Abstract
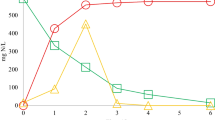
Tetrachloroethene (PCE) and trichloroethene (TCE) are common groundwater contaminants that also impact tidal flats, especially near urban and industrial areas. However, very little is known about dechlorinating microbial communities in tidal flats. Titanium pyrosequencing, 16S rRNA gene clone libraries, and dechlorinator-targeted quantitative real-time PCR (qPCR) characterized reductive dechlorinating activities and populations in tidal flat sediments collected from South Korea’s central west coast near Kangwha. In microcosms established with surface sediments, PCE dechlorination to TCE began within 10 days and 100% of the initial amount of PCE was converted to TCE after 37 days. cis-1,2-Dichloroethene (cis-DCE) was observed as dechlorination end product in microcosms containing sediments collected from deeper zones (i.e., 35–40 cm below ground surface). Pyrosequencing of bacterial 16S rRNA genes and 16S rRNA gene-targeted qPCR results revealed Desulfuromonas michiganensis-like populations predominanted in both TCE and cis-DCE producing microcosms. Other abundant groups included Desulfuromonas thiophila and Pelobacter acidigallici-like populations in the surface sediment microcosms, and Desulfovibrio dechloracetivorans and Fusibacter paucivorans-like populations in the deeper sediment microcosms. Dehalococcoides spp. populations were not detected in these sediments before and after incubation with PCE. The results suggest that tidal flats harbor novel, salt-tolerant dechlorinating populations and that titanium pyrosequencing provides more detailed insight into community structure dynamics of the dechlorinating microcosms than conventional 16S rRNA gene sequencing or fingerprinting methods.






Similar content being viewed by others
References
Abrahamsson K, Ekdahl JC, Pederson M (1995) Marine algae-a source of trichloroethylene and perchloroethylene. Limnol Oceanogr 40:1321–1326
Amos BK, Christ JA, Abriola LM, Pennell KD, Löffler FE (2007) Experimental evaluation and mathematical modeling of microbially enhanced tetrachloroethene (PCE) dissolution. Environ Sci Technol 41:963–970
Amos BK, Suchomel EJ, Pennell KD, Löffler FE (2009) Spatial and temporal distributions of Geobacter lovleyi and Dehalococcoides spp. during bioenganced PCE-NAPL dissolution. Environ Sci Technol 43:1977–1985
Andersson AF, Lindberg M, Jakobsson H, Bäckhed F, Nyrén P, Engstrand L (2008) Comparative analysis of human gut microbiota by barcoded pyrosequencing. PLoS One 3:e2836
Aulenta F, Beccari M, Majone M, Papini MP, Tandoi V (2008) Competition for H2 between sulfate reduction and dechlorination in butyrate-fed anaerobic cultures. Process Biochem 43:161–168
Bradley PM (2003) History and ecology of chloroethene biodegradation: a review. Bioremediat J 7:81–109
Cardenas E, Cole JR, Tiedje JM, Park J (2009) Microbial community analysis using RDP II (Ribosomal Database Project II): methods, tools and new advances. Environ Eng Res 14:3–9
Chang YC, Hatsu M, Jung K, Yoo YS, Takamizawa K (2000) Isolation and characterization of a tetrachloroethylene dechlorinating bacterium, Clostridium bifermentans DPH-1. J Biosci Bioeng 89:489–491
Cheng D, He J (2009) Isolation and characterization of “Dehalococcoides” sp. strain MB, which dechlorinates tetrachloroethene to trans-1,2-dechloroethene. Appl Environ Microbiol 75(18):5910–5918
Christ JA, Ramsburg CA, Abriola LM, Pennell KD, Löffler FE (2005) Coupling aggressive mass removal with microbial reductive dechlorination for remediation of DNAPL source zones: a review and assessment. Environ Health Perspect 113:465–477
Cole JR, Wang Q, Cardenas E, Fish J, Chai B, Farris RJ, Kulam-Syed-Mohideen AS, McGarrell DM, Marsh T, Garrity GM, Tiedje JM (2009) The Ribosomal Database Project: improved alignments and new tools for rRNA analysis. Nucleic Acids Res 37:D141–D145
Cupples AM, Spormann AM, McCarty PL (2003) Growth of a Dehalococcoides-like microorgasnism on vinyl chloride and cis-dichloroethene as electron acceptors as determined by competitive PCR. Appl Environ Microbiol 69:953–959
