Abstract
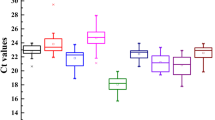
Validated internal controls are prerequisites to accurately normalize gene expression levels. Here, 14 candidate reference genes in Panax notoginseng were characterized. Primer specificity and amplification efficiency were evaluated for each gene. Candidates were subjected to transcript quantification in the root, fibrous root, rhizome, leaf, receptacle, pedicel, and fruit tissues. Expression stability (M value) and normalization factor variation (Vn/Vn+1) were determined by geNorm. 26S-2 and ACT-2 exhibited the highest expression stability among the tissues. Gene expression of dammarenediol synthase was accurately detected after normalization to 26S-2 and ACT-2 was performed. Results were consistent when each or both of 26S-2 and ACT-2 were applied as internal control. Hence, this study provides useful information to normalize gene expression accurately in the tissue-specific transcripts of P. notoginseng.



Similar content being viewed by others
References
Galli V, da Silva Messias R, Dos Anjos ESSD, Rombaldi CV (2013) Selection of reliable reference genes for quantitative real-time polymerase chain reaction studies in maize grains. Plant Cell Rep 32:1869–1877
He F, Zhu Y, Zhang Y (2008) Identification and characterization of differentially expressed genes involved in pharmacological activities of roots of Panax notoginseng during plant growth. Plant Cell Rep 27:923–930
Hub F, Zhong J (2008) Jasmonic acid mediates gene transcription of ginsenoside biosynthesis in cell cultures of Panax notoginseng treated with chemically synthesized 2-hydroxyethyl jasmonate. Process Biochem 43:113–118
Jia XH, Wang CQ, Liu JH, Li XW, Wang X, Shang MY, Cai SQ, Zhu S, Komatsu K (2012) Comparative studies of saponins in 1–3-year-old main roots, fibrous roots, and rhizomes of Panax notoginseng, and identification of different parts and growth-year samples. J Nat Med 67:339–349
Kundu A, Patel A, Pal A (2013) Defining reference genes for qPCR normalization to study biotic and abiotic stress responses in Vigna mungo. Plant Cell Rep 32:1647–1658
Lau AJ, Toh DF, Chua TK, Pang YK, Woo SO, Koh HL (2009) Antiplatelet and anticoagulant effects of Panax notoginseng: comparison of raw and steamed Panax notoginseng with Panax ginseng and Panax quinquefolium. J Ethnopharmacol 125:380–386
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods 25:402–408
Lovdal T, Lillo C (2009) Reference gene selection for quantitative real-time PCR normalization in tomato subjected to nitrogen, cold, and light stress. Anal Biochem 387:238–242
Mafra V, Kubo KS, Alves-Ferreira M, Ribeiro-Alves M, Stuart RM, Boava LP, Rodrigues CM, Machado MA (2012) Reference genes for accurate transcript normalization in citrus genotypes under different experimental conditions. PLoS ONE 7:e31263
Mallona I, Lischewski S, Weiss J, Hause B, Egea-Cortines M (2010) Validation of reference genes for quantitative real-time PCR during leaf and flower development in Petunia hybrida. BMC Plant Biol 10:4
Mehdi Khanlou K, Van Bockstaele E (2012) A critique of widely used normalization software tools and an alternative method to identify reliable reference genes in red clover (Trifolium pratense L.). Planta 236:1381–1393
Monteiro F, Sebastiana M, Pais MS, Figueiredo A (2013) Reference gene selection and validation for the early responses to downy mildew infection in susceptible and resistant Vitis vinifera cultivars. PLoS ONE 8:e72998
Singh K, Raizada J, Bhardwaj P, Ghawana S, Rani A, Singh H, Kaul K, Kumar S (2004) 26S rRNA-based internal control gene primer pair for reverse transcription-polymerase chain reaction-based quantitative expression studies in diverse plant species. Anal Biochem 335:330–333
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol Res 3:0034.1–0034.11
Wang M, Wang Q, Zhang B (2013) Evaluation and selection of reliable reference genes for gene expression under abiotic stress in cotton (Gossypium hirsutum L.). Gene 530:44–50
Yang CY, Wang J, Zhao Y, Shen L, Jiang X, Xie ZG, Liang N, Zhang L, Chen ZH (2010) Anti-diabetic effects of Panax notoginseng saponins and its major anti-hyperglycemic components. J Ethnopharmacol 130:231–236
Zhang G, Zhao M, Song C, Luo A, Bai J, Guo S (2012) Characterization of reference genes for quantitative real-time PCR analysis in various tissues of Anoectochilus roxburghii. Mol Biol Rep 39:5905–5912
Zhu X, Li X, Chen W, Chen J, Lu W, Chen L, Fu D (2012) Evaluation of new reference genes in papaya for accurate transcript normalization under different experimental conditions. PLoS ONE 7:e44405
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (NSFC, Grant No. 81260619) and the Scientific Research Project of Guangxi Educational Department (Grant No. 201204LX256). We thank Dr. Hui Yao (Peking Union Medical College, China), Ms. Juan Wang and Ms. Xiaoli Fan (Guilin Medical University, China) for their assistance in the experiments.
Supporting information
Supplementary Table 1—The RT-PCR primer sequences, amplicon size (bp), length of cDNA sequence and accession no. of the candidate genes in P. notoginseng
Supplementary Table 2—The slope, Y-Inter, R2 and amplification efficiency (Eff %) for candidate genes
Supplementary Figure 1—Standard curves of six reference genes and DDS gene were generated from qRT-PCR analyses
Supplementary Figure 2—Average expression stability (M) of six selected reference genes measured by geNorm values
Supplementary Figure 3—Optimal number of reference genes for normalization determined by the pairwise variation V
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
The online version of this article contains supplementary material, which is available to authorized users. The slope, Y-Inter, R 2 and amplification efficiency (Eff %) for candidate genes, the RT-PCR primer sequences, amplicon size, and accession number of the candidate genes, standard curves of genes and the optimal number of reference genes for normalization are available as Supporting Information.
Rights and permissions
About this article
Cite this article
Wu, Q., Ma, X., Zhang, K. et al. Identification of reference genes for tissue-specific gene expression in Panax notoginseng using quantitative real-time PCR. Biotechnol Lett 37, 197–204 (2015). https://doi.org/10.1007/s10529-014-1643-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-014-1643-x