Abstract
Cryptic ecologies, the Wallacean Shortfall of undocumented species’ geographical ranges and the Linnaean Shortfall of undescribed diversity, are all major barriers to conservation assessment. When these factors overlap with drivers of extinction risk, such as insular distributions, the number of threatened species in a region or clade may be underestimated, a situation we term ‘cryptic extinction risk’. The genus Lepidodactylus is a diverse radiation of insular and arboreal geckos that occurs across the western Pacific. Previous work on Lepidodactylus showed evidence of evolutionary displacement around continental fringes, suggesting an inherent vulnerability to extinction from factors such as competition and predation. We sought to (1) comprehensively review status and threats, (2) estimate the number of undescribed species, and (3) estimate extinction risk in data deficient and candidate species, in Lepidodactylus. From our updated IUCN Red List assessment, 60% of the 58 recognized species are threatened (n = 15) or Data Deficient (n = 21), which is higher than reported for most other lizard groups. Species from the smaller and isolated Pacific islands are of greatest conservation concern, with most either threatened or Data Deficient, and all particularly vulnerable to invasive species. We estimated 32 undescribed candidate species and linear modelling predicted that an additional 18 species, among these and the data deficient species, are threatened with extinction. Focusing efforts to resolve the taxonomy and conservation status of key taxa, especially on small islands in the Pacific, is a high priority for conserving this remarkably diverse, yet poorly understood, lizard fauna. Our data highlight how cryptic ecologies and cryptic diversity combine and lead to significant underestimation of extinction risk.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Scientists and conservationists have invested substantial effort assessing the status of significant portions of the world’s biodiversity (Rodrigues et al. 2006). Species-level conservation assessment is a first principle of all conservation planning, prioritization, and investment. For vertebrate taxa (which are generally better known) three key major impediments to determining conservation status and identifying declines have emerged. First, for rare species or those with cryptic ecologies (i.e. difficult-to-access microhabitats), sampling is frequently difficult, resulting in a lack of data necessary for conservation assessments, even under basic criteria such as extent of occurrence (McDonald 2004; Gillespie et al. 2020). For these difficult-to-sample species, population monitoring can be uninformative or prohibitively expensive, leaving conservation practitioners unable to measure population trends or the effectiveness of their management activities (Chadès et al. 2008). Second, the Wallacean Shortfall refers to the lack information on species’ distributions (Lomolino 2004), which impedes area-based assessment of conservation status. Third, is the Linnaean Shortfall (Brown and Lomolino 1998) or taxonomic impediment; that is, a large proportion of species diversity remains scientifically undocumented, with estimates ranging from 5 to 80 million unnamed species (NSB 1989). Listing of undescribed species on the International Union for Conservation of Nature (IUCN) Red List is discouraged (IUCN 2021) and knowledge of the distribution (i.e. the Wallacean Shortfall) and habitat requirements of candidate species (undescribed but recognized taxa) is frequently especially scant. Where threatened clades contain many difficult-to-sample, poorly known and/or undescribed species, there is a higher probablity that extinction risk will be underestimated, a situation that we term cryptic extinction risk (CER).
Although CER is, by its nature, hard to document, focused analyses of clades or regions where Linnaean Shortfall and/or cryptic ecologies are likely to operate provide opportunities to predict and highlight species groups and areas wherein it might be a major issue. For example, global-scale analyses have identified correlates of elevated extinction risk such as distribution size, insularity, body size, and ecology (Böhm et al. 2016; Ripple et al. 2017). Some studies have used these correlates to estimate proportions of data deficient species that are likely to be threatened (Tingley et al. 2013; Bland and Böhm 2016). Other expert panel-based analyses have also attempted to incorporate undescribed taxa into conservation prioritization exercises (Lintermans et al. 2020), thereby highlighting significant proportions of unnamed but threatened taxa. Across all analyses, small, and especially insular, distributions consistently emerge as key predictors of conservation concern. Indeed, insular areas such as the Pacific islands, Madagascar, and New Zealand have some of the most threatened biotas in the world (Carlquist 1974; Cheke and Hume 2008; Chapple et al. 2021). Accordingly, for taxa wherein insular distributions intersect with high levels of undescribed diversity and cryptic ecologies, CER may be acute.
The genus Lepidodactylus is a radiation of small (generally less the 10 cm adult body length), arboreal geckos with distributions spanning the tropical western Pacific (Figs. 1, 2; Oliver et al. 2018a). Recent phylogenetic analyses indicate that species currently placed in the genus Lepidodactylus are paraphyletic with respect to two lineages in the subgenus Luperosaurus, plus the entire genus Pseudogekko (Oliver et al. 2018a; Wood et al. 2020). Hereafter we use Lepidodactylus as a catchall for the combined clade of these lineages, comprising 58 described species currently, with phylogenetic analyses supporting a large number of additional, undescribed taxa (Oliver et al. 2018a; Eliades et al. 2021). In recent years, a suite of poorly known and/or highly restricted new Lepidodactylus species have also been described (Kraus 2019; Brown et al. 2020; Karkkainen et al. 2020; Eliades et al. 2021). A considerable, but as yet undocumented, number of described and candidate Lepidodactylus species are known only from small islands and atolls (e.g., Zug et al. 2003; Stubbs et al. 2017; Karin et al. 2018). Prior work on the Lepidodactylus also suggests that, like some other diverse insular lineages (Fernández‐Palacios et al. 2021; Richmond et al. 2021), this clade shows evidence of evolutionary displacement, with taxa concentrated away from species-rich lowland rainforests and into ‘marginal’ open, coastal or montane habitats, especially on the fringes of continental areas and larger islands like New Guinea and Borneo (Oliver et al. 2018a, 2020).
