Abstract
Reintroduction of grazing to counteract insect decline will lead to an increase of semi-open habitats. Semi-open habitats are highly heterogeneous, consisting of a mosaic of patches of trees or shrubs embedded in a matrix dominated by dwarf shrubs or grasses. Despite a lack of evidence, structural heterogeneity is expected to allow many species to co-occur, making semi-open habitats appear highly valuable for invertebrate conservation. We studied ground beetles in eight sites in two landscapes of Central Europe each encompassing semi-open, open, and forest habitats. Rapid response to environmental change and limited dispersal abilities make ground beetles an excellent model taxon to evaluate the effectiveness of such conservations measures. In both studied landscapes, ground beetle assemblages in semi-open habitats tended to be distinct and intermediate between those from the forest and open habitats. Species richness and functional diversity in semi-open habitats were similar to open habitats at site level. The majority of species entered the semi-open habitats, except for most threatened species, yet, few species were exclusively associated with semi-open habitats. We conclude that the continuous presence of many species in semi-open habitats likely results from mass effects rather than habitat heterogeneity per se. Our findings underline the conservation value of the existing forest, heathland, and grassland habitats over semi-open habitats which can, however, function as dispersal habitats and increase landscape connectivity. Strategies aiming at promoting semi-open habitats to counteract insect decline should target enhancing connectivity rather than the creation of habitats only.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Conservationists call for increasing structural diversity in forest and open habitats to support insect conservation (Samways et al., 2020). In various locations in Europe, this is achieved by the reintroduction of grazing, through trophic rewilding (sensu Svenning et al., 2016) or restoration and establishment of wood pastures (e.g. Garrido et al., 2021; Rösch et al., 2019; Vera, 2009) leading to an increase of semi-open habitats (e.g. Hall, 2018; Schulze et al., 2018). Semi-open habitats were once a common feature of temperate Europe (Sandom et al., 2014; Vera, 2000) and consist of a mosaic of patches of trees or shrubs embedded in a matrix dominated by grasses or dwarf shrubs (Garbarino and Bergmeier, 2014). Gradients in canopy closure affect light availability, temperature, and moisture at fine-scale, making semi-open habitats highly heterogeneous (Bergmeier et al., 2010; Díaz et al., 2013). The structural heterogeneity (cf. “habitat heterogeneity hypothesis” MacArthur and MacArthur, 1961) is assumed to offer a range of niches allowing many species to co-occur (Díaz et al., 2013) and is seen to influence assembly rules (Magura and Lövei, 2019). Consequently, semi-open habitats are regarded as highly valuable for conservation due to both their high species richness and unique assemblage composition (e.g. Bergmeier et al., 2010; Rösch et al., 2019). For invertebrates in semi-open habitats, however, the few existing studies showed non-equivocal results with few supporting their effectiveness for conservation (David et al., 1999; Horák et al., 2018; Talvi, 1995), while others did not come to the same conclusion (Gallé et al., 2017; Rösch et al., 2019; Somay et al., 2021; Sverdrup-Thygeson et al., 2010).
To assess the effectiveness of semi-open habitats for invertebrate conservation in the temperate region, four aspects must be considered. Firstly, considering the particular environmental conditions found in semi-open habitats, many authors assume that a set of specific species preferring semi-open conditions exist (e.g. Rösch et al., 2019; Sverdrup-Thygeson et al., 2010). Despite its importance for species conservation (Cáceres and Legendre, 2009), this knowledge remains limited for invertebrates and available evidence indicates that a rather small number of species likely rely on semi-open habitats (Díaz et al., 2013). Secondly, whether semi-open habitats do effectively harbour a large number of species seems affected by the scale at which species richness is considered (e.g. Moreno et al., 2016). Most studies that reported higher species richness in semi-open habitats focussed on γ-diversity (across habitat patches; e.g. Horák et al., 2018; Talvi, 1995) while α-diversities (at patch level) appeared at least similar to one of the adjacent habitats (e.g. Rösch et al., 2019; Somay et al., 2021). Thirdly, most studies were conducted in either the boreal (e.g. Sverdrup-Thygeson et al., 2010; Talvi, 1995) or the Mediterranean regions (e.g. David et al., 1999; Moreno et al., 2016; Taboada et al., 2006a) where some semi-open habitats are under protection (Bergmeier et al., 2010) while studies in the temperate region remain limited. Fourth, dwarf shrubs dominated semi-open habitats are a key element in the oceanic part of temperate Europe (Garbarino and Bergmeier, 2014), yet most studies on semi-open habitats focused on grass-dominated matrices (but see Eggers et al., 2010).
It is assumed that habitats with a higher functional diversity are more stable, better functioning, and, thus, of high conservation value (Cadotte et al., 2011). This can also be expected for semi-open habitats, however, in these habitats mainly taxonomic diversity has been studied and little attention has been paid to functional diversity (but see Jakobsson et al., 2020). The high niche availability in semi-open habitats is expected to increase the functional diversity as functionally divergent species occupy those diverse niches (MacArthur and MacArthur, 1961). For birds and bats, Jakobsson et al. (2020) found that increasing structural complexity in semi-open habitats increased the taxonomic diversity but no similar tendency in functional diversity was found. It is, though, likely that a relationship between habitat heterogeneity and functional diversity occurs as increased habitat heterogeneity drives a higher species richness which, in turn, increases functional diversity (but see Gerisch et al., 2012). In addition, as assemblages in semi-open habitats are expected to be a combination of species from open and forest habitats (Díaz et al., 2013), with usually distinct functional traits, semi-open habitats are likely to exhibit a high functional diversity.
Previous studies on diversity in semi-open habitats focussed on taxa mostly restricted to open (grasshoppers: Rösch et al., 2019; dung beetles: Somay et al., 2021) or forest habitats (saproxylic beetles: Horák et al., 2018; Sverdrup-Thygeson et al., 2010). Ground and tiger beetles (hereafter ground beetles), however, occur more evenly in all terrestrial habitats (Koivula, 2011; Lövei and Sunderland, 1996) and are known to respond to changes in moisture and temperature parameters at a very small spatial scale (Antvogel and Bonn, 2001; Koivula, 2011) making them suitable for studying habitat association in heterogeneous semi-open habitats. However, up to now, studies on ground beetle assemblages in semi-open habitats exist mostly for the Mediterranean (e.g. Taboada et al., 2006a, b) or the boreal regions (e.g. Söderström et al., 2001; Talvi, 1995) and are rare for the temperate one (but see Eggers et al., 2010).
In this study, we aim to evaluate the conservation potential of semi-open habitats for ground beetles in the temperate region where we sampled ground beetles in two climatically different landscapes containing remnants of semi-open habitats. We hypothesize that species richness and functional diversity will be higher in semi-open habitats than in adjacent forests, heathlands, or grasslands. Furthermore, we expect ground beetle assemblages to be distinct and intermediate between those of the forest and open habitats. Finally, we identify potential ground beetle species with a specific preference for semi-open habitats with a particular focus on threatened species.
