446
Finzi AC, Canham CD, van Breemen N (1998b) Canopy tree soil interactions within temperate forests: species effects on pH and cations. Ecol Appl 8:447–454
Goodale CL, Aber JD (2001) The long-term effects of land-use history on nitrogen cycling in northern hardwood forests. Ecol Appl 11:253–267. doi:10.1890/1051-0761(2001)011[0253:TLTEOL]2.0.CO;2
Gradowski T, Thomas SC (2006) Phosphorus limitation of sugar maple growth in central Ontario. For Ecol Manage 226:104–109. doi:10.1016/j.foreco.2005.12.062
Hall B, Motzkin G, Foster DR, Syfert M, Burk J (2002) Three hundred years of forest and land-use change in Massachusetts, USA. J Biogeogr 29:1319–1335. doi:10.1046/j.1365-2699.2002.00790.x
Hill DE, Sautter EH, Gunick WN (1980) Soils of connecticut. Connecticut agricultural experiment station bulletin number 787
Holland EA, Braswell BH, Sulzman J, Lamarque JF (2005) Nitrogen deposition onto the United States and western Europe: synthesis of observations and models. Ecol Appl 15:38–57. doi:10.1890/03-5162
Hyvonen R, Persson T, Andersson S, Olsson B, Agren GI, Linder S (2008) Impact of long-term nitrogen addition on carbon stocks in trees and soils in northern Europe. Biogeochemistry 89:121–137. doi:10.1007/s10533-007-9121-3
Jenkins JC, Chojnacky DC, Heath LS, Birdsey RA (2004) Comprehensive database of diameter-based biomass regressions for north American tree species. United States department of agriculture forest service general technical report NE-319. pp.48
Killingbeck KT (1996) Nutrients in senesced leaves: keys to the search for potential resorption and resorption proficiency. Ecology 77:1716–1727. doi:10.2307/2265777
Larson GJ, Stone BD (1980) Late Wisconsinian glaciation of New England. Kendall Hunt Publishing Company, Dubuque
Latty EF, Canham CD, Marks PL (2003) Beech bark disease in northern hardwood forests: the importance of nitrogen dynamics and forest history for disease severity. Can J For Res 33:257–268. doi:10.1139/x02-183
Lawrence D, Schlesinger WH (2001) Changes in soil phosphorus during 200 years of shifting cultivation in Indonesia. Ecology 82:2769–2780
Lea R, Tierson WC, Leaf AL (1979) Growth responses of northern hardwoods to fertilization. For Sci 25:597–604
LeBauer DS, Treseder KK (2008) Nitrogen limitation of net primary productivity in terrestrial ecosystems is globally distributed. Ecology 89:371–379. doi:10.1890/06-2057.1
Lindsay W, Vlek P (1977) Phosphate minerals. In: Dixon J, Weed S (eds) Minerals in soil environments. Soil Science Society of America, Madison, pp 639–672
Lovett GM, Weathers KC, Sobczak WV (2000) Nitrogen saturation and retention in forested watersheds of the Catskill Mountains, New York. Ecol Appl 10:73–84. doi:10.1890/1051-0761(2000)010[0073:NSARIF]2.0.CO;2
Lovett GM, Weathers KC, Arthur MA, Schultz JC (2004) Nitrogen cycling in a northern hardwood forest: do species matter? Biogeochemistry 67:289–308. doi:10.1023/B:BIOG.0000015786.65466.f5
Lowther JR (1980) Use of a single sulfuric acid-hydrogen peroxide digest for the analysis of Pinus radiata needles. Commun Soil Sci Plant Anal 11:175–188. doi:10.1080/00103628009367026
Magill AH, Aber JD, Berntson GM, McDowell WH, Nadelhoffer KJ, Melillo JM, Steudler P (2000) Long-term nitrogen additions and nitrogen saturation in two temperate forests. Ecosystems (N Y Print) 3:238–253. doi:10.1007/s100210000023
Magill AH, Aber JD, Currie WS, Nadelhoffer KJ, Martin ME, McDowell WH, Melillo JM, Steudler P (2004) Ecosystem response to 15 years of chronic nitrogen additions at the Harvard forest LTER, Massachusetts, USA. For Ecol Manage 196:7–28. doi:10.1016/j.foreco.2004.03.033
Magnani F, Mencuccini M, Borghetti M, Berbigier P, Berninger F, Delzon S, Grelle A, Hari P, Jarvis PG, Kolari P, Kowalski AS, Lankreijer H, Law BE, Lindroth A, Loustau D, Manca G, Moncrieff JB, Rayment M, Tedeschi V, Valentini R, Grace J (2007) The human footprint in the carbon cycle of temperate and boreal forests. Nature 447:848–850. doi:10.1038/nature05847
Melillo JM, Aber JD, Muratore JF (1982) Nitrogen and lignin control of hardwood leaf litter decomposition dynamics. Ecology 63:621–626. doi:10.2307/1936780
Mitchell H, Chandler R (1939) The nitrogen nutrition and growth of certain deciduous trees of northeastern United States. Black rock forest bulletin number 9
Nadelhoffer KJ, Emmett BA, Gundersen P, Kjonaas OJ, Koopmans CJ, Schleppi P, Tietema A, Wright RF (1999) Nitrogen deposition makes a minor contribution to carbon sequestration in temperate forests. Nature 398:145–148. doi:10.1038/18205