Examples of Lepidodactylus: a the newly described Lepidodactylus bisakol from the southern Bicol Peninsula, Philippines (Jason Fernandez and Rafe Brown); b the newly described Pseudogekko hungkag from Luzon Island, Philippines (Jason Fernandez and Rafe Brown); c an undescribed Lepidodactylus species from the Bismark Islands, Papua New Guinea (Steven Richards); and d Lepidodactylus flaviocularis known from only two specimens at one location on Guadalcanal, Solomon Islands (Scott Travers)
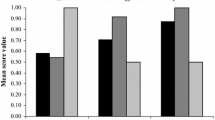
a Distribution of described Lepidodactylus species (black dots represent location records; www.gbif.org) across regions of the tropical western Pacific (excluding the widespread anthropogenically dispersed species L. lugubris). b Status of described Lepidodactylus species (n = 58) by region (DD data deficient, LC least concern, NT near threatened, VU vulnerable, EN endangered, CR critically endangered, EX Extinct)
In addition to a pattern of evolutionary displacement around continental fringes, Lepidodactylus are secretive and for most species are rarely observed in the wild. As such, they are often the most poorly known reptile species where they occur (Brown and Acalala 1978; McCoy 2006). For example, there may be decades between species sightings despite substantial search effort in suitable habitat (Wiles and Conry 1990; Crombie and Menz 2007; Meneses et al. 2020), or observations may be infrequent especially at localities where other sympatric geckos are abundant (Bucol et al. 2012). Further highlighting their apparent rarity, several recent Lepidodactylus species descriptions are based on only one or two specimens collected from a single locality (Siler et al. 2014; Kraus 2019; Karkkainen et al. 2020; Eliades et al. 2021). Lepidodactylus species are also known from hard-to-access microhabitats, exemplified by the use of small hollow chambers within epiphytic ant plants and arboreal termitaria (Brown and Alcalala 1978; Ineich 2008, 2019; Oliver et al. 2015; Brown et al. 2020). The combination of distributional patterns (e.g., small islands, fringe habitats, and mountains), rarity, and microhabitat use, suggests an inherent vulnerability of Lepidodactylus species to major habitat disturbance events and ecological displacement from invasive species on contemporary ecological scales. Highlighting this vulnerability was the recent extinction from the wild of the Christmas Island endemic Lepidodactylus listeri, linked to predation by the introduced snake Lycodon capucinus and other invasive species (Emery et al. 2021). Christmas Island is an Indian Ocean Australian territory with well-resourced conservation management and locally based biologists. In contrast, most other Lepidodactylus occur in rarely monitored areas, suggesting that there is a high likelihood that the threat to this clade is overlooked or ‘cryptic’ and that other declines and extinction events may be occurring presently but, to date, remain undocumented.
Here we use Lepidodactylus as a model group for assessing approaches to estimate and highlight CER, herein broadly defined as underestimated or overlooked extinction risk within specific taxonomic groups and/or particular regions. We assembled an expert panel to: (a) update our knowledge of distributions, threats and status, (b) estimate the number of undescribed species, and (c) arrive at a final characterization of CER for species of Lepidodactylus. In the context of evolutionary and ecological displacement (Oliver et al. 2018a), we predicted that, compared to most other lizard groups, a greater proportion of Lepidodactylus species would be: (1) Data Deficient, (2) highly threatened by invasive species, (3) ecologically rare, (4) range restricted, and (5) ultimately, threatened with extinction.
Methods
IUCN red list assessment
We included all recognized species of Lepidodactylus and Pseudogekko in our assessment. For Luperosaurus species, we excluded taxa (L. gulat, L. browni, and L. iskandari) recently shown to be part of a separate radiation: the genus Gekko (Wood et al. 2020). However, we conservatively included all species of true Luperosaurus (members of the clade containing the type species of the genus: L. cumingii), even if their phylogenetic relationships remain untested. There were 58 recognized species in our focal group at the time we started this assessment (December 2020), of which 36 species had previously been assessed under IUCN Red List criteria (15 Least Concern (LC), 14 Data Deficient (DD), three Vulnerable (VU), two Endangered (EN), one Critically Endangered (CR), and one Extinct in the wild (EX)). Most had not been assessed since 2011 or earlier. Twelve of the recognized species have been described in the last decade, and none of these had been assessed.
We conducted a two-day virtual workshop to assess the extinction risk of all 58 described Lepidodactylus species against IUCN Red List criteria. The workshop was attended by facilitators from IUCN and Secretariat of the Pacific Regional Environment Programme (SPREP) and a panel of 11 experts (RB, FK, UA, SE, RF, MG, BK, CG, MS, CS, PO) with extensive knowledge of, and field experience with, the species assessed. Input from three non-attending experts (LG, II, SR) was subsequently incorporated as well. Prior to the workshop, the IUCN facilitator (PB) collated data reported for each species from all available literature (e.g., geographic range, population abundance, ecology, threats, and conservation measures), and entered these into the IUCN’s Species Information Service (SIS) database. Data were reviewed systematically (species-by-species) by the workshop panel, and augmented with input from all panellists, to arrive at final consensus, resulting in revised conservation status.
To broadly assess the degree to which Lepidodactylus may be less known and/or more threatened than other reptile taxa, we compared the percentages of threatened and data deficient species to similar studies, which have synthetically revised the conservation status of groups of reptiles (Böhm et al. 2013; Tingley et al. 2019; Chapple et al. 2021).
Cryptic extinction risk
We compiled both published and unpublished genetic and morphological data to estimate the number of undescribed species of Lepidodactylus. Candidate species were included if they met one or more of the following criteria: (a) mitochondrial data indicating divergences (uncorrected p-distances) ≥ 10% (Oliver et al. 2009); (b) examination of relevant specimens, resulting in the combination of unique and diagnostic character sets (traditional categorical character state differences; or, in the case of measurements or meristic data, non-overlapping ranges of character values); or (c) unambiguous geographic disjunction from allied taxa coupled with photographic evidence of morphological distinctiveness. Descriptions of a number of these candidate species were in preparation at the time of our assessment (December 2020), and those taxa were still considered candidate species for this paper. We also emphasize that this list may be conservative. For example, there were no genetic or morphological data available for the widespread Fijian species Lepidodactylus manni; hence, this was considered as only one taxon (when we suspect it might constitute several species). Further, given the extreme rarity of many species (often known from a single specimen or locality), it seems likely that additional taxa remain completely overlooked. Conversely, we also emphasize that, for many of these candidate taxa, additional data will be required to validate the hypotheses that they represent distinct species. We are also aware of some recognised taxa that show very limited morphological or genetic divergence from other taxa, and may be synonyms (e.g., Lepidodactylus browni and Lepidodactylus orientalis or Lepidodactylus pantai and Lepidodactylus woodfordi; PMO pers. obs., Karin et al. 2021).