Materials and methods
Study landscapes, history, and habitat characteristics
The study was conducted in two landscapes in Germany with different climatic and edaphic conditions.
In both landscapes, the open habitats developed as the result of deforestation culminating in the Middle Ages and have been traditionally maintained by sheep grazing. When in the 19th century the traditional management became economically non-viable, many of the formerly grazed heathlands and dry calcareous grasslands have been afforested or transformed into arable lands (Beinlich and Plachter, 1995; Keienburg and Prüter, 2004).
The Lüneburger Heide (hereafter Heide) is located in the north-western German lowland (70–170 m.a.s.l.; 53.1675 N, 9.91345 E), has a suboceanic climate (mean annual precipitation: 854 mm, mean annual temperature 9 °C) on podzol as the predominant soil type (Keienburg and Prüter, 2004). The open habitats consist of dry heathland dominated by heather (Calluna vulgaris) and represent the remnants of formerly widespread heathland. The semi-open habitats developed more recently and comprise a mosaic dominated by heather with shrubs, single trees, or groups of trees (Betula pendula, Quercus robur, Pinus sylvestris), which resulted either from a combination of trees and shrubs encroachment and extensive sheep and goats grazing on former heathland or they were established by forest thinning followed by extensive grazing. Sheep and goats herds (about 0.8 animal/hectare) are grazing year-round in both open and semi-open habitats which are part of the same grazing area (personal communication Dirk Mertens, Verein Naturschutzpark). The forest habitats, which are not grazed, developed in the 19th century and are dominated by the same tree species as found in the semi-open habitats with the addition of Picea abies. Tree canopy cover (visually estimated for each pitfall trap) was on average 0% in the heathland, 20% in the semi-open, and 51% in the forest habitats (Table 1 and Online Resource Table A1).
The Schwäbische Alb (hereafter Alb) is located in a low mountain range in south-west Germany (450–1000 m.a.s.l. ; 48.2983 N, 9.24926 E), has a subcontinental climate (mean annual precipitation: 750 mm, mean annual temperature 7 °C) and limestone derived rendzic leptosol soil (Beinlich and Plachter, 1995). The open habitats, which are remnants of the former widespread grasslands, consist of semi-dry calcareous grassland dominated by Bromus erectus. In this landscape, the semi-open habitats resulted from a combination of tree encroachment and extensive sheep grazing. They comprise a mosaic dominated by the grass Bromus erectus with shrubs (Juniperus communis) and trees (Quercus robur and Sorbus aucuparia) isolated or in small groups. Sheep herds (about 3.5 to 4.6 animals/hectare) are grazing in a way that imitates the former transhumance (one grazing event in late spring followed by a second one in late summer to remove two-thirds of the biomass on each grazing event (Ministerium für Ernährung, Ländlichen Raum und Verbraucherschutz, 2021). Open and semi-open habitats are part of the same grazing area. The forest habitats are ungrazed and are mainly dominated by Fagus sylvatica. Tree canopy cover was on average 0%, 26%, and 77% in the open, semi-open, and forest habitats, respectively (Table 1 and Online Resource Table A1). More details about the studied landscapes and plant composition can be found in Travers et al. (2020).
We measured the proportion of the main habitat types (forest, semi-open, open (heathland or dry grassland depending on the landscape), other open habitats (include arable lands and other types of grassland)) in a 500 m radius centered on each sampling site (radius sufficiently large to include all three habitats) with the measuring tool using Google Earth Pro (version 7.3.4.8248). Urbanized and other artificial “habitats” (constructed, road networks, small gardens) were excluded as they are unlikely to offer suitable environments for the studied ground beetle species. In both landscapes, the semi-open habitats were not significantly different with regard to patch size, canopy cover, and proportion of forest, semi-open, open, and other open habitats (Table 1). Detailed information on the studied habitats and sites can be found in Online Resource Table A1.
Ground beetle sampling
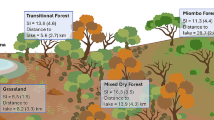
In each landscape, we selected eight sites (> 2 km apart) of which we surveyed half of them in 2013–2014 and the other half in 2014–2015 (Online Resource Table A1). Each site comprised patches of the three habitat types — open, semi-open, and forest habitats — in direct vicinity (Fig. 1). In each of the three habitats within a site, a row of five pitfall traps with 10 m between traps was used. The average distance between habitat rows was 218 m in the Heide (ranged from 81 to 809 m) and 207 m (ranged from 37 to 586 m) in the Alb.
Schematic representation of the sampling design based on one studied site from the Lüneburger Heide (a) in which the red-violet dots indicate the position of the trapping rows in the forest (green), semi-open (grey), and heathland (purple) and perspective at ground level in the forest (b), semi-open (c and e), and open heathland (d)
Corrected sample-based rarefaction curves (γ- diversity, a and b), rarefied species richness (α diversity, c and d), and functional diversity (Rao’s Q, e and f) of ground beetles for forest (green, solid line), semi-open (grey, dashed line), and open (purple, dotted line) habitats in the Lüneburger Heide (left column) and the Schwäbische Alb (right column). The (a) and (b) panels display 84% confidence intervals. Different letters in c) to f) above the boxplots indicate significant differences between the habitats (Online Resource 2) and the red-violet dot-dashed lines the mean value
A total of 120 traps were used in each landscape (8 sites x 3 habitats x 5 traps). In the Heide, traps were serviced year-round from January 2013 to February 2014 and from March 2014 to January 2015 every three weeks (except for extended trapping periods caused by snow cover in winter). In the Alb, traps were serviced every three weeks from April to September in 2013 and 2014. This resulted in 18 collection events per site in the Heide in 2013–2014 (1080 traps collected in total), 15 in 2014–2015 (900 traps), and, in the Alb, eight collection events in both years (960 traps). As pitfall traps, we used transparent plastic cups (500 ml, 10 cm diameter) filled with 150 ml of Renner solution (Renner, 1982) set flush with the ground. To reduce disturbance by sheep an acrylic glass roof was added in the Alb, 3–4 cm above the trap.
Distances to the habitat edges were measured as the distance between a given trap and the limit of the tree crowns at the forest or semi-open habitat edges. In the Heide, the distance from the heathland and forest edges to the sampled locations within the semi-open habitats ranged from 10 to 115 m. In the Alb, the distance to the grasslands and the forest edges ranged from 13 to 68 m and 33 to 187 m, respectively (Table 1 and Online Resource Table A1). The distance from the forest or open habitat edges did not significantly differ between both landscapes (Table 1).
