Abstract
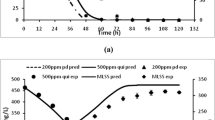
Biodegradation in the aquatic environment occurs in the presence of many chemicals, while standard simulation biodegradation tests are conducted with single chemicals. This study aimed to investigate the effect of the presence of additional chemicals on (1) biodegradation kinetics of individual chemicals and (2) the microbial composition in test systems. Parallel mixture and single substance experiments were conducted for 9 chemicals (phenethyl benzoate, oxacycloheptadec-10-en-2-one, α-ionone, methyl 2-naphthyl ether, decan-5-olide, octan-2-one, 2′-acetonaphthanone, methyl N-methylanthranilate, (+)-menthone) using inoculum from a Danish stream. Biotic and abiotic test systems were incubated at 12 °C for 1–30 days. Primary biodegradation kinetics were then determined from biotic/abiotic peak area ratios using SPME GC/MS analysis. The effect of the mixture on biodegradation varied with test chemical and was more pronounced for chemicals with lag-phases above 14 days: two chemicals degraded in the mixture but not when tested alone (i.e., positive mixture effect), and two degraded when tested alone but not in the mixture (i.e., negative mixture effect). Microbial composition (16S rRNA gene amplicon sequencing) was highly affected by 14 days incubation and the presence of the mixture (significant carbon source), but less by single chemicals (low carbon source). Growth on chemical mixtures resulted in consistent proliferation of Pseudomonas and Malikia, while specific chemicals increased the abundance of putative degraders belonging to Novosphingobium and Zoogloea. The chemical and microbiological results support (1) that simulation biodegradation kinetics should be determined in mixtures at low environmentally relevant concentrations and (2) that degradation times beyond some weeks are associated with more uncertainty.
Graphical abstract





Similar content being viewed by others
Data availability
All data generated during this study are included in this published article and its supplementary information files. Raw sequencing reads are available on GenBank (under BioProject PRJNA746252).
References
Albertsen M, Karst SM, Ziegler AS, Kirkegaard RH, Nielsen H (2015) Back to basics—the influence of DNA extraction and primer choice on phylogenetic analysis of activated sludge communities. PLoS ONE 10:e0132783. https://doi.org/10.1371/journal.pone.0132783
Apprill A, McNally S, Parsons R, Weber L (2015) Minor revision to V4 region SSU rRNA 806R gene primer greatly increases detection of SAR11 bacterioplankton. Aquat Microb Ecol 75:129–137. https://doi.org/10.3354/ame01753
Baboshin M, Golovleva L (2011) Multisubstrate kinetics of PAH mixture biodegradation: analysis in the double-logarithmic plot. Biodegradation 22:13–23. https://doi.org/10.1007/s10532-010-9370-z
Birch H, Andersen HR, Comber M, Mayer P (2017a) Biodegradation testing of chemicals with high Henry’s constants—separating mass and effective concentration reveals higher rate constants. Chemosphere 174:716–721. https://doi.org/10.1016/j.chemosphere.2017.02.003
Birch H, Hammershøj R, Comber M, Mayer P (2017b) Biodegradation of hydrocarbon mixtures in surface waters at environmentally relevant levels—effect of inoculum origin on kinetics and sequence of degradation. Chemosphere 184:400–407. https://doi.org/10.1016/j.chemosphere.2017.05.169
Birch H, Hammershøj R, Mayer P (2018) Determining biodegradation kinetics of hydrocarbons at low concentrations: covering 5 and 9 orders of magnitude of Kow and Kaw. Environ Sci Technol 52:2143–2151. https://doi.org/10.1021/acs.est.7b05624
Birch H, Sjøholm KK, Dechesne A, Sparham C, van Egmond R, Mayer P (2022) Biodegradation kinetics of fragrances, plasticizers, UV filters, and PAHs in a mixture—changing test concentrations over 5 orders of magnitude. Environ Sci Technol 56:293–301. https://doi.org/10.1021/acs.est.1c05583
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30:2114–2120. https://doi.org/10.1093/bioinformatics/btu170
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Pea AG, Goodrich JK, Gordon JI, Huttley GA, Kelley ST, Knights D, Koenig JE, Ley RE, Lozupone CA, McDonald D, Muegge BD, Pirrung M, Reeder J, Sevinsky JR, Turnbaugh PJ, Walters WA, Widmann J, Yatsunenko T, Zaneveld J, Knight R (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336
Desai AM, Autenrieth RL, Dimitriou-Christidis P, McDonald TJ (2008) Biodegradation kinetics of select polycyclic aromatic hydrocarbon (PAH) mixtures by Sphingomonas paucimobilis EPA505. Biodegradation 19:223–233. https://doi.org/10.1007/s10532-007-9129-3
Desiante WL, Minas NS, Fenner K (2021) Micropollutant biotransformation and bioaccumulation in natural stream biofilms. Water Res 193:116846. https://doi.org/10.1016/j.watres.2021.116846
