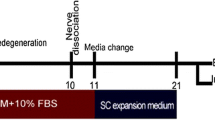
Abstract
Schwann cells (SCs) can be used to repair both the peripheral and central nervous systems. Therefore, establishment of a procedure to obtain activated, highly proliferative SCs, in an appropriate time for clinical applications, is a prerequisite. Purification is complicated by contamination with fibroblasts which often become the predominant cell type in an in vitro SC culture. This study describes a novel and efficient method to enrich SCs by utilizing the differential detachment properties of the two cell types. In culture, cells were treated with two different media and the chelator, EGTA, which detached SCs faster than fibroblasts and allowed for easy isolation of SCs. Within seven days, high yields of SCs with a purity of greater than 99% were achieved. This was confirmed by immunostaining characterization and flow-cytometric analyses using an antibody against the p75 low affinity nerve growth factor receptor (p75LNGFR). The entire procedure was completed in approximately 21 days. This method has the advantage of being technically easier, faster, and more efficient than other previously described methods. An SC culture that was about 99% homogenous was achieved.


Similar content being viewed by others
References
Assouline JG, Bosch EP, Lim R (1983) Purification of rat Schwann cells from cultures of peripheral nerve: an immunoselective method using surfaces coated with anti-immunoglobulin antibodies. Brain Res 277:389–392
Barakat DJ, Gaglani SM, Neravetla SR et al (2005) Survival, integration, and axon growth support of glia transplanted into the chronically contused spinal cord. Cell Transplant 14:225–240
Calderon-Martinez D, Garavito Z, Spinel C, Hurtado H (2002) Schwann cell-enriched cultures from adult human peripheral nerve: a technique combining short enzymatic dissociation and treatment with cytosine arabinoside (Ara-C). J Neurosci Methods 114:1–8
Casella GT, Bunge RP, Wood PM (1996) Improved method for harvesting human Schwann cells from mature peripheral nerve and expansion in vitro. Glia 17:327–338
Harvey AR, Plant GW, Tan MM (1995) Schwann cells and the regrowth of axons in the mammalian CNS: a review of transplantation studies in the rat visual system. Clin Exp Pharmacol Physiol 22:569–579
Hedayatpour A, Sobhani A, Bayati V et al (2007) A method for isolation and cultivation of adult Schwann cells for nerve conduit. Arch Iran Med 10:474–480
Ide C (1996) Peripheral nerve regeneration. Neurosci Res 25:101–121
Jin YQ, Liu W, Hong TH, Cao Y (2008) Efficient Schwann cell purification by differential cell detachment using multiplex collagenase treatment. J Neurosci Methods 170:140–148
Keilhoff G, Fansa H, Smalla KH et al (2000) Neuroma: a donor-age independent source of human Schwann cells for tissue engineered nerve grafts. Neuroreport 11:3805–3809
Komiyama T, Nakao Y, Toyama Y et al (2003) A novel technique to isolate adult Schwann cells for an artificial nerve conduit. J Neurosci Methods 122:195–200
Kromer LF, Cornbrooks CJ (1985) Transplants of Schwann cell cultures promote axonal regeneration in the adult mammalian brain. Proc Natl Acad Sci USA 82:6330–6334
Mackinnon SE, Dellon AL (1990) A study of nerve regeneration across synthetic (Maxon) and biologic (collagen) nerve conduits for nerve gaps up to 5 cm in the primate. J Reconstr Microsurg 6:117–121
Manent J, Oguievetskaia K, Bayer J et al (2003) Magnetic cell sorting for enriching Schwann cells from adult mouse peripheral nerves. J Neurosci Methods 123:167–173
Mauritz C, Grothe C, Haastert K (2004) Comparative study of cell culture and purification methods to obtain highly enriched cultures of proliferating adult rat Schwann cells. J Neurosci Res 77:453–461
Morrissey TK, Bunge RP, Kleitman N (1995a) Human Schwann cells in vitro. I. Failure to differentiate and support neuronal health under co-culture conditions that promote full function of rodent cells. J Neurobiol 28:171–189
Morrissey TK, Kleitman N, Bunge RP (1995b) Human Schwann cells in vitro. II. Myelination of sensory axons following extensive purification and heregulin-induced expansion. J Neurobiol 28:190–201
Oda Y, Okada Y, Katsuda S et al (1989) A simple method for the Schwann cell preparation from newborn rat sciatic nerves. J Neurosci Methods 28:163–169
Pannunzio ME, Jou IM, Long A et al (2005) A new method of selecting Schwann cells from adult mouse sciatic nerve. J Neurosci Methods 149:74–81
Pearse DD, Marcillo AE, Oudega M et al (2004) Transplantation of Schwann cells and olfactory ensheathing glia after spinal cord injury: does pretreatment with methylprednisolone and interleukin-10 enhance recovery? J Neurotrauma 21:1223–1239
Rothblum K, Stahl RC, Carey DJ (2004) Constitutive release of alpha4 type V collagen N-terminal domain by Schwann cells and binding to cell surface and extracellular matrix heparan sulfate proteoglycans. J Biol Chem 279:51282–51288
Verdu E, Rodriguez FJ, Gudino-Cabrera G et al (2000) Expansion of adult Schwann cells from mouse predegenerated peripheral nerves. J Neurosci Methods 99:111–117
Vroemen M, Weidner N (2003) Purification of Schwann cells by selection of p75 low affinity nerve growth factor receptor expressing cells from adult peripheral nerve. J Neurosci Methods 124:135–143
Weinstein DE, Wu R (1999) Isolation and purification of primary Schwann cells. In: Crawley JN, Gerfen CR, Rogawski MA, Sibley DR, Skolnick P, Wray S (eds) Current protocols in neuroscience. Wiley, New York, pp 3.17.11–3.17.19
Acknowledgment
This study was supported by a grant from Royan Institute, Iran.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Niapour, A., Karamali, F., Karbalaie, K. et al. Novel method to obtain highly enriched cultures of adult rat Schwann cells. Biotechnol Lett 32, 781–786 (2010). https://doi.org/10.1007/s10529-010-0230-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-010-0230-z