Ground beetles were counted and identified to species level in the laboratory using standard literature (Müller-Motzfeld, 2006) and nomenclature followed Schmidt et al. (2016). Species were classified as threatened if they were listed as vulnerable, endangered, or critically endangered on either the German Red List (Schmidt et al., 2016) or the Red List of the respective federal states (Assmann et al., 2003; Trautner et al., 2006). We selected six traits commonly used for ground beetles characterizing their spatial and temporal resources use (one continuous: mean body size (mm); five nominal variables: wing development (brachypterous, dimorphic, and macropterous), food preference (predator, herbivore, and omnivore), hibernation type (imagines, larvae, or both stages), reproduction time (spring, autumn, spring and autumn, and winter), and region-specific habitat preference (forest, open, wetland, and eurytopic)). Trait information was compiled from www.carabids.org (Homburg et al., 2014) amended with information from Nolte et al. (2019), and region-specific habitat preferences were based on the catalogue of the Gesellschaft für Angewandte Carabidologie (2009).
Statistical analyses
Due to the activity of large mammals, 37 traps (1.9%) in the Heide and 55 traps (5.7%) in the Alb were lost. The missing information was replaced by the average catch value corresponding to the remaining undisturbed traps of the given site, habitat, and trapping interval. Prior to statistical analyses, we pooled the catches of all five pitfall traps over the entire trapping period within each habitat and site to receive one sample per habitat and site which resulted in eight per habitat and landscape. Statistical analyses were performed using the software R, version 3.5.3 (R Core Team, 2019) with package ggplot2 for graphics (Wickam, 2016).
Sample-based rarefaction curves were used to estimate rarefied species richness for each habitat type (γ-diversity). Rarefaction curves for each habitat type in each landscape were calculated using the “specaccum” function (2500 permutations) with 84% confidence intervals (Payton et al., 2003). To correct for differences in numbers of individuals sampled, the rarefaction curves were re-plotted with an individual-based x-axis (Gotelli and Colwell, 2001). We calculated site-rarefied species richness (α-diversity) using the function “rarefy” considering the minimum number of individuals trapped in a site in each landscape (423 individuals in the Heide; 231 in the Alb). Both functions are part of the package vegan (Oksanen et al., 2019).
As functional diversity index, we used Rao’s quadratic entropy, which represents the mean trait distance between pairs of individuals in one assemblage (Botta-Dukát, 2005), calculated using the function “dbFD” of the FD package (Laliberté and Legendre, 2010). Rao’s quadratic entropy is expected to increase with increasing divergence of species traits independently from species richness which could be expected in semi-open habitats if species from both open and forest habitats co-occur.
As ground beetle assemblages and, thus, species richness and functional diversity can be influenced by landscape and site characteristics we checked for linear relationships with α- or functional diversity found in the semi-open habitats. We analysed site characteristics (patch size, canopy cover, and distance to the forest or open edges) and landscape characteristics (proportion of forest, semi-open, open, and other open habitats) by building two sets of linear models. No significant effect of any of the variables on α- or functional diversity was found (Online Resource Table A2). However, such questions should be addressed with targeted study designs.
The effect of habitat types and year of sampling on α- and functional diversity were first tested with a linear mixed model using the study site as a random factor. As the variance explained by the random factor was close to 0, it was excluded and linear models were used instead. Significant differences between habitat types were assessed, using the “glht” function from the multcomp package (Hothorn et al., 2008), with post–hoc tests of pairwise differences (Tukey test). Model validation was carried out graphically.
To compare the carabid beetle assemblages in the different habitat types, non-metric multidimensional scaling (NMDS, vegan package) was conducted for each landscape separately. We omitted carabid species with fewer than five individuals recorded per landscape. NMDS ordinations with Horn dissimilarity measures, which give more weight to rarer species (Jost et al., 2011), were restricted to two dimensions. Two ordinations were built for each landscape. The first one focussed exclusively on the semi-open habitats and using the function “envfit” (5 000 permutations) we checked whether the patch size, canopy cover, distance to the forest or open edges, proportion of forest, semi-open, open, and other open habitats significantly influenced the structure of the assemblages. The second ordination included all habitat types and was used to test for significant differences between all habitats, pairs of habitats, and sampling years using PERMANOVAs (5000 permutations) based on the dissimilarity matrices used for the ordination. The obtained p-values from “envfit” and those of the pairwise comparisons of habitats were corrected using the false discovery rate (function “p.ajust”; Benjamini and Hochberg, 1995).
Furthermore, we calculated the point-biserial correlation coefficient (rpb) for each species and habitat type combination to test for associations of carabid species with habitat types correcting for unequal group size. Rpb is the correlation coefficient between two vectors, one containing the species abundances in the habitat types and the other one the habitat type values, and ranges from − 1 (complete avoidance) to 1 (absolute preference). As rpb considers both species presence in the target habitat and absence in the other habitats, we further excluded species that occurred on fewer than three sites per landscape. Rpb was calculated using the function “signassoc” and the significance of association was tested using two‐sided permutation tests (1000 permutations) with p-values corrected for multiple testing (Šidák’s correction). We calculated 95% confidence intervals for the association with semi-open habitats by bootstrapping the observed data 1000 times with replacement using the function “strassoc”. Both functions are part of the package indicspecies (Cáceres and Legendre, 2009).
Results
In total, we sampled 28790 carabid beetles belonging to 96 species in the Heide (7849, 8671, and 12270 individuals in the forest, semi-open, and heathland habitats, respectively). We found 26 species with fewer than five individuals. In the Alb, we recorded 14 453 carabid beetles belonging to 95 species (6926, 3518, and 4009 individuals sampled in the forest, semi-open, and grassland habitats, respectively) with 19 species for which fewer than five individuals were recorded. We recorded 15 and 11 threatened species in the Heide and the Alb, respectively (Online Resource Table A3).
Corrected sample-based rarefaction curve slopes were similar for the three habitats in both landscapes, except for the forest in the Alb with a shallower slope, indicating similar sampling completeness was achieved in most habitats (Fig. 2a and b). Ground beetles exhibited a significantly higher γ-diversity in the semi-open habitats (not overlapping confidence interval) followed by the open and forest habitats in both studied landscapes (Heide: rarefied species richness for 7927 individuals of 71, 61.3, and 61 for semi-open, open, and forest habitats, respectively; Alb: rarefied species richness for 3708 individuals of 74, 67, and 37.4 for semi-open, open, and forest habitats, respectively).
No significant differences in α-diversity and functional diversity were found between the sampling years in both landscapes (Table 2 and Online Resource Table A4). Contrary to γ-diversity, in both landscapes, α-diversity was comparable between the semi-open and the open habitats while being higher than in the forests (Fig. 2c and d; Table 2 and Online Resource Table A4). In the Heide, functional diversity was similar between the three habitats while in the Alb both semi-open and open habitats had higher functional diversity compared to the forest (Fig. 2e and f; Table 2 and Online Resource Table A4).
