Abstract
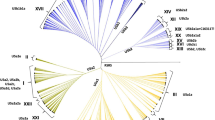
Deciphering the origin of invasive plant species, whether or not there have been multiple introductions, and genetic differentiations between invasive and native ranges is crucial in testing hypotheses underlying biological invasions. Here, we applied traditional population genetic analyses to unravel the phylogeographical relationships among invasive (North American) and native (North African, Mediterranean region, and Eurasian) range populations of Brassica nigra using chloroplast DNA. We sequenced chloroplast DNA intron (trnF–trnL) for 284 individuals representing 36 native and 15 invasive range populations of B. nigra. Thirty-two haplotypes were found over the whole data set. A similarity between the invasive range and native range populations in genetic diversity combined with results from analyses of molecular variance and gene genealogies suggest that invasive B. nigra populations were introduced from multiple sources in the native range. More generally, this study adds to the growing body of data on the genetic patterns involved in biological invasions that is crucial to our understanding of the evolutionary trajectories of invasive populations.

Similar content being viewed by others
References
Allendorf FW, Lundquist LL (2003) Introduction: population biology, evolution, and control of invasive species. Conserv Biol 17:24–30
Austerlitz F, Mariette S, Machon N et al (2000) Effects of colonization processes on genetic diversity: differences between annual plants and tree species. Genetics 154:1309–1321
Avise JC (2000) Phylogeography: the history and formation of species. Harvard University Press, Cambridge, MA
Bell DT, Muller CH (1973) Dominance of California annual grasslands by Brassica nigra. Am Midl Nat 90:277–299
Bossdorf O, Auge H, Lafuma L et al (2005) Phenotypic and genetic differentiation between native and introduced plant populations. Oecologia 144:1–11. doi:10.1007/s00442-005-0070-z
Chun YJ, Fumanal B, Laitung B, Bretagnolle F (2010) Gene flow and population admixture as the primary post-invasion processes in common ragweed (Ambrosia artemisiifolia) populations in France. New Phytol 185:1100–1107. doi:10.1111/j.1469-8137.2009.03129.x
Clement M, Posada D, Crandall KA (2000) TCS: a computer program to estimate gene genealogies. Mol Ecol 9:1657–1659. doi:10.1046/j.1365-294X.2000.01020.x
Dlugosch KM, Parker IM (2008) Founding events in species invasions: genetic variation, adaptive evolution, and the role of multiple introductions. Mol Ecol 17:431–449. doi:10.1111/j.1365-294X.2007.03538.x
Dobzhansky T (1952) Heterosis: a record of researches directed toward explaining and utilizing the vigor of hybrids, Nature and the origin of heterosis. Lowa State College Press, Ames, pp 218–223
Durka W, Bossdorf O, Prati D, Auge H (2005) Molecular evidence for multiple introductions of garlic mustard (Alliaria petiolata, Brassicaceae) to North America. Mol Ecol 14:1697–1706. doi:10.1111/j.1365-294X.2005.02521.x
Ellstrand NC, Schierenbeck KA (2000) Hybridization as a stimulus for the evolution of invasiveness in plants? Proc Natl Acad Sci U S A 97:7043–7050
Excoffier L, Smouse PE, Quattro JM (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics 131:479–491. doi:10.1007/s00424-009-0730-7
Excoffier L, Laval G, Schneider S (2005) Arlequin (version 3.0): an integrated software package for population genetics data analysis. Evol Bioinform Online 1:47–50. doi:10.1111/j.1755-0998.2010.02847.x
Facon B, Jarne P, Pointier JP, David P (2005) Hybridization and invasiveness in the freshwater snail Melanoides tuberculata: hybrid vigour is more important than increase in genetic variance. J Evol Biol 18:524–535. doi:10.1111/j.1420-9101.2005.00887.x
Feeny P, Rosenberry L (1982) Seasonal variation in the glucosinolate content of North American Brassica nigra and Dentaria species. Biochem Syst Ecol 10:23–32. doi:10.1016/0305-1978(82)90047-3
Freeland JR (2005) Molecular ecology. Wiley, New York
Fu YX (1997) Statistical tests of neutrality of mutations against population growth, hitchhiking and background selection. Genetics 147:915–925
Genton BJ, Shykoff JA, Giraud T (2005) High genetic diversity in French invasive populations of common ragweed, Ambrosia artemisiifolia, as a result of multiple sources of introduction. Mol Ecol 14:4275–4285. doi:10.1111/j.1365-294X.2005.02750.x
Gillis NK, Walters LJ, Fernandes FC, Hoffman EA (2009) Higher genetic diversity in introduced than in native populations of the mussel mytella charruana: evidence of population admixture at introduction sites. Divers Distrib 15:784–795. doi:10.1111/j.1472-4642.2009.00591.x