In addition to the data collected as part of our workshop assessment, we compiled a table of conservation-related attributes for all described and candidate species within Lepidodactylus, including: region (Sunda Shelf, including Borneo and Peninsular Malaysia; Philippines; Wallacea (based on phylogenetic evidence Christmas Island was considered part of Wallacea; Oliver et al. 2018a); West Melanesia, including Papua New Guinea (excluding Bougainville) and the Indonesian provinces of Papua and West Papua; and Pacific Islands, including Micronesia, Solomon Islands (including Bougainville), Vanuatu, Fiji, and Polynesia), extent of occurrence (area of minimum convex polygon around all location records or total land area where known only from an island < 1000 km2), number of specimens and sightings (from the GBIF and VertNet online databases and unpublished records from the panel), number of locations (minimum distance of 10 km separating locations, cf. Meiri et al. 2018), whether the taxon is restricted to small islands < 1000 km2, and year of last record. Consistent with Oliver et al. (2018a), we also recorded whether each species was known only from forest habitats (lowland rainforest, coastal forest, montane forest) versus also or only occurring in open habitats (e.g. beaches, disturbed anthropogenic landscapes or savannas), and whether the species is only known from lowland rainforest, a habitat type from which these geckos may be ecologically excluded in some areas (Oliver et al. 2018a, b).
Data analysis
We used binary logistic regression models to assess the drivers of DD and threatened status in described Lepidodactylus. For the DD models, we assigned species as DD (1), or data sufficient (0) if LC, Near Threatened (NT), VU, EN, or EX. For the threatened species models, we assigned species as threatened (1) if VU, EN, CR or EX, or non-threatened (0) if NT or LC, and removed DD species. We selected covariates that were available for all taxa (including DD and candidate species), including region (categorical), number of locations (continuous; defined above), small-island distributions (categorical), year of last record (continuous), obligate forest dweller (categorical; defined above), and obligate lowland rainforest dweller (categorical; defined above). We were unable to include additional potentially useful covariates, such as phylogeny, as these data were unavailable for all taxa. We fitted individual covariate models in R (R Core Team 2020) and retained covariates that significantly predicted threatened status (p = < 0.05). To avoid overfitting in the smaller data sufficient species pool, regions were fitted as individual covariates (e.g., Pacific Island region (1) versus other (0)). We modelled the retained covariates singularly and in all possible pairwise combinations, selecting the best-supported model as the highest R2 with a difference in Akaike information criterion (AIC) from the best-ranked model of < 2.0 (Symonds and Moussalli 2011).
To predict the numbers of DD and candidate species that are threatened, we applied the best-supported threatened logistic regression model using the predict () function in R, assigning species with a probability > 0.5 as potentially threatened.
Results
IUCN red list assessment
We found twenty-one (36%) of the described Lepidodactylus species to be DD, with insufficient data preventing adequate assessment of conservation status. This percentage is higher than the global percentages of DD reptiles reported at the Order level (0–24%, except for Amphisbaenia) and realm (0–33%) (Böhm et al. 2013), higher than any of the world’s skink subfamilies (3–15%) (Chapple et al. 2021), higher than any reptile family in Australia (0–18%) (Tingley et al. 2019), and higher than 25 of 26 reptile Families in Tanzania (0–27%). West Melanesia had the highest number of DD species (n = 6) (Fig. 2). Region was the best-supported logistic regression model predicting DD status (Table 1), with DD species more likely to occur on Sunda Shelf than the Philippines or Pacific Islands (Fig. 2).
We found fifteen (41%, calculated as (VU + EN + CR + EX)/(Total N-DD)) described Lepidodactylus species were threatened (VU, EN, CR) or extinct (EX). This percentage is higher than the global percentage of threatened reptiles reported for all reptile orders (7–21%) and all biogeographic realms (12–25%) except Oceania (42.9%) (Böhm et al. 2013), higher than any of the world’s skink subfamilies (0–30%) (Chapple et al. 2021), and higher than any reptile family in Australia (0–17%) (Tingley et al. 2019). The Pacific Islands region had the most threatened species, including five Critically Endangered, and two Vulnerable species (Fig. 2). The Philippines had the next highest number of threatened species, with two endemic taxa categorized as Endangered and three assessed as Vulnerable (Fig. 2). Number of locations and small-island endemism were included in the best-support logistic regression model to predict threat status (Table 1). Threatened species were recorded from significantly fewer locations than non-threatened species and were more likely to occur on small islands < 1000 km2 in area. There has been one documented Lepidodactylus extinction, with the Christmas Island endemic Lepidodactylus listeri last recorded in the wild in 2012.
Of the 58 described Lepidodactylus species, 33% (n = 19) are known only from their type localities. This is a higher percentage than previously documented in each of the world’s six gecko families (8–23%) and higher than in all but two of the world’s 42 lizard families (Meiri et al. 2018). Further, fewer than 50% of Lepidodactylus species are known from more than two locations. There was no significant difference in average extent of occurrence (EOO) across our five regions (Kruskal–Wallis chi squared = 3.71, df = 3, p = 0.30) for the 53 species with these data available. Ten (17%) of the described Lepidodactylus species have not been recorded in the last 20 years, including (date of last record shown in parentheses for each species): L. dialeukos (1938), L. euaensis (1992), L. gardineri (1982), L. labialis (1971), L. mutahi (1966), L. oortii (1923), L. shebae (1944), L. tepukapili (1998), L. zweifeli (1969), and Luperosaurus yasumai (1994).