The results of the NMDS analysis showed that the different habitats in each study landscape clustered together (Fig. 3). Assemblages in semi-open habitats were intermediate between the open and forest habitats, thus influenced by both adjacent habitats. PERMANOVAs revealed significant differences between the habitats for both landscapes along the first axis of the ordination with the forest and open habitat assemblages clearly separated and significantly different from each other. In the Heide, the assemblages from semi-open habitats showed higher similarity to those from the forests than from the open habitats, although all assemblages were significantly different. In the Alb, a trend existed for a difference between the assemblages from semi-open and open habitats while semi-open assemblages were significantly different from those from the forests. In addition, in the Heide, no effect of sampling year could be found, however, in the Alb a significant difference between the assemblages was found along the second axis (Table 3, Online Resource Fig. A1).
Non-metric multidimensional scaling ordinations of carabid beetle assemblages of forest (triangle, green), semi-open (diamond, grey), and open (circle, purple) habitats in the Lüneburger Heide (a) and the Schwäbische Alb (b) based on Horn distance (two dimensions). Minimum convex polygons are shown for forest (solid line), semi-open (dashed line), and open habitat (dotted line)
No correlation between single landscape or site variables and the ordination locations of semi-open habitats was found for the Heide. In the Alb, only the proportion of other open habitats significantly correlated with the second axis of the ordination (Online Resource Table A5).
In both landscapes, similar proportions of species were positively associated with the semi-open habitat only (Heide: 15.2%; Alb: 17.5%) and both the semi-open and open habitats (Heide: 17%; Alb: 17.5%). While a lower proportion of species was positively associated with both the semi-open and the forest habitats in the Heide (11.9%), this proportion was much lower in the Alb (5.3%). Those associations with the semi-open habitats only were significant for one species in the Heide (Amara similata) and three species in the Alb (Panagaeus bipustulatus, Carabus nemoralis, and C. granulatus). In addition, two species were significantly associated with either the semi-open and the open habitats (Nebria salina) or the semi-open and the forest habitats (Pterostichus niger) in the Heide (Online Resource Table A6). Two species (Badister bullatus and Pterostichus ovoideus) were also significantly positively associated with both the semi-open and the open habitats in the Alb (Fig. 4 and Online Resource Table A7).
Number of associated species (a and b) and threatened species (c and d) unique to or shared by the forest (green, solid line), the semi-open (grey, dashed line), and the open (purple, dotted line) habitats in the Lüneburger Heide (left column) and the Schwäbische Alb (right column). Values in parentheses correspond to the number of species significantly associated with the given habitat
Most of the threatened species were associated with the open habitats in both landscapes including five of them significantly associated with the heathlands in the Heide. In addition, in the Heide, only one species was positively associated with both semi-open and forest habitats (Carabus convexus) while, in the Alb, one species was positively associated with both the semi-open and the open habitats (Harpalus dimidiatus). The number of threatened species in the forest was overall very low and only one species was found to be significantly associated with forest in the Alb (Carabus irregularis) (Fig. 5, and Online Resource Tables A6 and A7).
Ground beetle species association with semi-open habitats in both studied landscapes. Species are grouped by habitat association with either forest (dark green, filled triangle), both forest and semi-open (light green, empty triangle), semi-open (grey, diamond), both the open and the semi-open (light purple, empty circle), and open (dark purple, filled circle) habitats and further sorted alphabetically. The symbols correspond to the point-biserial correlation coefficient and the error bars show the 95% confidence interval. Threatened species are written in red. Explanation about abbreviated species names can be found in Online Resource Table A2
A large number of species in both landscapes were neither positively nor negatively associated with semi-open habitats (Heide: 88.1% of the species; Alb: 84.2%). Only one species associated with forest habitats (1.7%) and four species associated with open habitats (6.8% including the threatened species Bembidion nigricorne and Bradycellus ruficollis) were significantly negatively associated with semi-open habitats in the Heide. In the Alb, nine of the forest-associated species (14% including the threatened species Carabus irregularis) and one of the open-associated species (1.8%) were significantly negatively associated with semi-open habitat (Fig. 5, Online Resource Tables A6 and A7).
Discussion
We found a higher γ-diversity of ground beetle species in semi-open habitats than in adjacent heathlands or grasslands, and forests in two landscapes in Germany. In contrast, both α- and functional diversity of ground beetles in semi-open habitats were similar to those of open habitats. The assemblages found in semi-open habitats were distinct yet intermediate between those of their adjacent open and forest habitats. About 40 to 45% of the species recorded in semi-open habitats showed a preference for semi-open habitats exclusively or in combination with either open or forest habitats. While most of the species recorded did not significantly avoid semi-open habitats, the small proportion (8–15%) of species avoiding it comprised numerous threatened ones. We found similar results in both studied landscapes irrespective of the dominant type of vegetation in the matrix and neither habitat nor landscape characteristics nor sampling year were influential in determining the difference between habitats or within the semi-open ones.
Ground beetle species richness and assemblages
A comparison of our results with other studies needs to consider the scale. At patch level, α-diversity was comparable to that of open habitats while at landscape level, γ diversity was highest in semi-open habitats. The former is in agreement with ground beetle studies from the Mediterranean regions (Taboada et al., 2006a, b) while the latter is comparable to a study in the boreal region (Talvi, 1995). This pattern appears to contradict most studies on semi-open habitats which are considered of high conservation value because of their higher species richness (e.g. Bergmeier et al., 2010). However, usually studies only report either α- or γ-diversity. Moreno et al. (2016) found similar discrepancies between landscape and site level diversities for other invertebrate groups in the Spanish Dehesa as a consequence of a higher species turnover between sites. In our case, no such difference in β-diversity existed (data not shown but see ordination in Fig. 3). Thus, similar richness values at site and landscape levels were expected. However although not significantly different on average, species richness at site level was higher in the semi-open than in the open habitats which likely resulted in the higher species richness found at landscape level.
Following the “habitat heterogeneity hypothesis”, we expected both higher species richness and functional diversity in semi-open habitats at site level due to the simultaneous presence of forest and open habitats ground beetle species (Díaz et al., 2013). Indeed, we found that semi-open habitat ground beetle assemblages consisted of species associated with both open or forest habitats. Contrary to expectation, species richness and functional diversity of the semi-open were not higher than those of open habitats in both landscapes. We also found several forest species in the open habitats. Forest ground beetles have a higher tendency to disperse into adjacent open habitats while open habitat species are less likely to enter forests (e.g. Lacasella et al., 2015; Magura, 2017). Despite being recorded in low numbers in the open habitats, those forest species contribute to the increase of both species richness and functional diversity. Thus, the similar species richness and functional diversity values found between semi-open and open habitats likely result from the latter having both increased species richness and functional diversity due to the presence of few forest species. In contrast, forest habitats displayed lower species richness and functional diversity attributable to the absence of open habitats species.