Hufbauer RA, Sforza R (2008) Multiple introductions of two invasive Centaurea taxa inferred from cpDNA haplotypes. Divers Distrib 14:252–261. doi:10.1111/j.1472-4642.2007.00424.x
Hulme PE (2009) Trade, transport and trouble: managing invasive species pathways in an era of globalization. J Appl Ecol 46:10–18. doi:10.1111/j.1365-2664.2008.01600.x
Kang M, Buckley YM, Lowe AJ (2007) Testing the role of genetic factors across multiple independent invasions of the shrub Scotch broom (Cytisus scoparius). Mol Ecol 16:4662–4673. doi:10.1111/j.1365-294X.2007.03536.x
Keller SR, Gilbert KJ, Fields PD, Taylor DR (2012) Bayesian inference of a complex invasion history revealed by nuclear and chloroplast genetic diversity in the colonizing plant, Silene latifolia. Mol Ecol 21:4721–4734. doi:10.1111/j.1365-294X.2012.05751.x
Kolbe JJ, Glor RE, Rodríguez Schettino L et al (2004) Genetic variation increases during biological invasion by a Cuban lizard. Nature 431:177–181. doi:10.1038/nature02807
Kolbe JJ, Glor RE, Schettino LR et al (2007) Multiple sources, admixture, and genetic variation in introduced Anolis lizard populations. Conserv Biol 21:1612–1625. doi:10.1111/j.1523-1739.2007.00826.x
Kolbe JJ, Larson A, Losos JB, de Queiroz K (2008) Admixture determines genetic diversity and population differentiation in the biological invasion of a lizard species. Biol Lett 4:434–437. doi:10.1098/rsbl.2008.0205
Lankau RA, Strauss SY (2008) Community complexity drives patterns of natural selection on a chemical defense of Brassica nigra. Am Nat 171:150–161. doi:10.1086/524959
Lavergne S, Molofsky J (2007) Increased genetic variation and evolutionary potential drive the success of an invasive grass. Proc Natl Acad Sci U S A 104:3883–3888. doi:10.1073/pnas.0607324104
Le Roux JJ, Brown GK, Byrne M et al (2011) Phylogeographic consequences of different introduction histories of invasive Australian Acacia species and Paraserianthes lophantha (Fabaceae) in South Africa. Divers Distrib 17:861–871. doi:10.1111/j.1472-4642.2011.00784.x
Le Roux JJ, Richardson DM, Wilson JRU, Ndlovu J (2013) Human usage in the native range may determine future genetic structure of an invasion: insights from Acacia pycnantha. BMC Ecol 13:37. doi:10.1186/1472-6785-13-37
Lee CE (2002) Evolutionary genetics of invasive species. Trends Ecol Evol 17:386–391. doi:10.1016/S0169-5347(02)02554-5
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451–1452. doi:10.1093/bioinformatics/btp187
Mack R, Simberloff D, Lonsdale MW et al (2000) Biotic invasions: causes, epidemiology, global consequences, and control. Ecol Appl 10:689–710
Meyerson LA, Cronin JT (2013) Evidence for multiple introductions of Phragmites australis to North America: detection of a new non-native haplotype. Biol Invasions 15:2605–2608. doi:10.1007/s10530-013-0491-2
Montarry J, Andrivon D, Glais I et al (2010) Microsatellite markers reveal two admixed genetic groups and an ongoing displacement within the French population of the invasive plant pathogen Phytophthora infestans. Mol Ecol 19:1965–1977. doi:10.1111/j.1365-294X.2010.04619.x
Nei M, Maruyama T, Chakraborty R (1975) The bottleneck effect and genetic variability in populations. Evolution (N Y) 29:1–10
Novak SJ, Mack RN (1993) Genetic variation in Bromus tectorum introduced populations. Heredity (Edinb) 71:167–176
Oduor AMO, Lankau RA, Strauss SY, Gomez JM (2011) Introduced Brassica nigra populations exhibit greater growth and herbivore resistance but less tolerance than native populations in the native range. New Phytol 191:536–544
Oduor AMO, Strauss SY, Garcia Y, et al (2013) Herbivores mediate different competitive and facilitative responses of native and invader populations of Brassica nigra. Ecology 94:2288–2298
Orians CM (2000) The effects of hybridization in plants on secondary chemistry: implications for the ecology and evolution of plant—Herbivore interactions. Am J Bot 87:1749–1756. doi:10.2307/2656824
Pimentel D, McNair S, Janecka J et al (2001) Economic and environmental threats of alien plant, animal and microbe invasions. Agric Ecosyst Environ 84:1–20
R Development Core Team (2013) A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rius M, Darling JA (2014) How important is intraspecific genetic admixture to the success of colonising populations? Trends Ecol Evol 29:233–242. doi:10.1016/j.tree.2014.02.003