The main listed threats to Lepidodactylus are not evenly distributed across the regions (Fig. 3). Specifically, deforestation, and agriculture were the dominant threats in the two western regions (Sunda Shelf and the Philippines) and in West Melanesia, whereas invasive geckos and predators were dominant threats east of Wallace’s Line (Fig. 3).
Distribution of key threats nominated to described Lepidodactylus species across regions of the tropical western Pacific, including numbers of species affected by each threat. Note that not all species have listed threats and some species have multiple threats, thus total numbers across threat categories will not match the number of species in each region
Cryptic extinction risk
Our panel identified 32 candidate Lepidodactylus species in addition to the 58 currently described, which is 35% of known species diversity in this clade. Candidate species were non-randomly distributed, with the highest numbers in West Melanesia (n = 13) and the Philippines (n = 10) (Fig. 4). West Melanesia and Wallacea each had more candidate species than described species (Fig. 4). The Pacific Islands had the lowest number of candidates relative to described species (3 versus 16) (Fig. 4.). Twenty of these 32 candidate species have been included in a published clade-wide phylogeny (Oliver et al. 2018a).
Half (n = 16) of these candidate species were only known from islands of < 1000 km2, and 78% (n = 25) are known from only one location. Candidate species were also typically rare despite substantial search effort involving many person hours or search nights per specimen (e.g. Eliades et al. 2021). Most of the candidate species have been collected on relatively recent expeditions, with 84% (n = 27) first collected post-2000 and 63% (n = 20) collected post-2010 (Table S1), and 78% (n = 25) of candidate species have already been referred to in the published literature.
Our threatened status logistic regression model predicted an additional three and 15 threatened species from the DD and candidate species pools, respectively (Table 2). Eight of the taxa predicted to be threatened are from West Melanesia, four are from the Philippines, and three are from each of Wallacea and the Pacific Islands (Table S1). Priority Lepidodactylus species for conservation and field surveys comprise 36.7% of all species in the clade (Tables 2, S1).
Discussion
Lepidodactylus geckos are relatively highly threatened, both in terms of the number and percentage of taxa of concern; importantly, the number of threatened species was underestimated prior to this study. Our results demonstrate the value of combining standard, predominately range-based IUCN Red List assessments of described taxa with explicit consideration of what we term cryptic extinction risk. Specifically, although our updated conservation assessment of recognised species more than doubled the number of threatened species (from six to 15), this estimate was again more than doubled (to 33) through our model predictions of threatened status in Data Deficient and undescribed candidate species. Considering CER in conservation assessment could benefit other inherently vulnerable biotic groups with high levels of documented, but undescribed, taxonomic diversity.
Cryptic extinction risk
Our work provides a demonstration of the extent to which the Linnean Shortfall and other knowledge gaps (e.g. Wallacean Shortfall) have led to underestimation of extinction risk in an exemplary clade of poorly known species, typified by cryptic ecology and underestimated species diversity. Although Lepidodactylus species are relatively small lizards and often an inconspicuous part of many lizard communities across our study region (Brown et al. 2013; Sanguila et al. 2016), our data indicate that, with close to one hundred species, Lepidodactylus is the most diverse gecko radiation across the Philippines and much of the Pacific. Our panel also noted that many Lepidodactylus are exceedingly rare (e.g., Lepidodactylus flaviocularis, Luperosaurus spp. on large islands; Oliver et al. 2020; Eliades et al. 2021), especially on larger islands where there are richer gecko assemblages and on small islands, in the presence of invasive geckos (e.g., Hemidactylus spp.). This scarcity likely reflects genuine rarity or species’ reliance on difficult-to-access microhabitats (Grismer 2011; Oliver et al. 2015; Brown et al. 2020; Eliades et al. 2021). Either possibility could also be linked to ecological displacement and/or susceptibility to predation. A tendency for Lepidodactylus species to be ecologically displaced is consistent with broader and deeper patterns of evolutionary displacement around continental fringes (Oliver et al. 2018a, b, 2020). We argue that this combination of cryptic ecology, ecological vulnerability, and high levels of unrecognised species diversity has led to a serious and likely ongoing underestimation of both the number and percentage of Lepidodactylus species that are threatened—and potentially already extinct (see below).
Our approach to estimating threatened status differs from other assessment methods of particular biotic groups, such as expert elicitation of extinction percentage risk (Lintermans et al. 2020; Geyle et al. 2021), in that potential at-risk taxa can be identified from single species’ geographical occurences and few other data. Our approach is also distinct from other methods seeking to overcome the taxonomic impediment, such as mapping centers of phylogenetic endemism (Rosauer et al. 2018), because data deficient and undescribed candidate species from small islands can be identified as priorities for conservation management without any phylogenetic data, or overlap with significant hotspots of diversity or distinctiveness. Consequently, our method of assessing CER may be most appropriate for insular species groups that combine traits such as documented but undescribed diversity, many localized endemics, and often highly cryptic ecologies. An obvious further candidate group would be the other major Pacific lizard radiation of skinks in the genus Emoia. It may be also applicable to some invertebrate groups such as mygalomorph spiders wherein described diversity has been documented and many taxa show localised ranges (e.g. Rix et al. 2020).
