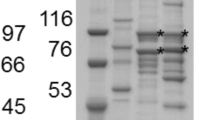
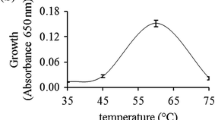
Abstract
The anaerobic thermophilic bacterium, Clostridium thermocellum, is a potent cellulolytic microorganism that produces large extracellular multienzyme complexes called cellulosomes. To isolate C. thermocellum organisms that possess effective cellulose-degrading ability, new thermophilic cellulolytic strains were screened from more than 800 samples obtained mainly from agriculture residues in Thailand using microcrystalline cellulose as a carbon source. A new strain, C. thermocellum S14, having high cellulose-degrading ability was isolated from bagasse paper sludge. Cellulosomes prepared from S14 demonstrated faster degradation of microcrystalline cellulose, and 3.4- and 5.6-fold greater Avicelase activity than those from C. thermocellum ATCC27405 and JW20 (ATCC31449), respectively. Scanning electron microscopic analysis showed that S14 had unique cell surface features with few protuberances in contrast to the type strains. In addition, the cellulosome of S14 was resistant to inhibition by cellobiose that is a major end product of cellulose hydrolysis. Saccharification tests conducted using rice straw soaked with sodium hydroxide indicated the cellulosome of S14 released approximately 1.5-fold more total sugars compared to that of ATCC27405. This newly isolated S14 strain has the potential as an enzyme resource for effective lignocellulose degradation.





Similar content being viewed by others
References
Bayer EA, Lamed R (1986) Ultrastructure of the cell surface cellulosome of Clostridium thermocellum and its interaction with cellulose. J Bacteriol 167:828–836
Bayer EA, Kenig R, Lamed R (1983) Adherence of Clostridium thermocellum to cellulose. J Bacteriol 156:818–827
Bayer EA, Shoham Y, Lamed R (2008) Cellulosome-enhanced conversion of biomass: on the road to bioethanol. In: Wall JD, Harwood CS, Demain AL (eds) Bioenergy. ASM Press, Washington, DC, pp 75–96
Béguin P, Millet J, Aubert JP (1992) Cellulose degradation by Clostridium thermocellum: from manure to molecular biology. FEMS Microbiol Lett 9:523–528
Bensadoun A, Weinstein D (1976) Assay of proteins in the presence of interfering materials. Anal Biochem 70:241–250
Brown SD, Raman B, McKeown CK, Kale SP, He Z, Mielenz JR (2007) Construction and evaluation of a Clostridium thermocellum ATCC 27405 whole-genome oligonucleotide microarray. Appl Biochem Biotechnol 137–140:663–674
Dashtban M, Schraft H, Qin W (2009) Fungal bioconversion of lignocellulosic residues; opportunities & perspectives. Int J Biol Sci 5:578–595
Demain AL, Newcomb M, David JH (2005) Cellulase, Clostridia, and Ethanol. Microbiol Mol Biol Rev 69:124–154
Freier D, Mothershed CP, Wiegel J (1988) Characterization of Clostridium thermocellum JW20. Appl Environ Microbiol 54:204–211
Fujino T, Béguin P, Aubert JP (1993) Organization of a Clostridium thermocellum gene cluster encoding the cellulosomal scaffolding protein CipA and a protein possibly involved in attachment of the cellulosome to the cell surface. J Bacteriol 175:1891–1899
Gerhardt P, Murray RGE, Costilow RN, Nester EW, Wood WA, Krieg NR, Phillips GB (1981) Manual of methods for general bacteriology. American Society of Microbiology, Washington, DC
Hungate RE (1969) A roll-tube method for cultivation of strict anaerobes. Methods Microbiol 3:117–132
Johnson EA, Reese T, Demain AL (1982a) Inhibition of Clostridium thermocellum cellulase by end products of cellulolysis. J Appl Biochem 4:64–71
Johnson EA, Sakajoh M, Halliwell G, Media A, Demain AL (1982b) Saccharification of complex cellulosic substrates by the cellulase system from Clostridium thermocellum. Appl Environ Microbiol 43:1125–1132
