Abstract
Objectives
As 5-aminolevulinic acid synthase (ALAS), the key enzyme for 5-aminolevulinic acid (ALA) synthesis, is unstable, we have sought to find thermostable ALASs from thermophilic organisms.
Results
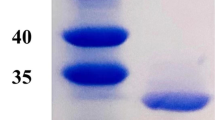
Three ALASs from thermophiles Geobacillus thermoglucosidasius (GT-ALAS), Laceyella sacchari (LS-ALAS) and Pseudomonas alcaliphila (PA-ALAS) were purified and characterized. All enzymes were more stable than two previously studied ALASs from Rhodopseudomonas palustris and Rhodobacter sphaeroides. There was almost no activity change after 60 h at 37 °C for the three thermostable enzymes. This contrasts with the other two enzymes which lost over 90 % activities in just 1 h. Furthermore, the specific activity of LS-ALAS (7.8 U mg−1) was also higher than any previously studied ALASs.
Conclusions
Thermostable ALASs were found in thermophilic organisms and this paves the way for developing cell free processes for enzymatic production of ALA from bulk chemicals succinate and glycine.





Similar content being viewed by others
References
Bolt EL, Kryszak L, Zeilstra-Ryalls J, Shoolingin-Jordan PM, Warren MJ (1999) Characterization of the Rhodobacter sphaeroides 5-aminolaevulinic acid synthase isoenzymes, HemA and HemT, isolated from recombinant Escherichia coli. Eur J Biochem 265:290–299
Burnham BF (1970) δ-Aminolevulinic acid synthase (Rhodopseudomonas sphaeroides). Methods Enzymol 17:195–204
Choi HP, Hong JW, Rhee KH, Sung HC (2004) Cloning, expression, and characterization of 5-aminolevulinic acid synthase from Rhodopseudomonas palustris KUGB306. FEMS Microbiol Lett 236:175–181
Fu W, Lin J, Cen P (2010) Expression of a hemA gene from Agrobacterium radiobacter in a rare codon optimizing Escherichia coli for improving 5-aminolevulinate production. Appl Biochem Biotechnol 160:456–466
Kaufholz AL, Hunter GA, Ferreira GC, Lendrihas T, Hering V, Layer G, Jahn M, Jahn D (2013) Aminolaevulinic acid synthase of Rhodobacter capsulatus: high-resolution kinetic investigation of the structural basis for substrate binding and catalysis. Biochem J 451:205–216
Lee DH, Jun WJ, Shin DH, Cho HY, Hong BS (2005) Effect of culture conditions on production of 5-aminolevulinic acid by recombinant Escherichia coli. Biosci Biotechnol Biochem 69:470–476
Lin J, Fu W, Cen P (2009) Characterization of 5-aminolevulinate synthase from Agrobacterium radiobacter, screening new inhibitors for 5-aminolevulinate dehydratase from Escherichia coli and their potential use for high 5-aminolevulinate production. Bioresour Technol 100:2293–2297
Lou JW, Zhu L, Wu MB, Yang LR, Lin JP, Cen PL (2014) High level soluble expression of the hemA gene from Rhodobacter capsulatus and comparative study of its enzymatic properties. J Zhejiang Univ Sci B 15:491–499
Mauzerall D, Granick S (1956) The occurrence and determination of delta-amino-levulinic acid and porphobilinogen in urine. J Biol Chem 219:435–446
Zhang YH (2010) Production of biocommodities and bioelectricity by cell-free synthetic enzymatic pathway biotransformations: challenges and opportunities. Biotechnol Bioeng 105:663–677
Zhang L, Chen J, Chen N, Sun J, Zheng P, Ma Y (2013) Cloning of two 5-aminolevulinic acid synthase isozymes HemA and HemO from Rhodopseudomonas palustris with favorable characteristics for 5-aminolevulinic acid production. Biotechnol Lett 35:763–768
Zhang J, Kang Z, Chen J, Du G (2015) Optimization of the heme biosynthesis pathway for the production of 5-aminolevulinic acid in Escherichia coli. Sci Rep 5:8584
Zhou X, Szeker K, Janocha B, Bohme T, Albrecht D, Mikhailopulo IA, Neubauer P (2013) Recombinant purine nucleoside phosphorylases from thermophiles: preparation, properties and activity towards purine and pyrimidine nucleosides. FEBS J 280:1475–1490
Acknowledgments
This work was supported by the National Key Basic Research Program of China (973 Program) (Nos. 2012CB725203, 2011CBA00804), National High Technology Research and Development Program of China (863 Program) (No. 2012AA022103) and Natural Science Foundation of Tianjin (No. 12JCYBJC33000).
Supporting information
Supplementary Table 1—Bacterial strains and plasmids used in this study.
Supplementary Table 2—The NCBI protein IDs of the studied enzymes.
Supplementary Fig. 1—Comparison of amino acid sequences of ALASs from different organisms.
Supplementary Fig. 2—Initial enzymatic reaction rates measured at various substrate concentrations.
Supplementary Fig. 3—Influence of PLP addition during the purification process on the specific activities of ALASs.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Meng, Q., Zhang, Y., Ma, C. et al. Purification and functional characterization of thermostable 5-aminolevulinic acid synthases. Biotechnol Lett 37, 2247–2253 (2015). https://doi.org/10.1007/s10529-015-1903-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-015-1903-4