Function of semi-open habitats for ground beetles
The presumed mechanism driving the high species richness or functional diversity in semi-open habitats is niche differentiation resulting from the higher habitat heterogeneity, which allows more species adapted to a range of different abiotic conditions to coexist. However, habitat patches are not isolated from one another and mass effects could also be an important contributor to species richness. Mass effects could increase assemblages’ species richness as a result of recurrent dispersal of individuals from adjacent habitats with a higher reproduction success (Mouquet and Loreau, 2002; Shmida and Wilson, 1985). Mass effects have been found to be neglectable compared to niche differentiation across similar habitats (Brooks et al., 2008). However, mass effects are expected to be maximal when differences between habitats are moderate such as between semi-open habitats and adjacent open and forest habitats (“intermediate difference hypothesis” Kunin, 1998). While ground beetle species have limited and varying dispersal abilities, our sampling locations within the semi-open habitats are well within the range of what could be covered by such taxa within one activity period (e.g. Baars, 1979; Rijnsdorp, 1980; Vermeulen, 1994). In semi-open habitats, neither the narrow glades of heathland or grassland-like patches nor the small wooded patches dominated by trees or shrubs are likely to support sufficient reproduction of most open or forest ground beetle species found in this study (Noordijk et al., 2011; Petit, 1994; Vermeulen, 1994). Thus, the continuous presence of many species in semi-open habitats at comparable abundance with either forest or open habitats is likely to depend on immigration from the adjacent source habitats. Consequently, mass effects rather than habitat heterogeneity are likely to be the main driver of the high species richness and functional diversity of semi-open habitats. In some instances, semi-open habitats will allow species preferring distinct habitats to exploit a greater proportion of the landscape, for example for foraging (e.g. Rijnsdorp, 1980; Taboada et al., 2011).
While the potential of semi-open habitats as corridors has rarely been investigated (but see Eggers et al., 2010; Travers et al., 2020), semi-open habitats can have positive effects on landscape connectivity. In our study, only a limited number of species (8 to 15%) tended to effectively avoid semi-open habitats, though, the high tendency of a large number of forest and open habitat species to enter semi-open habitats indicates that semi-open habitats can be also used for dispersal (Eggers et al., 2010). Of course, if intended to be used for dispersal in addition to habitat quality, semi-open habitat location and dimension will have to be adapted to the target species to avoid that they instead act as a sink.
We found that 15 to 18% of the analysed species were exclusively positively associated with semi-open habitats. Thus, semi-open habitat patches do appear to function as a habitat for some species preferring the particular environmental conditions prevailing in those patches. The addition of those species, to the above-mentioned forest and open habitat species, explains the higher species richness of our semi-open habitats. However, only a few species typically associated with forest edges (Heide: Leistus ferrugineus and Pterostichus quadrifoveolatus; Alb: Panagaeus bipustulatus) are likely to be truly dependent on semi-open habitats, while the remaining species being either eurytopic, open, or (more rarely) forest habitat specialist species (Gesellschaft für Angewandte Carabidologie, 2009). In concordance with our results, the semi-open Dehesa habitats have similarly been shown to predominantly host eurytopic and open habitat specialist species (Taboada et al., 2011, 2006a). In our case, many of the open habitat specialists are associated with regular disturbance as occurring in arable fields or ruderal habitats and their preference for semi-open habitat is likely caused by disturbances from both the effect of trees and shrub management and trampling from grazing animals. Similarly, Oedipoda caerulescens, a grasshopper also dependent on such frequently disturbed microsites was found in semi-open habitats (Rösch et al., 2019).
Some authors considered semi-open habitats as ecotones, given their intermediate nature combined with their high proportions of forest-open habitat edges (Bergmeier et al., 2010; Somay et al., 2021). However, studies focussing on ground beetles at forest-open habitat ecotones found assemblages highly similar to the forest assemblages (e.g. Heliölä et al., 2001; Lacasella et al., 2015) while we retrieved semi-open habitat assemblages which were distinct from the forest ones. In addition, studies on ecotones found an absence or a very low number of species that were associated with this particular environment. We comparatively found a higher number of species associated with semi-open habitats. Similar dissimilarities between forest ecotones and semi-open habitats have been reported for dung beetles (Somay et al., 2021). While forest edges entail a narrow, abrupt transition between open and forest habitats with environmental characteristics most similar to the forest interior (Heliölä et al., 2001), semi-open habitats, like those studied here, are much wider and appear to exhibit distinct environmental conditions (Travers et al., 2020). Consequently, semi-open habitats appear to be different from an ecotone and should likely not be regarded as such.
Difference between studied landscapes
Although generally consistent for both landscapes, our results differed in some respects. The ground beetle assemblages of semi-open habitats in the Heide were more similar to the ones in the forest while they were more similar to the open habitat assemblages in the Alb. A comparatively higher proportion of species associated with forest habitats in the Alb avoided the semi-open habitats while this proportion was lower in the Heide. The opposite was found for species associated with open habitats in both landscapes. Few studies compared assemblages from semi-open habitats in different locations. Studying dung beetles across Hungary, Somay et al. (2021) did not find a comparable alteration of the composition of semi-open habitat assemblages under similar climatic conditions and in the presence of an identical matrix. In contrast, a similar pattern was found for plants in our studied landscapes (some sites in common, Travers et al., 2020). Furthermore, the Ellenberg indicator values for the plant assemblages found in the semi-open habitats in the Heide indicated higher moisture than for the Alb (see Travers et al., 2020). The different climatic conditions between the two studied landscapes may be the driver for this pattern. The microclimatic conditions in the semi-open habitats under oceanic climate are likely to be more similar to those in forests; thus in the Heide, a larger number of forest-associated species, preferring on average cooler and moister conditions (Thiele, 1977), enter the semi-open habitats. Under sub-continental climate, in contrast, habitat conditions in the semi-open are likely to be more similar to those in open habitats (see Travers et al., 2020); thus in the Alb, a larger proportion of open-associated species, preferring dryer and sunnier conditions (Thiele, 1977), enter the semi-open habitats. In addition, dwarf shrubs such as Calluna vulgaris, which dominate the semi-open matrix in the Heide, were shown to alter microclimatic conditions by stabilising humidity and temperature levels within the vegetation layer (Gimingham, 1972). Thus, the matrix could contribute to reinforcing the difference between the two landscapes. In dry calcareous grasslands, such as those found in the Alb, no dwarf shrub species occur so humidity levels depend strictly on weather conditions. However, it remains to be investigated further by considering semi-open habitats with oceanic climate and grassland vegetation. Nevertheless, Somay’s expectation (Somay et al., 2021) that assemblages from semi-open habitats are intermediate between those of forest and open habitats independently from locations can now be extended to further taxa (ground beetles or plants) and presumably to the entire temperate region.