Spatial patterns of extinction risk and threats
We found that extinction risk in Lepidodactylus was not randomly distributed but concentrated in small-island systems, especially the Pacific Islands region and, to a lesser extent, West Melanesia and Wallacea. This geographic variation in vulnerability is consistent with research on Pacific island birds demonstrating frequent losses of more than 50% of bird species on islands throughout the western Pacific following the arrival of humans (Steadman 2006). Some islands of Wallacea also show evidence of Holocene extinction and turnover in key faunal elements (Turvey et al. 2017; Louys et al. 2021). Although fossil remains of smaller lizards are uncommon on these islands, prehistoric extinctions of unassignable gecko taxa have been documented in Fiji, Tonga and the Mariana Islands (Pregill 1993, 1998; Pregill and Steadman 2014). More recently, extirpations of several small gecko species have been recorded throughout the tropical northwest Pacific (Pregill and Steadman 2009). These observations, together with the recent extinction of Lepidodactylus listeri from Christmas Island (Andrew et al. 2018), the highly isolated and localized distributions of many taxa (e.g., Lepidodactylus paurolepis from Palau and L. oligoporus from the Mortlock Islands), and the number of undescribed species from poorly sampled locations (Eliades et al. 2021), point to the loss of a much richer evolutionary history than has been documented (Oliver et al. 2018a), and a likelihood of future extinction events in these regions. Even during this work we gathered reports that the Rotuma Island endemic (Lepidodactylus gardneri) has not been recently seen despite active searching (Monifa Fiu, pers. obs.), raising grave concerns about the persistence of this small island endemic.
As with extinction risk, we also found that threats to Lepidodactylus species were not randomly distributed. A dominant role of invasive species was identified east of Wallace’s Line and especially in the Pacific Islands, where many small-island endemic Lepidodactylus species occur. The importance of invasive species is consistent with the idea that Lepidodactylus are particularly susceptible to novel predators and ecological displacement, and supported by the likely role of an introduced snake (Lycodon capucinus) in the recent extinction of Lepidodactylus listeri (Emery et al. 2021). In this context, the human-assisted expansion of L. capucinus east of Wallace’s Line, through the Lesser Sundas and into islands around New Guinea (O’Shea et al. 2018) is particularly concerning. In contrast, in more western regions and on larger landmasses, habitat loss through deforestation was the major threat to the many obligate forest-dwelling Lepidodactylus species (e.g., Das et al. 2008; Siler et al. 2014; Kraus and Oliver 2019; Brown et al. 2020; Eliades et al. 2021), a pattern consistent with global trends for small vertebrates (Ripple et al. 2017).
Steps towards understanding and conserving biodiversity in rare and undescribed taxa
The true conservation status of a significant majority (59%) of Lepidodactylus taxa (candidate and Data Deficient species) remains unknown. Many species are very difficult to find, let alone effectively monitor (Brown and Alcala 1978). Thus, although we encourage targeted searches, novel sampling methods (e.g., Bell 2009), and efforts to identify key microhabitats (Brown et al. 2013; Sanguila et al. 2016), reward per unit effort is likely to be low, especially for rare species from larger island systems (e.g., Sunda Shelf and the Philippines) (Eliades et al. 2021). Many key areas that need to be surveyed are also both logistically and politically challenging to access (e.g., Luperosaurus joloensis, known only from four specimens, collected from three locality es in the Sulu Archipelago and Western Mindanao Island; Supsup et al. 2020). Given these limitations, the following research and conservation actions may further knowledge of Lepidodactylus diversity and enhance species conservation:
-
1.
Integrative and innovative approaches to resolving species diversity. This is particularly important for an array of remote island taxa from the Pacific. Relatively new techniques for systematics such as target capture genomic studies for obtaining DNA from old specimens with degraded DNA and CT scanning may provide avenues to resolve species status for taxa known from few or very old specimens. Although it is preferable to describe species based on a robust, statistical sample size and vouchered series of genotyped animals, the extreme rarity of many Lepidodactylus species suggests this will often not be possible, and taxonomic studies should proceed, providing there is no doubt of evolutionary distinctiveness. A complete phylogentic picture for the clade would also allow for phylogeny to be included in models predicting threatened or DD status, perhaps improving the accuracy of predictions.
-
2.
Targeted surveys. Although Lepidodactylus species are extremely difficult to detect in many ecological systems, targeteted surveys on remote islands or in habitats that may serve as refuges for vulnerable species could be informative (Table S1). Numerous small-island endemics from the Pacific urgently require re-survey to confirm that they have not been extirpated (e.g., Lepidodactylus gardineri, L. oligoporus, L. tepukapili, and L. sp. Nuguria). We again note that recent biodiversity surveys have failed to locate L. gardineri in Rotuma (Monifa Fiu, pers. comm.). Several species, known only from small islands, are also surprisingly abundant on these islands (e.g., Lepidodactylus mitchelli on Boia Boia Waga Island, L. sp. Kur on Kur Island, and Luperosaurus macgregori on Babuyan Claro Island). These specific cases may provide instances for which more meaningful data on population trends could illuminate key ecological interactions associated with long-term persistence of unique species on isolated small islands.
-
3.
Building within-region expertise to monitor species. The (apparent) last refuges of many Lepidodactylus taxa are remote and difficult to access. This situation has only been compounded by the Covid-19 pandemic, with restrictions making travel difficult or impossible throughout the region. Building and supporting local expertise and capacity may effectively address these issues and could also strengthen the ownership countries have in protecting their threatened endemic species.
-
4.
Strengthen quarantine safeguards on remaining island refuges. Invasive species are almost certainly the most important threat to many small-island Lepidodactylus species. On islands that are frequently visited by people effective quarantine is challenging. For remote islands (e.g., Rotuma, Tuvalu and Tonium Island), however, strategies to prevent colonization by invasive rats, wolf snakes (Lycodon), common house geckos, and non-native lizards may be the only feasible strategy for preventing the extinction of many endemic species. Such strategies are currently being funded and implemented in the Pacific Islands region (e.g., PRISMSS Programme www.sprep.org/prismss/protect-our-islands), though awareness of protecting small-island endemics, such as Lepidodactylus, needs to be raised.
-
5.
