Abstract
Objective
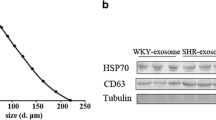
This study is to explore the exact roles of extracellular vesicle (EVs) miRNAs from brain microvascular pericytes in the pathogenesis of hypertension.
Results
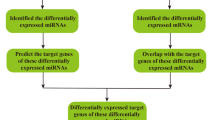
Forty-eight significantly differentially expressed miRNAs were identified, of which 17 were found to be upregulated and 31 were found to be downregulated in brain microvascular pericytes of spontaneous hypertensive rats, compared with that of normotension Wistar Kyoto rats. The GO enrichment analysis verified that the target genes were enriched in signaling pathways and molecular functions, such as metal ion binding, nucleotide binding and ATP binding. The KEGG analysis indicated that the target genes were enriched in Linoleic acid, alpha-linolenic acid and sphingolipid metabolism pathways.
Conclusions
Several EV derived miRNAs, such as miR-21-5p, let-7c-5p and let-7a-5p, were found to be abnormally expressed in brain microvascular pericytes obtained from spontaneous hypertensive rats, compared with that of normotension Wistar Kyoto rats. The results of our research provide more insights into the functional link between brain microvascular pericytes and the pathogenesis of hypertension.







Similar content being viewed by others
Abbreviations
- UTR:
-
Untranslated regions
- EVs:
-
Extracellular vesicles
- SCI:
-
Spinal cord injury
- TEM:
-
Transmission electron microscopy
- NTA:
-
Nanoparticle tracking analysis
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- SHR:
-
Spontaneous hypertensive rats
- WKY:
-
Wistar Kyoto rats
- GO:
-
Gene ontology
- BP:
-
Biology process
- CC:
-
Cellular component
- MF:
-
Molecular function
- SLC7A1:
-
Solute carrier family 7 member 1
References
Alipoor SD et al (2019) Serum exosomal miRNAs are associated with active pulmonary tuberculosis. Dis Markers 2019:1907426. https://doi.org/10.1155/2019/1907426
Armulik A, Genove G, Betsholtz C (2011) Pericytes: developmental, physiological, and pathological perspectives, problems, and promises. Dev Cell 21:193–215. https://doi.org/10.1016/j.devcel.2011.07.001
Bhome R, Del Vecchio F, Lee GH, Bullock MD, Primrose JN, Sayan AE, Mirnezami AH (2018) Exosomal microRNAs (exomiRs): small molecules with a big role in cancer. Cancer Lett 420:228–235. https://doi.org/10.1016/j.canlet.2018.02.002
Borodzicz S, Czarzasta K, Kuch M, Cudnoch-Jedrzejewska A (2015) Sphingolipids in cardiovascular diseases and metabolic disorders. Lipids Health Dis 14:55. https://doi.org/10.1186/s12944-015-0053-y
Carrick D et al (2018) Hypertension, microvascular pathology, and prognosis after an acute myocardial infarction. Hypertension 72:720–730. https://doi.org/10.1161/hypertensionaha.117.10786
Cengiz M et al (2015) Circulating miR-21 and eNOS in subclinical atherosclerosis in patients with hypertension. Clin Exp Hypertension 37:643–649. https://doi.org/10.3109/10641963.2015.1036064
Chen X, Liang H, Zhang J, Zen K, Zhang CY (2012) Secreted microRNAs: a new form of intercellular communication. Trends Cell Biol 22:125–132. https://doi.org/10.1016/j.tcb.2011.12.001
Ebrahimkhani S et al (2017) Exosomal microRNA signatures in multiple sclerosis reflect disease status. Sci Rep 7:14293. https://doi.org/10.1038/s41598-017-14301-3
Enright AJ, John B, Gaul U, Tuschl T, Sander C, Marks DS (2003) MicroRNA targets in Drosophila. Genome Biol 5:R1. https://doi.org/10.1186/gb-2003-5-1-r1
Fenger M, Linneberg A, Jeppesen J (2015) Network-based analysis of the sphingolipid metabolism in hypertension. Front Genet 6:84. https://doi.org/10.3389/fgene.2015.00084
Ferland-McCollough D, Slater S, Richard J, Reni C, Mangialardi G (2017) Pericytes, an overlooked player in vascular pathobiology. Pharmacol Ther 171:30–42. https://doi.org/10.1016/j.pharmthera.2016.11.008
Gaceb A, Barbariga M, Ozen I, Paul G (2018) The pericyte secretome: potential impact on regeneration. Biochimie 155:16–25. https://doi.org/10.1016/j.biochi.2018.04.015
Garcia-Contreras M, Shah SH, Tamayo A, Robbins PD, Golberg RB, Mendez AJ, Ricordi C (2017) Plasma-derived exosome characterization reveals a distinct microRNA signature in long duration type 1 diabetes. Sci Rep 7:5998. https://doi.org/10.1038/s41598-017-05787-y
Hannafon BN et al (2016) Plasma exosome microRNAs are indicative of breast cancer. Breast Cancer Res 18:90. https://doi.org/10.1186/s13058-016-0753-x
Hornick NI, Huan J, Doron B, Goloviznina NA, Lapidus J, Chang BH, Kurre P (2015) Serum exosome MicroRNA as a minimally-invasive early biomarker of AML. Sci Rep 5:11295. https://doi.org/10.1038/srep11295
Langmead B, Trapnell C, Pop M, Salzberg SL (2009) Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol 10:R25. https://doi.org/10.1186/gb-2009-10-3-r25