Lamed R, Setter E, Kenig R, Bayer EA (1983) The cellulosome-a discrete cell surface organelle of Clostridium thermocellum which exhibits separate antigenic, cellulose-binding and various cellulolytic activities. Biotechnol Bioeng Symp 13:163–181
Ljungdahl LG, Coughlan MP, Mayer F, Mori Y, Hon-nami H, Hon-nami K (1988) Macrocellulase complexes and yellow affinity substance from Clostridium thermocellum. In: Wood WA, Kellogg ST (eds) Methods in enzymology, vol 160. Academic Press, New York, pp 483–500
Lynd LR, Weimer PJ, van Zyl WH, Pretorius IS (2002) Microbial cellulose utilization: fundamentals and biotechnology. Microbiol Mol Biol Rev 66:506–577
Mayer F, Coughlan MP, Mori Y, Ljungdahl L (1987) Macromolecular organization of the cellulolytic enzyme complex of Closridium thermocellum as revealed by electron microscopy. Appl Eviron Microbiol 53:2785–2792
McCann MC, Carpita NC (2008) Designing the deconstruction of plant cell walls. Curr Opin Plant Biol 11:314–320
Morag E, Bayer EA, Lamed R (1992) Affinity digestion for the near-total recovery of purified cellulosome from Clostridium thermocellum. Enzyme Microb Technol 14:289–292
Mori Y (1992) Comparison of the Cellulolytic Systems of Clostridium thermocellum YM4 and JW20. Biotechnol Lett 14:131–136
Nelson N (1944) A photometric adaptation of the Somogyi method for the determination of glucose. J Biol Chem 153:375–380
Ng TK, Weimer PJ, Zeikus JG (1977) Cellulolytic and physiological properties of Clostridium thermocellum. Arch Microbiol 114:1–7
Ohmiya K, Sakka K, Kimura T, Morimoto K (2003) Application of microbial genes to recalcitrant biomass utilization and environmental conservation. J Biosci Bioeng 95:549–561
Ozkan M, Desai SG, Zhang Y, Stevenson DM, Beane J, White EA, Guerinot ML, Lynd LR (2001) Characterization of 13 newly isolated strains of anaerobic, cellulolytic, thermophilic bacteria. J Ind Microbiol Biotechnol 27:275–280
Raman B, Pan C, Hurst GB, Rodriguez M Jr, McKeown CK, Lankford PK, Samatova NF, Mielenz JR (2009) Impact of pretreated Switchgrass and biomass carbohydrates on Clostridium thermocellum ATCC 27405 cellulosome composition: a quantitative proteomic analysis. PLoS One 4:e5271
Schwarz WH (2001) The cellulosome and cellulose degradation by anaerobic bacteria. Appl Microbiol Biotechnol 56:634–649
Sukhumavasi J, Ohmiya K, Shimizu S, Ueno K (1988) Clostridium Jousi sp. nov., a cellulolytic, moderate thermophilic species from Thai compost. Int J Syst Bacteriol 38:179–182
Sun Y, Cheng J (2002) Hydrolysis of lignocellulosic materials for ethanol production: a review. Bioresour Technol 83:1–11
Viljoen JA, Fred EB, Peterson WH (1926) The fermentation of cellulose by thermophilic bacteria. J Agric Sci 16:1–17
Virunanon C, Chantaroopamai S, Denduangbaripant J, Chulalaksananukul W (2008) Solventogenic-cellulolytic Clostridia from 4-step-screening process in agricultural waste and cow intestinal tract. Anaerobe 14:109–117
Wood WA, Kellogg ST (1988) Methods in enzymology, vol 160. Academic Press, New York
Acknowledgments
C. Tachaapaikoon was supported by the Visiting Research Fellowship Program of Japan International Research Center for Agricultural Sciences (JIRCAS) and Thailand Research Fund (TRF Grant). This work was conducted as part of a research project funded by a grant (Development of Biomass Utilization Technologies for Revitalizing Rural Areas) from the Ministry of Agriculture, Forestry, and Fisheries of Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Chakrit Tachaapaikoon and Akihiko Kosugi contributed equally to this work.
Rights and permissions
About this article
Cite this article
Tachaapaikoon, C., Kosugi, A., Pason, P. et al. Isolation and characterization of a new cellulosome-producing Clostridium thermocellum strain. Biodegradation 23, 57–68 (2012). https://doi.org/10.1007/s10532-011-9486-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-011-9486-9