Edgar RC (2013) UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods 10:996–998. https://doi.org/10.1038/nmeth.2604
Egli T (2010) How to live at very low substrate concentration. Water Res 44:4826–4837. https://doi.org/10.1016/j.watres.2010.07.023
Ellis TG, Smets BF, Grady CPL (1998) Effect of simultaneous biodegradation of multiple substrates on the extant biodegradation kinetics of individual substrates. Water Environ Res 70:27–38. https://doi.org/10.2175/106143098x126865
European Chemicals Agency (2016) Guidance on information requirements and chemical safety assessment Chapter R.16: Environ Exposure Assessment
European Chemicals Agency (2017) Guidance on information requirements and chemical safety assessment chapter R.7b: endpoint specific guidance. ECHA, European Chemicals Agency, Helsinki, Finland
European Parliament (2006) Regulation (EC) No 1907/2006 of the European Parliament and of the Council of 18 December 2006
Guha S, Peters CA, Jaffe PR (1999) Multisubstrate biodegradation kinetics of naphthalene, phenanthrene, and pyrene mixtures. Biotechnol Bioeng 65:491–499
Hammershøj R, Birch H, Redman AD, Mayer P (2019) Mixture effects on biodegradation kinetics of hydrocarbons in surface water: increasing concentrations inhibited degradation whereas multiple substrates did not. Environ Sci Technol 53:3087–3094. https://doi.org/10.1021/acs.est.9b00638
Hammershøj R, Birch H, Sjøholm KK, Mayer P (2020a) Accelerated passive dosing of hydrophobic complex mixtures—controlling the level and composition in aquatic tests. Environ Sci Technol 54:4974–4983. https://doi.org/10.1021/acs.est.9b06062
Hammershøj R, Sjøholm KK, Birch H, Brandt KK, Mayer P (2020b) Biodegradation kinetics testing of two hydrophobic UVCBs—potential for substrate toxicity supports testing at low concentrations. Environ Sci Process Impacts 22:2172–2180. https://doi.org/10.1039/d0em00288g
Helbling DE, Hammes F, Egli T, Kohler HPE (2014) Kinetics and yields of pesticide biodegradation at low substrate concentrations and under conditions restricting assimilable organic carbon. Appl Environ Microbiol 80:1306–1313. https://doi.org/10.1128/AEM.03622-13/SUPPL_FILE/ZAM999105124SO1.PDF
Knightes CD, Peters CA (2006) Multisubstrate biodegradation kinetics for binary and complex mixtures of polycyclic aromatic hydrocarbons. Environ Toxicol Chem 25:1746–1756. https://doi.org/10.1897/05-483R.1
Kowalczyk A, Martin TJ, Price OR, Snape JR, van Egmond RA, Finnegan CJ, Schäfer H, Davenport RJ, Bending GD (2015) Refinement of biodegradation tests methodologies and the proposed utility of new microbial ecology techniques. Ecotoxicol Environ Saf 111:9–22. https://doi.org/10.1016/j.ecoenv.2014.09.021
Legendre P, Gallagher ED (2001) Ecologically meaningful transformations for ordination of species data. Oecologia 129:271–280. https://doi.org/10.1007/s004420100716
Li Z, McLachlan MS (2019) Biodegradation of chemicals in unspiked surface waters downstream of wastewater treatment plants. Environ Sci Technol 53:1884–1892. https://doi.org/10.1021/acs.est.8b05191
Li P, Wang X, Stagnitti F, Li L, Gong Z, Zhang H, Xiong X, Austin C (2005) Degradation of phenanthrene and pyrene in soil slurry reactors with immobilized bacteria Zoogloea sp. Environ Eng Sci 22:390–399. https://doi.org/10.1089/EES.2005.22.390
Li Z, Sobek A, Radke M (2015) Flume experiments to investigate the environmental fate of pharmaceuticals and their transformation products in streams. Environ Sci Technol 49:6009–6017. https://doi.org/10.1021/acs.est.5b00273
Magoc T, Salzberg SL (2011) FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27:2957–2963. https://doi.org/10.1093/bioinformatics/btr507
Mansfeldt C, Deiner K, Mächler E, Fenner K, Eggen RIL, Stamm C, Schönenberger U, Walser JC, Altermatt F (2020) Microbial community shifts in streams receiving treated wastewater effluent. Sci Total Environ 709:135727. https://doi.org/10.1016/j.scitotenv.2019.135727
National Food Institute DTU (2015) Danish (Q)SAR Database. National Food Institute, Technical University of Denmark. http://qsardb.food.dtu.dk/database/index.html. Accessed 13 Apr 2021
Nogales J, García JL, Díaz E (2019) Degradation of aromatic compounds in Pseudomonas: a systems biology view. In: Rojo F (ed) Aerobic utilization of hydrocarbons, oils, and lipids. Springer, Cham, pp 639–687
OECD 301 (1992) OECD guideline for testing of chemicals. Ready Biodegrad 1–62
OECD 309 (2004) OECD guideline for the testing of chemicals. Aerobic mineralisation in surface water—simulation biodegradation test
Okpokwasili GC, Nweke CO (2006) Microbial growth and substrate utilization kinetics. Afr J Biotechnol 5:305–317. https://doi.org/10.5897/AJB2006.000-5041