Li H et al (2016) MicroRNA-21 Lowers blood pressure in spontaneous hypertensive rats by upregulating mitochondrial translation. Circulation 134:734–751. https://doi.org/10.1161/circulationaha.116.023926
Liu X, Fortin K, Mourelatos Z (2008) MicroRNAs: biogenesis and molecular functions. Brain Pathol 18:113–121. https://doi.org/10.1111/j.1750-3639.2007.00121.x
Liu X, Yuan W, Yang L, Li J, Cai J (2019) miRNA profiling of exosomes from spontaneous hypertensive rats using next-generation sequencing. J Cardiovasc Trans Res 12:75–83. https://doi.org/10.1007/s12265-017-9784-7
Majzunova M, Dovinova I, Barancik M, Chan JY (2013) Redox signaling in pathophysiology of hypertension. J Biomed Sci 20:69. https://doi.org/10.1186/1423-0127-20-69
Mao X, Cai T, Olyarchuk JG, Wei L (2005) Automated genome annotation and pathway identification using the KEGG Orthology (KO) as a controlled vocabulary. Bioinformatics 21:3787–3793. https://doi.org/10.1093/bioinformatics/bti430
Murthy M, Kurz T, O'Shaughnessy KM (2017) WNK signalling pathways in blood pressure regulation. Cell Mol Life Sci 74:1261–1280. https://doi.org/10.1007/s00018-016-2402-z
Ren W et al (2019) Exosomal miRNA-107 induces myeloid-derived suppressor cell expansion in gastric cancer. Cancer Manage Res 11:4023–4040. https://doi.org/10.2147/cmar.S198886
Sekar D, Shilpa BR, Das AJ (2017) Relevance of microRNA 21 in different types of hypertension. Curr Hypertens Rep 19:57. https://doi.org/10.1007/s11906-017-0752-z
Serne EH, de Jongh RT, Eringa EC, Ijzerman RG, Stehouwer CD (2007) Microvascular dysfunction: a potential pathophysiological role in the metabolic syndrome. Hypertension 50:204–211. https://doi.org/10.1161/hypertensionaha.107.089680
Shen E, Diao X, Wei C, Wu Z, Zhang L, Hu B (2010) MicroRNAs target gene and signaling pathway by bioinformatics analysis in the cardiac hypertrophy. Biochem Biophys Res Commun 397:380–385. https://doi.org/10.1016/j.bbrc.2010.05.116
Spijkers LJ et al (2011) Hypertension is associated with marked alterations in sphingolipid biology: a potential role for ceramide. PLoS ONE 6:e21817. https://doi.org/10.1371/journal.pone.0021817
Sugimachi K et al (2015) Identification of a bona fide microRNA biomarker in serum exosomes that predicts hepatocellular carcinoma recurrence after liver transplantation. Br J Cancer 112:532–538. https://doi.org/10.1038/bjc.2014.621
Tang Y, Zhao Y, Song X, Song X, Niu L, Xie L (2019) Tumor-derived exosomal miRNA-320d as a biomarker for metastatic colorectal cancer. J Clin Lab Anal 33:e23004. https://doi.org/10.1002/jcla.23004
Tsukamoto I, Sugawara S (2018) Low levels of linoleic acid and alpha-linolenic acid and high levels of arachidonic acid in plasma phospholipids are associated with hypertension. Biomed Reps 8:69–76. https://doi.org/10.3892/br.2017.1015
Villarroya-Beltri C, Gutierrez-Vazquez C, Sanchez-Madrid F, Mittelbrunn M (2013) Analysis of microRNA and protein transfer by exosomes during an immune synapse. Methods Mol Biol 1024:41–51. https://doi.org/10.1007/978-1-62703-453-1_4
Wu Q, Yuan X, Han R, Zhang H, Xiu R (2019) Epitranscriptomic mechanisms of N6-methyladenosine methylation regulating mammalian hypertension development by determined spontaneously hypertensive rats pericytes. Epigenomics. https://doi.org/10.2217/epi-2019-0148
Xin H et al (2012) Exosome-mediated transfer of miR-133b from multipotent mesenchymal stromal cells to neural cells contributes to neurite outgrowth. Stem Cells 30:1556–1564. https://doi.org/10.1002/stem.1129
Yuan X, Wu Q, Liu X, Zhang H, Xiu R (2018) Transcriptomic profile analysis of brain microvascular pericytes in spontaneously hypertensive rats by RNA-Seq. Am J Transl Res 10:2372–2386
Yuan X et al (2019) Exosomes derived from pericytes improve microcirculation and protect blood-spinal cord barrier after spinal cord injury in mice. Front Neurosci 13:319. https://doi.org/10.3389/fnins.2019.00319
Zhang J, Li S, Li L, Li M, Guo C, Yao J, Mi S (2015) Exosome and exosomal microRNA: trafficking, sorting, and function. Genomics Proteomics Bioinf 13:17–24. https://doi.org/10.1016/j.gpb.2015.02.001
Acknowledgements
This study was supported by the innovation fund of the Chinese Academy of Medical Sciences and Peking Union Medical College (Grant Nos. 3332014006 and 3332015123), the CAMS Initiative for Innovative Medicine (CAMS-I2M) (Grant No. 2016-I2M-3-006) and National Natural Science Foundation of China (Grant No. 81801433).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wu, Q., Yuan, X., Li, B. et al. Differential miRNA expression analysis of extracellular vesicles from brain microvascular pericytes in spontaneous hypertensive rats. Biotechnol Lett 42, 389–401 (2020). https://doi.org/10.1007/s10529-019-02788-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-019-02788-x