Ollinger SV, Aber JD, Lovett GM, Milham SE, Lathrop RG, Ellis JM (1993) A spatial model of atmospheric deposition for the northeastern United States. Ecol Appl 3:459–472. doi:10.2307/1941915
Pare D, Bernier B (1989) Origin of the phosphorus deficiency observed in declining sugar maple stands in the Quebec Appalachians. Can J For Res 19:24–34. doi:10.1139/x89-004
Pastor JD, Aber JD, McClaugherty CA, Melillo JM (1984) Aboveground production and N and P cycling along a nitrogen mineralization gradient on Blackhawk Island, Wisconsin. Ecology 65:256–268. doi:10.2307/1939478
Safford LO (1973) Fertilization increases diameter growth of birch–beech–maple trees in New Hampshire. USDA forest service research note NE-182
Schlesinger WH (1997) Biogeochemistry: an analysis of global change, 2nd edn. Academic Press, New York
Schoenau J, Karamanos R (1993) Sodium bicarbonte-extractable P, K, and N. In: Carter M (ed) Soil sampling and methods of analysis. Lewis Publishers, Boca Raton, pp 51–58
Siccama TG (1971) Presettlement and present forest vegetation in northern Vermont with special reference to Chittenden county. Am Midl Nat 85:153–172. doi:10.2307/2423919
Templer PH, Dawson TE (2004) Nitrogen uptake by four tree species of the Catskill Mountains, New York: Implications for forest N dynamics. Plant Soil 262:251–261. doi:10.1023/B:PLSO.0000037047.16616.98
Tiessen H, Moir JO (1993) Characterization of available P by sequential extraction. In: Carter M (ed) Soil Sampling and Methods of Analysis. Lewis Publishers, Boca Raton, pp 75–86
Underwood AJ (1997) Experiments in ecology: their logical design and interpretation using analysis of variance. Cambridge University Press, New York
van Breemen N, Finzi AC, Canham CD (1997) Canopy tree - soil interactions within temperate forests: effects of soil elemental composition and texture on species distributions. Can J For Res 27:1110–1116
Van Breemen N, Boyer EW, Goodale CL, Jaworski NA, Paustian K, Seitzinger SP, Lajtha K, Mayer B, Van Dam D, Howarth RW, Nadelhoffer KJ, Eve M, Billen G (2002) Where did all the nitrogen go? Fate of nitrogen inputs to large watersheds in the northeastern USA. Biogeochemistry 57:267–293
Verhoeven JTA, Koerselman W, Meuleman AFM (1996) Nitrogen- or phosphorus-limited growth in herbaceous, wet vegetation: relations with atmospheric inputs and management regimes. Trends Ecol Evol 11:494–497
Vitousek PM (1982) Nutrient cycling and nutrient use efficiency. Am Nat 119:553–572
Vitousek PM, Farrington H (1997) Nutrient limitation and soil development: Experimental test of a biogeochemical theory. Biogeochemistry 37:63–75
Walker TW, Syers JK (1976) The fate of phosphorus during pedogenesis. Geoderma 15:1–19
Ward WW, Bowersox TW (1970) Upland oak response to fertilization with nitrogen, phosphorus and calcium. For Sci 16:113–120
Wilmot TR, Ellsworth DS, Tyree MT (1995) Relationships among crown condition, growth, and stand nutrition in 7 northern Vermont sugarbushes. Can J For Res 25:386–397
Wilmot TR, Ellsworth DS, Tyree MT (1996) Base cation fertilization and liming effects on nutrition and growth of Vermont sugar maple stands. For Ecol Manag 84:123–134
Winer H (1955) History of the Great Mountain Forest, Litchfield County, Connecticut. Ph.D. Dissertation, Yale University, p 276
Yanai RD (1992) Phosphorus budget of a 70-Year-Old northern Hardwood forest. Biogeochemistry 17:1–22
Acknowledgments
Thanks the Great Mountain Forest Corporation and the State of Connecticut, Department of Environmental Protection, Natural Area Preserves Program for permission to work in their forestlands. Thanks also to Jody Bronson who helped with the logistical support for this research at the Great Mountain Forest. Edward Brzostek set up and fertilized the research plots and also made repeated dendrometer band measurements; his help was invaluable. Christy Goodale, Matthew Weand and Ruth Yanai provided helpful comments on an earlier draft of this manuscript. This research was supported by the USDA-CSREES (2001-00782).
Author information
Authors and Affiliations
Corresponding author
Appendix
Rights and permissions
About this article
Cite this article
Finzi, A.C. Decades of atmospheric deposition have not resulted in widespread phosphorus limitation or saturation of tree demand for nitrogen in southern New England. Biogeochemistry 92, 217–229 (2009). https://doi.org/10.1007/s10533-009-9286-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-009-9286-z