Implications for conservation
Any proposed solution for biodiversity conservation needs to be objectively evaluated (Godet and Devictor, 2018) and species richness has often been used to set conservation priorities (e.g. Gutiérrez et al., 2004). The high species richness found in semi-open habitats in combination with the occurrence of threatened species are repeatedly used to justify their conservation value (e.g. Gallé et al., 2017; Rösch et al., 2019; Somay et al., 2021). However, no or only limited consideration has been given to the process leading to the high species richness or the strength of the association between a given species and semi-open habitats. For ground beetles, we found that high species richness and functional diversity were presumably a result of only a few semi-open preferring species living in these habitats combined with those species regularly dispersing from both forest and open habitats into semi-open habitats due to mass effects. Thus, it appears, that neither species richness nor functional diversity are suitable tools for assessing the effectiveness of semi-open habitats as habitats with unique properties for biodiversity conservation. In addition, most threatened species, for which we evaluated habitat preference, were found to avoid semi-open habitats. Our findings question the conservation value of semi-open habitats for ground beetles in the temperate region.
Instead, our results underline the value of preserving the existing forest, heathland, and grassland habitats as such. The expected increase in semi-open habitats in the European temperate region through trophic rewilding or restoration and establishment of wood pastures may thus possibly be detrimental to many ground beetle species, including many threatened ones if they come to replace existing valuable habitats. Such measures should consequently be implemented with caution. However, as semi-open habitats appear to host a few specialized ground beetle species and considering that modern landscapes are mostly void of such habitat types, they do appear vital for the conservation of (at least) those few species. Also, the property of semi-open habitats to attract a large number of species from adjacent habitats could be beneficial for the conservation of ground beetles if such habitats are designed and established in a way so that they function as dispersal corridors.
Thus, both trophic rewilding or restoration and establishment of wood pastures, which have been suggested as solutions for insect conservation (Samways et al., 2020), might be especially effective for ground beetle conservation when they target enhancing connectivity rather than habitat creation. Given the similarity between our results and findings from the Mediterranean region for invertebrates (Díaz et al., 2013), a careful re-examination of already published evidence on the conservation value of semi-open habitats is required.
Availability of data
The dataset generated and analysed during the current study is available from the corresponding author on reasonable request.
Code Availability
Not applicable.
References
Antvogel H, Bonn A (2001) Environmental parameters and microspatial distribution of insects: a case study of carabids in an alluvial forest. Ecography (Cop) 24:470–482. https://doi.org/10.1111/j.1600-0587.2001.tb00482.x
Assmann T, Dormann W, Främbs H, Gürlich S, Handke K, Huk T, Sprick PT (2003) Rote Liste der in Niedersachsen und Bremen gefährdeten Sandlaufkäfer und Laufkäfer mit Gesamtartenverzeichnis. Informationsdienst Naturschutz Niedersachsen 23:70–95
Baars MA (1979) Patterns of movement of radioactive carabid beetles. Oecologia 44:125–140. https://doi.org/10.1007/bf00346411
Beinlich B, Plachter H (1995) Schutz und Entwicklung der Kalkmagerrasen der Schwäbischen Alb. Beihefte zu den Veröffentlichungen für Naturschutz und Landschaftspfl. Baden-Württembergs 83:1–520
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc 57:289–300. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x
Bergmeier E, Petermann J, Schröder E (2010) Geobotanical survey of wood-pasture habitats in Europe: diversity, threats and conservation. Biodivers Conserv 19:2995–3014. https://doi.org/10.1007/s10531-010-9872-3
Botta-Dukát Z (2005) Rao’s quadratic entropy as a measure of functional diversity based on multiple traits. J Veg Sci 16:533–540. https://doi.org/10.1111/j.1654-1103.2005.tb02393.x
Brooks DR, Perry JN, Clark SJ, Heard MS, Firbank LG, Holdgate R, Mason NS, Shortall CR, Skellern MP, Woiwod IP (2008) National-scale metacommunity dynamics of carabid beetles in UK farmland. J Anim Ecol 77:265–274. https://doi.org/10.1111/j.1365-2656.2007.01331.x
Cáceres M De, Legendre P (2009) Associations between species and groups of sites: indices and statistical inference. Ecology 90:3566–3574. https://doi.org/10.1890/08-1823.1
Cadotte MW, Carscadden K, Mirotchnick N (2011) Beyond species: functional diversity and the maintenance of ecological processes and services. J Appl Ecol 48:1079–1087. https://doi.org/10.1111/j.1365-2664.2011.02048.x
David J-F, Devernay S, Loucougaray G, Floc’h E Le (1999) Belowground biodiversity in a Mediterranean landscape: relationships between saprophagous macroarthropod communities and vegetation structure. Biodivers Conserv 8:753–767. https://doi.org/10.1023/A:1008842313832
Díaz M, Tietje WD, Barrett RH (2013) Effects of management on biological diversity and endangered species. In: Campos P, Huntsinger L, Pro O, Starrs JL, Diaz PF, Standiford M, Montero RB (eds) Mediterranean Oak Woodland Working Landscapes: Dehesas of Spain and Ranchlands of California. Springer Netherlands, Dordrecht, pp 213–243. https://doi.org/10.1007/978-94-007-6707-2_8
Eggers B, Matern A, Drees C, Eggers J, Härdtle W, Assmann T (2010) Value of semi-open corridors for simultaneously connecting open and wooded habitats: a case study with ground beetles. Conserv Biol 24:256–266. https://doi.org/10.1111/j.1523-1739.2009.01295.x
Gallé R, Urák I, Nikolett G-S, Hartel T (2017) Sparse trees and shrubs confers a high biodiversity to pastures: Case study on spiders from Transylvania. PLoS ONE 12:e0183465. https://doi.org/10.1371/journal.pone.0183465
Garbarino M, Bergmeier E (2014) Plant and vegetation diversity in European wood-pastures, in: European Wood-Pastures in Transition. Routledge, pp 131–149
Garrido P, Edenius L, Mikusiński G, Skarin A, Jansson A, Thulin C-G (2021) Experimental rewilding may restore abandoned wood-pastures if policy allows. Ambio 50:101–112. https://doi.org/10.1007/s13280-020-01320-0
