Abstract
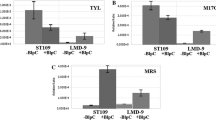
Streptococcus thermophilus B59671 produces a bacteriocin with anti-pediococcal activity, but genes required for its production are not characterized. Genome sequencing of S. thermophilus has identified a genetic locus encoding a quorum sensing (QS) system that regulates production of class II bacteriocins. However, in strains possessing this gene cluster, production of bacteriocin like peptides (Blp) was only observed when excess pheromone was provided. PCR analysis revealed this strain possessed blpC, which encodes the 30-mer QS pheromone. To investigate if BlpC regulates bacteriocin production in S. thermophilus B59671, an integrative vector was used to replace blpC with a gene encoding for kanamycin resistance and the resulting mutant did not inhibit the growth of Pediococcus acidilactici. Constitutive expression of blpC from a shuttle vector restored the bacteriocin production, confirming the blp gene cluster is essential for bacteriocin activity in S. thermophilus B59671.



Similar content being viewed by others
References
Bolotin A, Quinquis B, Renault P, Sorokin A et al (2004) Complete sequence and comparative genome analysis of the dairy bacterium Streptococcus thermophilus. Nat Biotechnol 22:1554–1558
de Saizieu A, Gardes C, Flint N, Wagner C et al (2000) Microarray-based identification of a novel Streptococcus pneumoniae regulon controlled by an autoinduced peptide. J Bacteriol 182:4696–4703
Du L, Somkuti GA, Renye JA Jr (2012) Molecular analysis of the bacteriocin-encoding plasmid pDGL1 from Enterococcus durans and genetic characterization of the durancin GL locus. Microbiology 158:1523–1532
Federal Register (1988) Nisin preparation: affirmation of GRAS status as a direct human food ingredient. Fed Reg 54:11247–11251
Fontaine L, Boutry C, Guedon E, Guillot A et al (2007) Quorum-sensing regulation of the production of Blp bacteriocins in Streptococcus thermophilus. J Bacteriol 189:7195–7205
Gilbreth SE, Somkuti GA (2005) Thermophilin 110: a bacteriocin of Streptococcus thermophilus ST110. Curr Microbiol 51:175–182
Hols P, Hancy F, Fontaine L, Grossiord B et al (2005) New insights in the molecular biology and physiology of Streptococcus thermophilus revealed by comparative genomics. FEMS Microbiol Rev 29:435–463
Ivanova I, Miteva V, Stefanova Ts, Pantev A et al (1998) Characterization of a bacteriocin produced by Streptococcus thermophilus 81. Int J Food Microbiol 42:147–158
Kabuki T, Uenishi H, Seto Y, Yoshioka T, Nakajima H (2009) A unique lantibiotic, thermophilin 1277, containing a disulfide bridge and two thioether bridges. J Appl Microbiol 106:853–862
Kang X, Ling N, Sun G, Zhou Q, Zhang L, Sheng Q (2012) Complete genome sequence of Streptococcus thermophilus strain MN-ZLW-002. J Bacteriol 194:4428–4429
Makarova K, Slesarev A, Wolf Y, Sorokin A et al (2006) Comparative genomics of the lactic acid bacteria. Proc Natl Acad Sci 103:15611–15616
Marciset O, Jeronimus-Stratingh MC, Mollet B, Poolman B (1997) Thermophilin 13, a nontypical antilisterial poration complex bacteriocin, that functions without a receptor. J Biol Chem 272:14277–14284
Mathot AG, Beliard E, Thuault D (2003) Streptococcus thermophilus 580 produces a bacteriocin potentially suitable for inhibition of Clostridium tyrobutyricum in hard cheese. J Dairy Sci 86:3068–3074
Mills S, Stanton C, Hill C, Ross RP (2011) New developments and applications of bacteriocins and peptides in foods. Annu Rev Food Sci Technol 2:299–329
Renye JA Jr, Somkuti GA (2012) Vector-mediated chromosomal integration of the glutamate decarboxylase gene in Streptococcus thermophilus. Biotechnol Lett 34:549–555
Sambrook J, Maniatis T, Fritsch EF (1989) Molecular cloning a laboratory manual, 2nd edn. Cold Spring Harbor Lab Press, New York
Shatalin KY, Neyfakh AA (2005) Efficient gene inactivation in Bacillus anthracis. FEMS Microbiol Lett 245:315–319
Solaiman DK, Somkuti GS (1993) Shuttle vectors developed from Streptococcus thermophilus native plasmid. Plasmid 30:67–78
Somkuti GA, Solaiman DK (1997) STP2201, a chromosomal promoter sequence of Streptococcus thermophilus. Curr Microbiol 35:180–185
Somkuti GS, Steinberg DH (1988) Genetic transformation of Streptococcus thermophilus by electroporation. Biochimie 70:579–585
Stougaard P, Molin S (1981) Vertical dye-buoyant density gradients for rapid analysis and preparation of plasmid DNA. Anal Biochem 118:191–193
Sun Z, Chen X, Wang J, Zhao W et al (2011) Complete genome sequence of Streptococcus thermophilus strain ND03. J Bacteriol 193:793–794
Villani F, Pepe O, Mauriello G, Salzano G et al (1995) Antilisterial activity of thermophilin 347, a bacteriocin produced by Streptococcus thermophilus. Int J Food Microbiol 25:179–190
Ward DJ, Somkuti GA (1995) Characterization of a bacteriocin produced by Streptococcus thermophilus ST134. Appl Microbiol Biotechnol 43:330–335
Acknowledgments
We thank D. Steinberg, M. Toht and E. Lawrence (St. Joseph’s University) for technical assistance with plasmid and strain development, and D. Needleman for assistance with nucleic acid sequencing.
Author information
Authors and Affiliations
Corresponding author
Additional information
Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture. USDA is an equal opportunity provider and employer.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Renye, J.A., Somkuti, G.A. BlpC-regulated bacteriocin production in Streptococcus thermophilus . Biotechnol Lett 35, 407–412 (2013). https://doi.org/10.1007/s10529-012-1095-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-012-1095-0