Habitat protection. Indonesia and Malaysia are among the top ten countries for global forest loss since 2000, and rates of deforestation have accelerated over the last decade in Papua New Guinea and the Solomon Islands (Global Forest Watch 2014). Highlighting the threats to forest-dwelling Lepidodactylus in Melanesia is the case of Woodlark Island (874 km2) in PNG, inhabited by L. kwasnickae and at least 47 other endemic plant and animal species, wherein there are plans for near complete deforestation for palm oil plantations (Kraus 2021). Expansion of the terrestrial protected area in these countries, all of which fell short of the Aichi Target of 17% of terrestrial area by 2020 (Malaysia 13.3%, Indonesia 12.2%, PNG 3.7%, Solomon Islands 1.8%; UNEP-WCMC and IUCN 2020), could help to secure the futures of forest-dwelling Lepidodactylus, and other endemic species, particularly in lowland rainforests and coastal habitats.
Conclusion
Our data on Lepidodactylus highlight how a combination of cryptic ecologies and unrecognized species diversity can lead to significant underestimation of conservation threat, here termed Cryptic Extinction Risk (CER). Accounting for CER in conservation assessment thus has the potential to highlight at-risk taxa and geographic areas of the concern, which may otherwise be overlooked. Our results also highlight the urgency for surveys targeting threatened Pacific Island Lepidodactylus species, as well as the importance of strengthening quarantine safeguards in this region. The CER may be useful for highlighting conservation issues in other taxa that combine aspects of a workable but incomplete taxonomic framework, poorly documented geographical distributions, many localized endemics, and cryptic or otherwise poorly known ecologies.
Data availability
Data are available in the supplementary material, IUCN Red List, Global Biodiversity Information Facility and on request to authors.
Code availability
Not applicable.
References
Andrew P, Cogger H, Driscoll D et al (2018) Somewhat saved: a captive breeding programme for two endemic Christmas Island lizard species, now extinct in the wild. Oryx 52:171–174. https://doi.org/10.1017/S0030605316001071
Bell TP (2009) A novel technique for monitoring highly cryptic lizard species in forests. Herpetol Conserv Biol 4:415–425
Bland LM, Böhm M (2016) Overcoming data deficiency in reptiles. Biol Conserv 204:16–22. https://doi.org/10.1016/j.biocon.2016.05.018
Böhm M, Collen B, Baillie JEM et al (2013) The conservation status of the world’s reptiles. Biol Conserv 157:372–385. https://doi.org/10.1016/j.biocon.2012.07.015
Böhm M, Williams R, Bramhall HR et al (2016) Correlates of extinction risk in squamate reptiles: the relative importance of biology, geography, threat and range size. Glob Ecol Biogeogr 25:391–405. https://doi.org/10.1111/geb.12419
Brown WC, Alcala A (1978) Philippine lizards of the Family Gekkonidae. Silliman University Press, Philippines
Brown JH, Lomolino MV (1998) Biogeography, 2nd edn. Sinauer Press, Sunderland, Massachusetts
Brown RM, Siler CD et al (2013) The amphibians and reptiles of Luzon Island, Philippines, VIII: the herpetofauna of Cagayan and Isabela Provinces, northern Sierra Madre Mountain Range. Zookeys 266:1–120. https://doi.org/10.3897/zookeys.266.3982
Brown RM, Meneses CG, Wood PL et al (2020) Unexpected discovery of another new species of Philippine false gecko (Gekkonidae; Pseudogekko) from the Bicol Peninsula of Luzon Island. Herpetologica 76:315–329. https://doi.org/10.1655/Herpetologica-D-19-00029.1
Bucol A, Alcala A, Averia L et al (2012) Checklist of the Herpetofauna of Siquijor Island, Philippines. Philipp Sci 48:100–122. https://doi.org/10.3860/psci.v48i0.2483
Carlquist S (1974) Island biology. Columbia University Press, New York
Chadès I, McDonald-Madden E, McCarthy MA et al (2008) When to stop managing or surveying cryptic threatened species. Proc Natl Acad Sci USA 105:13936–13940. https://doi.org/10.1073/pnas.0805265105
Chapple DG, Roll R, Böhm M et al (2021) Conservation status of the world’s skinks (Scincidae): taxonomic and geographic patterns in extinction risk. Biol Cons 257:109101. https://doi.org/10.1016/j.biocon.2021.109101
Cheke AS, Hume JP, (2008) Lost Land of the Dodo: The Ecological History of the Mascarene Islands. T. & A. D. Poyser, London
Crombie RI, Menz R (2007) Lepidodactylus paurolepis Ota, Fisher. Ineich and Case Sauria 29:2
Das I, Lakin M, Kandaung P (2008) New species of Luperosaurus (Squamata: Gekkonidae) from the Crocker Range Park, Sabah, Malaysia (Borneo). Zootaxa 1719:53–60
Eliades SJ, Brown RM, Huang W, et al (2021) Taxonomic Revision of Scaly-toed Geckos (Reptilia: Gekkonidae: Lepidodactylus) in the Northern Philippines, with Descriptions of Four New Species. Herpetological Monographs 35
Emery JP, Mitchell NJ, Cogger H et al (2021) The lost lizards of Christmas Island: a retrospective assessment of factors driving the collapse of a native reptile community. Conserv Sci Pract 3:e358. https://doi.org/10.1111/csp2.358
Fernández-Palacios JM, Otto R, Borregaard MK et al (2021) Evolutionary winners are ecological losers among oceanic island plants. J Biogeogr. https://doi.org/10.1111/jbi.14143
Geyle HM, Tingley R, Amey AP et al (2021) Reptiles on the brink: identifying the Australian terrestrial snake and lizard species most at risk of extinction. Pacific Conserv Biol 27:3–12. https://doi.org/10.1071/PC20033