Gerisch M, Agostinelli V, Henle K, Dziock F (2012) More species, but all do the same: contrasting effects of flood disturbance on ground beetle functional and species diversity. Oikos 121:508–515. https://doi.org/10.1111/j.1600-0706.2011.19749.x
Gesellschaft für Angewandte Carabidologie (2009) Lebensraumpräferenzen der Laufkäfer Deutschlands - Wissensbasierter Katalog. Angew. Carabidol. Suppl. V,1–45
Gimingham CH (1972) Ecology of Heathlands, Science Paperbacks. Chapman and Hall
Godet L, Devictor V (2018) What conservation does. Trends Ecol Evol 33:720–730. https://doi.org/10.1016/j.tree.2018.07.004
Gotelli NJ, Colwell RK (2001) Quantifying biodiversity: procedures and pitfalls in the measurement and comparison of species richness. Ecol Lett 4:379–391. https://doi.org/10.1046/j.1461-0248.2001.00230.x
Gutiérrez D, Menéndez R, Méndez M (2004) Habitat-based conservation priorities for carabid beetles within the Picos de Europa National Park, northern Spain. Biol Conserv 115:379–393. https://doi.org/10.1016/S0006-3207(03)00154-X
Hall SJG (2018) A novel agroecosystem: Beef production in abandoned farmland as a multifunctional alternative to rewilding. Agric Syst 167:10–16. https://doi.org/10.1016/j.agsy.2018.08.009
Heliölä J, Koivula M, Niemelä J (2001) Distribution of carabid beetles (Coleoptera, Carabidae) across a boreal forest-clearcut ecotone. Conserv Biol 15:370–377. https://doi.org/10.1046/j.1523-1739.2001.015002370.x
Homburg K, Homburg N, Schäfer F, Schuldt A, Assmann T (2014) Carabids.org – a dynamic online database of ground beetle species traits (Coleoptera, Carabidae). Insect Conserv Divers 7:195–205. https://doi.org/10.1111/icad.12045
Horák J, Pavlíček J, Kout J, Halda JP (2018) Winners and losers in the wilderness: response of biodiversity to the abandonment of ancient forest pastures. Biodivers Conserv 27:3019–3029. https://doi.org/10.1007/s10531-018-1585-z
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biometrical J 50:346–363. https://doi.org/10.1002/bimj.200810425
Jakobsson S, Wood H, Ekroos J, Lindborg R (2020) Contrasting multi-taxa functional diversity patterns along vegetation structure gradients of woody pastures. Biodivers Conserv 29:3551–3572. https://doi.org/10.1007/s10531-020-02037-y
Jost L, Choa A, Chazdon RL (2011) Compositional similarity and β (beta) diversity. In: Magurran AE, Mcgill BJ (eds) Biological Diversity: Frontiers in Measurement and Assessment. Oxford University Press, pp 66–84
Keienburg T, Prüter J (2004) Conservation and management of Central European lowland heathlands. Case study: Lüneburger Heide nature reserve, north-west Germany. Mitteilungen aus der NNA 15:1–64
Koivula M (2011) Useful model organisms, indicators, or both? Ground beetles (Coleoptera, Carabidae) reflecting environmental conditions. Zookeys 100:287–317. https://doi.org/10.3897/zookeys.100.1533
Kunin WE (1998) Biodiversity at the edge: A test of the importance of spatial “mass effects” in the Rothamsted Park Grass experiments. Proc. Natl. Acad. Sci. 95, 207–212. https://doi.org/10.1073/pnas.95.1.207
Lacasella F, Gratton C, De Felici S, Isaia M, Zapparoli M, Marta S, Sbordoni V (2015) Asymmetrical responses of forest and “beyond edge” arthropod communities across a forest–grassland ecotone. Biodivers Conserv 24:447–465. https://doi.org/10.1007/s10531-014-0825-0
Laliberté E, Legendre P (2010) A distance-based framework for measuring functional diversity from multiple traits. Ecology 91:299–305. https://doi.org/10.1890/08-2244.1
Lövei GL, Sunderland KD (1996) Ecology and behavior of ground beetles (Coleoptera: Carabidae). Annu Rev Entomol 41:231–256. https://doi.org/10.1146/annurev.en.41.010196.001311
MacArthur RH, MacArthur JW (1961) On bird species diversity. Ecology 42:594–598. https://doi.org/10.2307/1932254
Magura T (2017) Ignoring functional and phylogenetic features masks the edge influence on ground beetle diversity across forest-grassland gradient. For Ecol Manage 384:371–377. https://doi.org/10.1016/j.foreco.2016.10.056
Magura T, Lövei GL (2019) Environmental filtering is the main assembly rule of ground beetles in the forest and its edge but not in the adjacent grassland. Insect Sci 26:154–163. https://doi.org/10.1111/1744-7917.12504
Ministerium für Ernährung, Ländlichen Raum und Verbraucherschutz (2021) Landschaftspflege mit Schafen. URL https://www.landwirtschaft-bw.info/pb/,Lde/3650826_3651464_2305950_2316374_2316446 (accessed 12.20.21)
Moreno G, Gonzalez-Bornay G, Pulido F, Lopez-Diaz ML, Bertomeu M, Juárez E, Diaz M (2016) Exploring the causes of high biodiversity of Iberian dehesas: the importance of wood pastures and marginal habitats. Agrofor Syst 90:87–105. https://doi.org/10.1007/s10457-015-9817-7
Mouquet N, Loreau M (2002) Coexistence in metacommunities: the regional similarity hypothesis. Am Nat 159:420–426. https://doi.org/10.1086/338996
Müller-Motzfeld G, Freude H, Harde K, Lohse G, Klausnitzer B (eds) (2006): Die Käfer Mitteleuropas. Spektrum-Verlag, Heidelberg/Berlin, p. 521
Nolte D, Boutaud E, Kotze DJ, Schuldt A, Assmann T (2019) Habitat specialization, distribution range size and body size drive extinction risk in carabid beetles. Biodivers Conserv 28:1267–1283. https://doi.org/10.1007/s10531-019-01724-9
Noordijk J, Schaffers AP, Heijerman T, Sýkora KV (2011) Using movement and habitat corridors to improve the connectivity for heathland carabid beetles. J Nat Conserv 19:276–284. https://doi.org/10.1016/j.jnc.2011.05.001
Oksanen J, Blanchet GF, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2019) vegan: Community Ecology Package
Payton ME, Greenstone MH, Schenker N (2003) Overlapping confidence intervals or standard error intervals: what do they mean in terms of statistical significance? J Insect Sci 3:34. https://doi.org/10.1093/jis/3.1.34
Petit S (1994) Diffusion of forest carabid beetles in hedgerow network landscapes. In: Desender K, Dufrêne M, Loreau M, Luff ML, Maelfait J-P (eds) Carabid Beetles: Ecology and Evolution. Springer Netherlands, Dordrecht, pp 337–341. https://doi.org/10.1007/978-94-017-0968-2_51
Core Team R (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Renner K (1982) Coleopterenfänge mit Bodenfallen am Sandstrand der Ostseeküste, ein Beitrag zum Problem der Lockwirkung von Konservierungsmitteln. Faun-ökol Mitt 5:137–146
Rijnsdorp AD (1980) Pattern of movement in and dispersal from a dutch forest of Carabus problematicus Hbst. (Coleoptera, Carabidae) Oecologia 45:274–281. https://doi.org/10.1007/bf00346470.