Cupples AM, Spormann AM, McCarty PL (2004) Comparative evaluation of chloroethene dechlorination to ethene by Dehalococcoides-like microorgasnisms. Environ Sci Technol 38:4768–4774
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32(5):1792–1797
Gribble GW (1994) The abundant natural sources and uses of chlorinated chemicals. Am J Public Health 84:1183
Gribble GW (2003) The diversity of naturally produced organohalogens. Chemosphere 52:289–297
Harms G, Layton AC, Dionisi HM, Gregory R, Garrett M, Hawkins SA, Robinson KG, Sayler GS (2003) Real-time PCR quantification of nitrifying bacteria in a municipal wastewater treatment plant. Environ Sci Technol 37:3430351
He J, Ritalahti KM, Aiello MR, Löffler FE (2003a) Complete detoxification of vinyl chloride by an anaerobic enrichment culture and identification of the reductively dechlorinating population as a Dehalococcoides species. Appl Environ Microbiol 69:996–1003
He J, Ritalahti KM, Yang K, Koenigsberg SS, Löffler FE (2003b) Detoxification of vinyl chloride to ethene coupled to growth of an anaerobic bacterium. Nature 424:62–65
Holliger C, Hahn D, Harmsen H, Ludwig W, Schumacher W, Tindall B, Vazquez F, Weiss N, Zehnder AJB (1998) Dehalobacter restrictus gen. nov. and sp. nov., a strictly anaerobic bacterium that reductively dechlorinates tetra- and trichloroethene in an anaerobic respiration. Arch Microbiol 169:313–321
Kassenga GR, Pardue JH (2006) Effect of competitive terminal electron acceptor processes on dechlorination of cis-1,2-dichloroethene and 1,2-dichloroethane in constructed wetland soils. FEMS Microbiol Ecol 57:311–323
Kim HS, Kweon JH (2010) Cleaning of lubricating products from machinery parts using subcritical water. KSCE J Civ Eng 14:1–6
Kittelmann S, Friedrich MW (2008) Novel uncultured Chloroflexi dechlorinate perchloroethene to trans-dichloroethene in tidal flat sediments. Environ Microbiol 10:1557–1570
Koh C (2001) The Korean tidal flat: environment, biology and human. Seoul National University Press, Seoul
Krumholz LR (1997) Desulfuromonas chloroethenica sp. nov. uses tetrachloroethylene and trichloroethylene as electron acceptors. Int J Syst Bacteriol 47:1262–1263
Krumholz LR, Sharp R, Fishbain SS (1996) A freshwater anaerobe coupling acetate oxidation to tetrachloroethylene dehalogenation. Appl Environ Microbiol 62:4108–4113
Lane DJ (1991) 16S/23S rRNA sequencing. Wiley, Chichester, pp 177–203
Löffler FE, Edwards EA (2006) Harnessing microbial activities for environmental cleanup. Curr Opin Biotechnol 17:274–284
Löffler FE, Sanford RA, Tiedje JM (1996) Initial characterization of a reductive dehalogenase from Desulfitobacterium chlororespirans Co23. Appl Environ Microbiol 62:3809–3813
Löffler FE, Ritalahti KM, Tiedje JM (1997) Dechlorination of chloroethenes is inhibited by 2-Bromoethanesulfonate in the absence of methanogens. Appl Environ Microbiol 63:4982–4985
Löffler FE, Sun Q, Li J, Tiedje M (2000) 16S rRNA gene-based detection of tetrachloroethene (PCE)-dechlorinating Desulfuromonas and Dehalococcoides species. Appl Environ Microbiol 66:1369–1374
Luijten MLGC, de Weert J, Smidt H, Boschker HTS, de Vos WM, Schraa G, Stams AJM (2003) Description of Sulfurospirillum halorespirans sp. nov., an anaerobic, tetrachloroethene-respiring bacterium, and transfer of Dehalospirillum multivorans to the genus Sulfurospirillum as Sulfurospirillum multivorans comb. nov. Int J Syst Evol Microbiol 53:787–793
Magnuson JK, Romine MF, Burris DR, Kingsley MT (2000) Trichloroethene reductive dehalogenase from Dehalococcoides ethenogenes: sequence of tceA and substrate range characterization. Appl Environ Microbiol 66:5141–5147
Maymo-Gatell X, Chien YT, Gossett JM, Zinder SH (1997) Isolation of a bacterium that reductively dechlorinates tetrachloroethene to ethene. Science 276:1568–1571
Maymo-Gatell X, Nijenhuis I, Zinder SH (2001) Reductive dechlorination of cis-1,2-dichloroethene and vinyl chloride by “Dehalococcoides ethenogenes”. Environ Sci Technol 35:516–521
