Abstract
Objective
To explore the influence of ultra-low carbon dioxide partial pressure (pCO2) on the monoclonal antibody (mAb) N-glycosylation profile in Chinese hamster ovary (CHO) cell culture.
Results
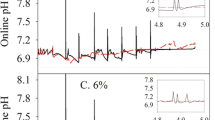
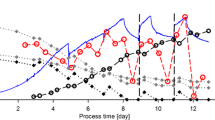
In fed-batch bioreactor cultures, lowering the pCO2 in the medium (< 25 mmHg) via increasing headspace aeration decreased the cell viability and mAb production in CHO cells. Additionally, mAb galactosylation under low pCO2 was approximately 27.45 ± 2.13%, noticeably higher than that observed under normal pCO2 (21.36 ± 1.66%) at harvest. However, all of the relevant intracellular nucleotide sugar concentrations were dramatically decreased to approximately 50% of the levels found under normal pCO2 on day 7. Real-time PCR revealed that the upregulation of galactosylation-related glycosyltransferase genes and substrate transporter genes played a critical role in the improved galactosylation under the ultra-low pCO2 condition.
Conclusions
In the bioreactor culture processes, ultra-low pCO2 demonstrated a positive effect on mAb galactosylation.



Similar content being viewed by others
References
Andersen DC, Bridges T, Gawlitzek M, Hoy C (2000) Multiple cell culture factors can affect the glycosylation of Asn-184 in CHO-produced tissue-type plasminogen activator. Biotechnol Bioeng 70:25–31
Batra J, Rathore AS (2016) Glycosylation of monoclonal antibody products: current status and future prospects. Biotechnol Prog 32:1091–1102
Brunner M, Fricke J, Kroll P, Herwig C (2017) Investigation of the interactions of critical scale-up parameters (pH, pO2 and pCO2) on CHO batch performance and critical quality attributes. Bioprocess Biosyst Eng 40:251–263
Chee Furng Wong D, Tin Kam Wong K, Tang Goh L, Kiat Heng C, Gek Sim Yap M (2005) Impact of dynamic online fed-batch strategies on metabolism, productivity and N-glycosylation quality in CHO cell cultures. Biotechnol Bioeng 89:164–177
Dietmair S, Timmins NE, Gray PP, Nielsen LK, Kromer JO (2010) Towards quantitative metabolomics of mammalian cells: development of a metabolite extraction protocol. Anal Biochem 404:155–164
Fan Y et al (2015) Amino acid and glucose metabolism in fed-batch CHO cell culture affects antibody production and glycosylation. Biotechnol Bioeng 112:521–535
Feng H-T, Wong N, Wee S, Lee M (2008) Simultaneous determination of 19 intracellular nucleotides and nucleotide sugars in Chinese Hamster ovary cells by capillary electrophoresis. J Chromatogr B 870:131–134
Kimura R, Miller WM (1997) Glycosylation of CHO-derived recombinant tPA produced under elevated pCO2. Biotechnol Prog 13:311–317
Liu H, Nowak C, Shao M, Ponniah G, Neill A (2016) Impact of cell culture on recombinant monoclonal antibody product heterogeneity. Biotechnol Prog 32:1103–1112
Mostafa SS, Gu X (2003) Strategies for improved dCO2 removal in large-scale fed-batch cultures. Biotechnol Prog 19:45–51
Saldova R et al (2014) Association of N-glycosylation with breast carcinoma and systemic features using high-resolution quantitative UPLC. J Proteome Res 13:2314–2327
Schmelzer AE, Miller WM (2002) Hyperosmotic stress and elevated pCO2 alter monoclonal antibody charge distribution and monosaccharide content. Biotechnol Prog 18:346–353
Yoon SK, Ahn Y-H, Han K (2001) Enhancement of recombinant erythropoietin production in CHO cells in an incubator without CO2 addition. Cytotechnology 37:119–132
Zanghi JA, Schmelzer AE, Mendoza TP, Knop RH, Miller WM (1999) Bicarbonate concentration and osmolality are key determinants in the inhibition of CHO cell polysialylation under elevated pCO2 or pH. Biotechnol Bioeng 65(2):182–191
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 21406066, 21106045), the National High Technology Research and Development Program of China (863 Program) (No. 2012AA02A303), the Fundamental Research Funds for the Central Universities (No. 22221818014), and the Development Funds of Shanghai Zhangjiang National Independent Innovation Demonstration Zone (No. ZJ2015-ZD-002).
Supporting information
Supplementary Table 1—Primers used in real-time PCR to quantify the expression levels of specific N-galactosylation-related genes.
Supplementary Fig. 1—The glycosylation profiles of 2-AB-labeled N-glycans separated using UPLC under normal pCO2 (0.05 vvm, red) and low pCO2 (0.4 vvm, black) on day 11. Symbols: square, N-acetylglucosamine; circle, mannose; rhombus, galactose; triangle, fucose; star, sialic acid.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10529_2018_2586_MOESM1_ESM.tif
The glycosylation profiles of 2-AB-labeled N-glycans separated using UPLC under normal pCO2 (0.05 vvm, red) and low pCO2 (0.4 vvm, black) on day 11. Symbols: square, N-acetylglucosamine; circle, mannose; rhombus, galactose; triangle, fucose; star, sialic acid. Supplementary material 1 (TIFF 43375 kb)
Rights and permissions
About this article
Cite this article
Wang, C., Wang, J., Chen, M. et al. Ultra-low carbon dioxide partial pressure improves the galactosylation of a monoclonal antibody produced in Chinese hamster ovary cells in a bioreactor. Biotechnol Lett 40, 1201–1208 (2018). https://doi.org/10.1007/s10529-018-2586-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-018-2586-4