Rösch V, Hoffmann M, Diehl U, Entling MH (2019) The value of newly created wood pastures for bird and grasshopper conservation. Biol Conserv 237:493–503. https://doi.org/10.1016/j.biocon.2019.07.036
Samways MJ, Barton PS, Birkhofer K, Chichorro F, Deacon C, Fartmann T, Fukushima CS, Gaigher R, Habel JC, Hallmann CA, Hill MJ, Hochkirch A, Kaila L, Kwak ML, Maes D, Mammola S, Noriega JA, Orfinger AB, Pedraza F, Pryke JS, Roque FO, Settele J, Simaika JP, Stork NE, Suhling F, Vorster C, Cardoso P (2020) Solutions for humanity on how to conserve insects. Biol Conserv 242:108427. https://doi.org/10.1016/j.biocon.2020.108427
Sandom CJ, Ejrnæs R, Hansen MDD, Svenning J-C (2014) High herbivore density associated with vegetation diversity in interglacial ecosystems. Proc. Natl. Acad. Sci. 111, 4162–4167. https://doi.org/10.1073/pnas.1311014111
Schmidt J, Trautner J, Müller-Motzfeld G (2016) Rote Liste und Gesamtartenliste der Laufkäfer Deutschlands. Naturschutz und Biol Vielfalt 70:139–204
Schulze KA, Rosenthal G, Peringer A (2018) Intermediate foraging large herbivores maintain semi-open habitats in wilderness landscape simulations. Ecol Modell 379:10–21. https://doi.org/10.1016/j.ecolmodel.2018.04.002
Shmida A, Wilson MV (1985) Biological determinants of species diversity. J Biogeogr 12:1–20. https://doi.org/10.2307/2845026
Söderström B, Svensson B, Vessby K, Glimskär A (2001) Plants, insects and birds in semi-natural pastures in relation to local habitat and landscape factors. Biodivers Conserv 10:1839–1863. https://doi.org/10.1023/A:1013153427422
Somay L, Szigeti V, Boros G, Ádám R, Báldi A (2021) Wood pastures: a transitional habitat between forests and pastures for dung beetles assemblages. Forests 12:25. https://doi.org/10.3390/f12010025
Svenning J-C, Pedersen PBM, Donlan CJ, Ejrnæs R, Faurby S, Galetti M, Hansen DM, Sandel B, Sandom CJ, Terborgh JW, Vera FWM (2016) Science for a wilder anthropocene: synthesis and future directions for trophic rewilding research. Proc. Natl. Acad. Sci. 113, 898–906. https://doi.org/10.1073/pnas.1502556112
Sverdrup-Thygeson A, Skarpaas O, Ødegaard F (2010) Hollow oaks and beetle conservation: the significance of the surroundings. Biodivers Conserv 19:837–852. https://doi.org/10.1007/s10531-009-9739-7
Taboada A, Kotze DJ, Salgado JM, Tárrega R (2011) The value of semi-natural grasslands for the conservation of carabid beetles in long-term managed forested landscapes. J Insect Conserv 15:573–590. https://doi.org/10.1007/s10841-010-9359-2
Taboada A, Kotze DJ, Tárrega R, Salgado JM (2006a) Traditional forest management: do carabid beetles respond to human-created vegetation structures in an oak mosaic landscape? For. Ecol Manage 237:436–449. https://doi.org/10.1016/j.foreco.2006.09.077
Taboada A, Kotze J, Salgado J, Tárrega R (2006b) The influence of habitat type on the distribution of carabid beetles in traditionally managed “dehesa” ecosystems in NW Spain. Entomol Fenn 17:284–295. https://doi.org/10.33338/ef.84348
Talvi T (1995) Carabid beetle assemblages (Coleoptera) in a wooded meadow and in the adjacent habitats on the Saaremaa Island. Estonia Entomol Fenn 6:169–175. https://doi.org/10.33338/ef.83854
Thiele H-U (1977) Carabid beetles in their environments: a study on habitat selection by adaptations in physiology and behaviour. Springer-Verlag, Berlin Germany
Trautner J, Bräunicke M, Kiechle J, Kramer M, Rietze J, Wolf-Schwenninger K, Schanowski A (2006) Rote Liste und Artenverzeichnis der Laufkäfer Baden-Württembergs (Coleoptera: Carabidae s. l.). Landesanstalt für Umweltschutz Baden-Württemberg
Travers E, Pitz WT, Fichtner A, Matthies D, Härdtle W (2020) The role of semi-open habitats as dispersal corridors for plant species of woodlands and open habitats. Appl Veg Sci 24:e12526. https://doi.org/10.1111/avsc.12526
Vera FWM (2009) Large-scale nature development–The Oostvaardersplassen. Br Wildl 20:28
Vera FWM (2000) Grazing ecology and forest history. CABI Publishing, Wallingford. https://doi.org/10.1079/9780851994420.0000
Vermeulen HJW (1994) Corridor function of a road verge for dispersal of stenotopic heathland ground beetles Carabidae. Biol Conserv 69:339–349. https://doi.org/10.1016/0006-3207(94)90433-2
Wickam H (2016) Elegant graphics for data analysis
Acknowledgements
We like to thank the landowners and the nature conservation authorities that provided us with the authorization to work on areas under their care. This study was funded by the federal agency for nature conservation (Bundesamt für Naturschutz, Grant Number: FKZ 3512 85 0100). Thanks to Wiebke Schuett for proofreading the manuscript. We thank the editor and two anonymous reviewers for their constructive comments, which helped improve the manuscript.
Funding
This study was funded by the federal agency for nature conservation (Bundesamt für Naturschutz, Grant Number: FKZ 3512 85 0100).
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Conceptualization: Thorsten Assmann; Methodology: Thorsten Assmann; Formal analysis: Estève Boutaud, Dorothea Nolte, Claudia Drees; Visualization: Estève Boutaud; Investigation: Estève Boutaud, Dorothea Nolte, Ingmar Harry; Writing - original draft preparation: Estève Boutaud; Writing - review and editing: Estève Boutaud, Dorothea Nolte, Ingmar Harry, Thorsten Assmann, Claudia Drees; Funding acquisition: Thorsten Assmann; Supervision: Thorsten Assmann, Claudia Drees.
Corresponding author
Ethics declarations
Conflict of interest/Competiting interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Communicated by Andreas Schuldt.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Boutaud, E., Nolte, D., Harry, I. et al. Conservation value of semi-open habitats for ground beetles (Coleoptera: Carabidae, Cicindelidae) in Central Europe. Biodivers Conserv 31, 1469–1489 (2022). https://doi.org/10.1007/s10531-022-02402-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-022-02402-z