Gillespie GR, Fukuda Y, McDonald P (2020) Using non-systematically collected data to evaluate the conservation status of elusive species: a case study on Australia’s Oenpelli python. Wildl Res 47:146–157. https://doi.org/10.1071/WR19112
Global Forest Watch (2014) Global Forest Watch. In: World Resour. Inst. www.globalforestwatch.org. Accessed 18 Jun 2021
Grismer LL (2011) Lizards of Peninsular Malaysia, Singapore, and their adjacent archipelagos: Their description, distribution, and natural history. Edition Chimaira, Frankfurt
Ineich I (2008) A new arboreal Lepidodactylus (Reptilia: Gekkonidae) from Espiritu Santo Island, Vanuatu: from egg to holotype. Zootaxa 38:26–38
Ineich I (2019) How habitat disturbance benefits geckos: conservation implications. CR Biol 333:76–82. https://doi.org/10.1016/j.crvi.2009.11.005
IUCN (2021) The IUCN Red List of Threatened Species. https://www.iucnredlist.org
Karin BR, Stubbs AL, Arifin U et al (2018) The herpetofauna of the Kei Islands (Maluku, Indonesia): comprehensive report on new and historical collections, biogeographic patterns, conservation concerns, and an annotated checklist of species from Kei Kecil, Kei Besar, Tam, and Kur. Raffles Bull Zool 66:704–738
Karin BR, Oliver PM, Stubbs AL et al (2021) Who’s your daddy? On the identity and distribution of the paternal hybrid ancestor of the parthenogenetic gecko Lepidodactylus lugubris (Reptilia: Squamata: Gekkonidae). Zootaxa 4999:87–100
Karkkainen DT, Richards SJ, Kraus F et al (2020) A new species of small Lepidodactylus (Squamata: Gekkonidae) from Salawati Island, Indonesia. Isr J Ecol Evol 66:180–189. https://doi.org/10.1163/22244662-bja10001
Kraus F (2019) New species of Lepidodactylus (Squamata: Gekkonidae) from New Guinea and adjacent islands. Zootaxa 4651:305–329
Kraus F (2021) A herpetofauna with dramatic endemism signals an overlooked biodiversity hotspot. Biodivers Conserv. https://doi.org/10.1007/s10531-021-02242-3
Kraus F, Oliver PM (2019) A new species of Lepidodactylus (Squamata: Gekkonidae) from the mountains of northeastern Papua New Guinea: Older than the hills. Zootaxa 4718:549–561
Lintermans M, Geyle HM, Beatty S et al (2020) Big trouble for little fish: identifying Australian freshwater fishes in imminent risk of extinction. Pacific Conserv Biol 26:365–377. https://doi.org/10.1071/PC19053
Lomolino MV (2004) Conservation biogeography. In: Lomolino MV, Heaney LR (eds) Frontiers of Biogeography: new directions in the geography of nature. Sinauer Associates, Sunderland, Massachusetts, pp 293–296
Louys J, Braje TJ, Chang CH et al (2021) No evidence for widespread island extinctions after Pleistocene hominin arrival. Proc Natl Acad Sci 118(20):e2023005118. https://doi.org/10.1073/pnas.2023005118
McCoy M (2006) Reptiles of the Solomon Islands. Pensoft Publishers, Bulgaria
McDonald LL (2004) Sampling rare populations. In: Thompson W (ed) Sampling rare or elusive species: concepts, designs and techniques for estimating population parameters. Island Press, Washington, pp 11–42
Meiri S, Bauer AM, Allison A et al (2018) Extinct, obscure or imaginary: the lizard species with the smallest ranges. Divers Distrib 24:262–273. https://doi.org/10.1111/ddi.12678
Meneses CG, Siler CD, Gonzalez JCT et al (2020) Molecular phylogenetic estimates of evolutionary affinities and the first reports of phenotypic variation in two secretive, endemic reptiles from the Romblon Island Group, central Philippines) from Luzon Island, the Philippines. Philipp J Syst Biol 14:1–20
NSB (1989) Loss of biological diversity: a global crisis requiring international solutions. Washington, D.C.
O’Shea M, Kusuma KI, Kaiser H (2018) First Record of the Island Wolfsnake, Lycodon capucinus (H. Boie in F. Boie 1827), from New Guinea, with comments on its widespread distribution and confused taxonomy, and a new record for the common sun skink Eutropis multifasciata (Kuhl 1820). IRCF Reptil Amphib 25:70–84
Oliver PM, Adams A, Lee MSY, Hutchinson MN, Doughty P (2009) Cryptic diversity in vertebrates: molecular data double estimates of species diversity in a radiation of Australian lizards (Diplodactylus, Gekkota). Proc R Soc B Biol Sci 276:2001–2007. https://doi.org/10.1098/rspb.2008.1881
Oliver PM, Parker F, Tallowin O (2015) Further records of reptiles and amphibians utilising ant plant (Rubiaceae) domatia in New Guinea. Herpetol Notes 8:239–241
Oliver PM, Blom MP, Cogger HG et al (2018a) Insular biogeographic origins and high phylogenetic distinctiveness for a recently depleted lizard fauna from Christmas Island. Australia Biol Lett 14:20170696. https://doi.org/10.1098/rsbl.2017.0696
Oliver PM, Brown RM, Kraus F et al (2018b) Lizards of the lost arcs: Mid-Cenozoic diversification, persistence and ecological marginalization in the west pacific. Proc R Soc B Biol Sci 285:20171760. https://doi.org/10.1098/rspb.2017.1760
Oliver PM, Brown RM, Karin BR, Lee Grismer L (2020) Mountain endemism in Malesian geckos: Can biotic interactions push lizards up hills? Isr J Ecol Evol 66:190–201. https://doi.org/10.1163/22244662-20191089
Pregill GK (1993) Fossil lizards from the late Qauternary of ’Eua, Tonga. Pac Sci 47:101–114
Pregill GK (1998) Squamate reptiles from prehistoric sites in the Mariana Islands. Copeia 1998:64–75
Pregill GK, Steadman DW (2009) The prehistory and biogeography of terrestrial vertebrates on Guam, Mariana Islands. Divers Distrib 15:983–996. https://doi.org/10.1111/j.1472-4642.2009.00603.x