Müller JA, Rosner BM, von Abendroth G, Meshluham-Simon G, McCarty PL, Spormann AM (2004) Molecular identification of the catabolic vinyl chloride reductase from Dehalococcoides sp. strain VS and its environmental distribution. Appl Environ Microbiol 70:4880–4888
Neumann A, Scholz-Muramatsu H, Diekert G (1994) Tetrachloroethene metabolism of Dehalospirillum multivorans. Arch Microbiol 162:295–301
Ravot G, Magot M, Fardeau ML, Patel BKC, Thomas P, Garcia JL, Ollivier B (1999) Fusibacter paucivorans gen. nov., sp. nov., an anaerobic, thiosulfate-reducing bacterium from an oil-producing well. Int J Syst Bacteriol 49:1141–1147
Scholz-Muramatsu H, Neumann A, Meβmer M, Moore E, Diekert G (1995) Isolation and characterization of Dehalospirillum multivorans gen. nov., sp. nov., a tetrachloroethene-utilizing, strictly anaerobic bacterium. Arch Microbiol 163:48–56
Sharma PK, McCarty PL (1996) Isolation and characterization of a facultative bacterium that reductively dehalogenates tetrachloroethene to cis-1,2-dichloroethene. Appl Environ Microbiol 62:761–765
Smidt H, de Vos WM (2004) Anaerobic microbial dehalogenation. Annu Rev Microbiol 58:43–73
Sun B, Cole JR, Sanford RA, Tiedje JM (2000) Isolation and characterization of Desulfovibrio dechloracetivorans sp. nov., a marine dechlorinating bacterium growing by coupling the oxidation of acetate to the reductive dechlorination of 2-Chlorophenol. Appl Environ Microbiol 66:2408–2413
Sung Y, Ritalahti KM, Sanford RA, Urbance JW, Flynn SJ, Tiedje JM, Löffler FE (2003) Characterization of two tetrachloroethene-reducing, acetate-oxidizing anaerobic bacteria, and their description as Desulfuromonas michiganensis sp. nov. Appl Environ Microbiol 69:2964–2974
Sung Y, Fletcher KE, Ritalahti KM, Apkarian RP, Ramos-Hernández N, Sanford RA, Mesbah NM, Löffler FM (2006a) Geobacter lovleyi sp. nov. strain SZ, a novel metal-reducing and tetrachloroethene-dechlorinating bacterium. Appl Environ Microbiol 72:2775–2782
Sung Y, Ritalahti KM, Apkarian RP, Löffler FE (2006b) Quantitative PCR confirms purity of strain GT, a novel trichloroethene-to-ethene-respiring Dehalococcoides isolate. Appl Environ Microbiol 72:1980–1987
Suyama A, Iwakiri R, Kai K, Tokunaga T, Sera N, Furukawa K (2001) Isolation and characterization of Desulfitobacterium sp. strain Y51 capable of efficient dehalogenation of tetrachloroethene and polychloroethanes. Biosci Biotechnol Biochem 65:1474–1481
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
US Environmental Protection Agency (2006) Superfund information system, National Priorities List (NPL) sites. http://cfpub.epa.gov/supercpad/cursites/srchsites.cfm
Wilms R, Sass H, Köpke B, Köster J, Cypionka H, Engelen B (2006) Specific bacterial, archaeal, and eukaryotic communities in tidal-flat sediments along a vertical profile of several meters. Appl Environ Microbiol 72:2756–2764
Zinder SH, Gossett JM (1995) Reductive dechlorination of tetrachloroethene by a high rate anaerobic microbial consortium. Environ Health Perspect 103:5–7
Acknowledgments
We gratefully acknowledge Dr. James M. Tiedje from Center for Microbial Ecology at Michigan State University for his valuable review and advice. This study was supported by Korea Ministry of Environment as “The Eco-technopia 21 Project” (051-071-031) and WCU (World Class University) program through the National Research Foundation of Korea funded by the Ministry of Education, Science and Technology (R33-10076).
Author information
Authors and Affiliations
Corresponding author
Additional information
Part of SI BioMicroWorld2009.
Rights and permissions
About this article
Cite this article
Lee, J., Lee, T.K., Löffler, F.E. et al. Characterization of microbial community structure and population dynamics of tetrachloroethene-dechlorinating tidal mudflat communities. Biodegradation 22, 687–698 (2011). https://doi.org/10.1007/s10532-010-9429-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-010-9429-x