Quast C, Pruesse E, Yilmaz P, Gerken J, Schweer T, Yarza P, Peplies J, Glöckner FO (2013) The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res 41:D590–D596. https://doi.org/10.1093/nar/gks1219
Reardon KF, Mosteller DC, Rogers JB, DuTeau NM, Kim KH (2002) Biodegradation kinetics of aromatic hydrocarbon mixtures by pure and mixed bacterial cultures. Environ Health Perspect 110:1005–1011
Révész F, Farkas M, Kriszt B, Szoboszlay S, Benedek T, Táncsics A (2020) Effect of oxygen limitation on the enrichment of bacteria degrading either benzene or toluene and the identification of Malikia spinosa (Comamonadaceae) as prominent aerobic benzene-, toluene-, and ethylbenzene-degrading bacterium: enrichment, isolation and whole-genome analysis. Environ Sci Pollut Res Int 27:31130–31142. https://doi.org/10.1007/S11356-020-09277-Z
Schmidt SK, Alexander M (1985) Effects of dissolved organic carbon and second substrates on the biodegradation of organic compounds at low concentrations. Appl Environ Microbiol 49:822–827. https://doi.org/10.1128/aem.49.4.822-827.1985
Schmidt SK, Simkins S, Alexander M (1985) Models for the kinetics of biodegradation of organic compounds not supporting growth. Appl Environ Microbiol 50:323–331. https://doi.org/10.1128/AEM.50.2.323-331.1985
Schwarzenbach RP, Gschwend PM, Imboden DM (2017) Environmental organic chemistry, 3rd edn. Wiley, Hoboken
Seller C, Honti M, Singer H, Fenner K (2020) Biotransformation of chemicals in water-sediment suspensions: influencing factors and implications for persistence assessment. Environ Sci Technol Lett 7:854–860. https://doi.org/10.1021/acs.estlett.0c00725
Sjøholm KK, Birch H, Hammershøj R, Saunders DMV, Dechesne A, Loibner AP, Mayer P (2021) Determining the temperature dependency of biodegradation kinetics for 34 hydrocarbons while avoiding chemical and microbial confounding factors. Environ Sci Technol 55:11091–11101. https://doi.org/10.1021/acs.est.1c02773
Sjøholm KK, Dechesne A, Lyon D, Saunders DMV, Birch H, Mayer P (2022) Linking biodegradation kinetics, microbial composition and test temperature—testing 40 petroleum hydrocarbons using inocula collected in winter and summer. Environ Sci Process Impacts 24:152–160. https://doi.org/10.1039/d1em00319d
Stravs MA, Pomati F, Hollender J (2019) Biodiversity drives micropollutant biotransformation in freshwater phytoplankton assemblages. Environ Sci Technol 53:4265–4273. https://doi.org/10.1021/acs.est.8b07018
Stringfellow WT, Aitken MD (1995) Competitive metabolism of naphthalene, methylnaphthalenes, and fluorene by phenanthrene-degrading pseudomonads. Appl Environ Microbiol 61:357–362. https://doi.org/10.1128/AEM.61.1.357-362.1995
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naïve Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73:5261–5267. https://doi.org/10.1128/AEM.00062-07
Wang J, Wang C, Li J, Bai P, Li Q, Shen M, Li R, Li T, Zhao J (2018) Comparative genomics of degradative novosphingobium strains with special reference to microcystin-degrading Novosphingobium sp. THN1. Front Microbiol 9:2238
Zou H, Radke M, Kierkegaard A, Macleod M, McLachlan MS (2015) Using chemical benchmarking to determine the persistence of chemicals in a Swedish Lake. Environ Sci Technol 49:1646–1653. https://doi.org/10.1021/es505548k
Acknowledgements
The authors thank Global Product Compliance (GPC) for financial support, Roger Van-Egmond for useful comments and input on the manuscripts and Hanne Bøggild for technical assistance in the laboratory.
Funding
This study was funded by the company Global Product Compliance (GPC). The role of the sponsor was to provide a list of chemicals of interest to their industries from which the authors selected test chemicals. The sponsor was not involved in the study design, collection, analysis, and interpretation of data or writing of manuscript.
Author information
Authors and Affiliations
Contributions
Study conception, funding and methodology was performed by HB and PM. HB performed the experiments, wrote the original draft for the manuscript, and prepared the figures. AD contributed to the data interpretation on microbial composition. All authors improved the manuscript through comments and text suggestions and all authors approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Authors are required to disclose financial interests that are directly or indirectly related to the work submitted for publication. The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Birch, H., Dechesne, A., Knudsmark Sjøholm, K. et al. Biodegradation of chemicals tested in mixtures and individually: mixture effects on biodegradation kinetics and microbial composition. Biodegradation 34, 139–153 (2023). https://doi.org/10.1007/s10532-022-10009-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-022-10009-y