Pregill GK, Steadman DW (2014) The prehistory of terrestrial reptiles and birds in the central lau group, fiji. Florida Museum Nat Hist Bull 53:1–25
R Core Team (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Richmond JQ, Ota H, Grismer LL et al (2021) Influence of niche breadth and position on the historical biogeography of seafaring scincid lizards. Biol J Linn Soc Lond 132:74–92. https://doi.org/10.1093/biolinnean/blaa172
Ripple WJ, Wolf C, Newsome TM et al (2017) Extinction risk is most acute for the world’s largest and smallest vertebrates. Proc Natl Acad Sci USA 114:10678–10683. https://doi.org/10.1073/pnas.1702078114
Rix MG, Wilson JD, Harvey MS (2020) First phylogenetic assessment and taxonomic synopsis of the open-holed trapdoor spider genus Namea (Mygalomorphae: Anamidae): a highly diverse mygalomorph lineage from Australia’s tropical eastern rainforests. Invertebr Syst 34:679–726. https://doi.org/10.1071/IS20004
Rodrigues ASL, Pilgrim JD, Lamoreux JF et al (2006) The value of the IUCN Red List for conservation. Trends Ecol Evol 21:71–76. https://doi.org/10.1016/j.tree.2005.10.010
Rosauer DF, Byrne M, Blom MP et al (2018) Real-world conservation planning for evolutionary diversity in the Kimberley, Australia, sidesteps uncertain taxonomy. Conserv Lett 11:e12438. https://doi.org/10.1111/conl.12438
Sanguila MB, Cobb CD et al (2016) The amphibians and reptiles of Mindanao Island, southern Philippines, II: the herpetofauna of northeast Mindanao and adjacent islands. Zookeys 624:1–132. https://doi.org/10.3897/zookeys.624.9814
Siler CD, Welton LJ, Davis DR et al (2014) Taxonomic revision of the Pseudogekko compresicorpus Complex (Reptilia: Squamata: Gekkonidae), with descriptions of three new species. Herpetol Monogr 28:110–139. https://doi.org/10.1655/HERPMONOGRAPHS-D-14-00005
Steadman DW (2006) Extinction and biogeography of tropical Pacific birds. The University of Chicago Press, Chicago
Stubbs AL, Karin BR, Arifin U et al (2017) A new species of Lepidodactylus (Reptilia: Squamata: Gekkonidae) from the Kei Islands, Maluku, Indonesia. Zootaxa 4350:91–105
Supsup C, Puna NM et al (2020) Amphibians and reptiles of Cebu, Philippines: the poorly understood herpetofauna of an island with very little remaining natural habitat. Asian Herpetol Res 7:151–179
Symonds MRE, Moussalli A (2011) A brief guide to model selection, multimodel inference and model averaging in behavioural ecology using Akaike’s information criterion. Behav Ecol Sociobiol 65:13–21. https://doi.org/10.1007/s00265-010-1037-6
Tingley R, Hitchmough RA, Chapple DG (2013) Life-history traits and extrinsic threats determine extinction risk in New Zealand lizards. Biol Conserv 165:62–68. https://doi.org/10.1016/j.biocon.2013.05.028
Tingley R, Macdonald SL, Mitchell NJ et al (2019) Geographic and taxonomic patterns of extinction risk in Australian squamates. Biol Conserv 238:108203. https://doi.org/10.1016/j.biocon.2019.108203
Turvey ST, Crees JJ, Hansford J et al (2017) Quaternary vertebrate faunas from Sumba, Indonesia: implications for Wallacean biogeography and evolution. Proc R Soc B Biol Sci 284:20171278. https://doi.org/10.1098/rspb.2017.1278
UNEP-WCMC, IUCN (2020) Protected Planet: The World Database on Protected Areas (WDPA). https://www.protectedplanet.net/. Accessed 18 Jun 2021
Wiles GJ, Conry PJ (1990) Terrestrial vertebrates of the Ngerukewid Islands Wildlife Preserve, Palau Islands. Micronesica 23:41–66
Wood PL Jr, Guo X, Travers SL et al (2020) Parachute geckos free fall into synonymy: Gekko phylogeny, and a new subgeneric classification, inferred from thousands of ultraconserved elements. Mol Phylogenet Evol 146:106731. https://doi.org/10.1016/j.ympev.2020.106731
Zug GR, Watling D, Alefaio S, Ludescher C (2003) A new gecko (Reptilia: Squamata: Genus Lepidodactylus) from Tuvalu, South-central Pacific. Proc Biol Soc Washingt 116:38–46
Acknowledgements
We thank Sven Mecke for providing data on some Pacific Lepidodactylus species. Research in the Philippines has been facilitated by the Biodiversity Management Bureau, the Department of the Environment Natural Resources, and made possible grants from the U. S. National Science Foundation, including DEB 0073199, 0910341, 0743491, 0640737, 1418895, 1654388, and EF-0334952 (to RMB and KU-based graduate students), IOS 1353683 and DEB 1657648 and 0804115 (to CDS); Fulbright and Fulbright-Hays grants (to CDS); the Rufford Foundation (to CGM); the Philippine Commission on Higher Education-IDIG, and Philippine Department of Science and Technology-GIA (to MBS). Research in the Pacific Islands has been funded by many groups including Mohamed bin Zayed, Conservation International, Island Conservation, Wildlife Conservation Society, IUCN Oceania, SPREP, CEPF, San Diego Zoo Global, and U.S. Geological Survey. Any use of trade, firm or product names is for illustrative purposes and does not imply endorsement by the U.S. Government.
Funding
No funding to declare.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declare that they have conflict of interest.
Additional information
Communicated by Pedro Aragón.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
McDonald, P.J., Brown, R.M., Kraus, F. et al. Cryptic extinction risk in a western Pacific lizard radiation. Biodivers Conserv 31, 2045–2062 (2022). https://doi.org/10.1007/s10531-022-02412-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-022-02412-x