Roman J, Darling JA (2007) Paradox lost: genetic diversity and the success of aquatic invasions. Trends Ecol Evol 22:454–464. doi:10.1016/j.tree.2007.07.002
Rosenthal DM, Ramakrishnan AP, Cruzan MB (2008) Evidence for multiple sources of invasion and intraspecific hybridization in Brachypodium sylvaticum (Hudson) Beauv. in North America. Mol Ecol 17:4657–4669. doi:10.1111/j.1365-294X.2008.03844.x
Sakai AK, Allendorf FW, Holt JS et al (2001) The population biology of invasive species. Annu Rev Ecol Syst 32:305–332
Smith CI, Pellmyr O, Althoff DM et al (2008) Pattern and timing of diversification in Yucca (Agavaceae): specialized pollination does not escalate rates of diversification. Proc Biol Sci 275:249–258. doi:10.1098/rspb.2007.1405
Stepien CA, Taylor CD, Dabrowska KA (2002) Genetic variability and phylogeographic patterns of a nonindigenous species invasion: a comparison of exotic versus native zebra and quagga mussel populations. J Evol Biol 15:314–328
Taberlet P, Gielly L, Pautou G, Bouvet J (1991) Universal primers for amplification of three non-coding regions of chloroplast DNA. Plant Mol Biol 17:1105–1109. doi:10.1007/BF00037152
Tajima F (1989) Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 123:585–595
Taylor DR, Keller SR (2007) Historical range expansion determines the phylogenetic diversity introduced during contemporary species invasion. Evolution (N Y) 61:334–345. doi:10.1111/j.1558-5646.2007.00037.x
Turner KG, Hufbauer RA, Rieseberg LH (2014) Rapid evolution of an invasive weed. New Phytol 202:309–321. doi:10.1111/nph.12634
Uller T, Leimu R (2011) Founder events predict changes in genetic diversity during human-mediated range expansions. Glob Chang Biol 17:3478–3485. doi:10.1111/j.1365-2486.2011.02509.x
Verhoeven KJF, Macel M, Wolfe LM, Biere A (2011) Population admixture, biological invasions and the balance between local adaptation and inbreeding depression. Proc R Soc B Biol Sci 278:2–8. doi:10.1098/rspb.2010.1272
Voisin M, Engel CR, Viard F (2005) Differential shuffling of native genetic diversity across introduced regions in a brown alga: aquaculture vs. maritime traffic effects. Proc Natl Acad Sci U S A 102:5432–5437. doi:10.1073/pnas.0501754102
Westman AL, Kresovich S (1999) Simple sequence repeat (SSR)—based marker variation in Brassica nigra genebank accessions and weed populations. Euphytica 109:85–92
Whitney KD, Gabler CA (2008) Rapid evolution in introduced species, “invasive traits” and recipient communities: challenges for predicting invasive potential. Divers Distrib 14:569–580. doi:10.1111/j.1472-4642.2008.00473.x
Williams DA, Overholt WA, Cuda JP, Hughes CR (2005) Chloroplast and microsatellite DNA diversities reveal the introduction history of Brazilian peppertree (Schinus terebinthifolius) in Florida. Mol Ecol 14:3643–3656. doi:10.1111/j.1365-294X.2005.02666.x
Zardus JD, Hadfield MG (2005) Multiple origins and incursions of the Atlantic barnacle Chthamalus proteus in the Pacific. Mol Ecol 14:3719–3733. doi:10.1111/j.1365-294X.2005.02701.x
Zhang B, Edwards OR, Kang L, Fuller SJ (2012) Russian wheat aphids (Diuraphis noxia) in China: native range expansion or recent introduction? Mol Ecol 21:2130–2144. doi:10.1111/j.1365-294X.2012.05517.x
Acknowledgments
We thank Yedra García for help with DNA extraction and Sharon Y. Strauss for comments on an earlier version of this manuscript. This study was supported by a Grant from the foundation Banco Bilbao Vizcaya Argentaria (BBVA-BIOCON06/064). We are grateful to North Central Regional Plant Introduction Station (NCRPIS)-USA, IPK, Gatersleben (Germany) and Real Jardín Botánico de Madrid (RJB-CSIC) for providing us with Brassica nigra seeds from their germplasm collections. We received additional B. nigra seeds from field collections done by Richard A. Lankau, Mirka Marcel,and Jeffrey K. Conner. A.M.O. Oduor received further financial support from a Ph.D. scholarship granted by Agencia Española de Cooperación Internacional para el Desarrollo (MAEC-AECID).
Author information
Authors and Affiliations
Corresponding author
Additional information
Chloroplast DNA (cpDNA) sequences of the region trnF–trnL have been published in the GenBank (accession numbers KF947115–KF947398).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Oduor, A.M.O., Gómez, J.M., Herrador, M.B. et al. Invasion of Brassica nigra in North America: distributions and origins of chloroplast DNA haplotypes suggest multiple introductions. Biol Invasions 17, 2447–2459 (2015). https://doi.org/10.1007/s10530-015-0888-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-015-0888-1