Abstract
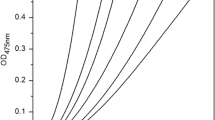
A novel tyrosinase inhibitor, an arbutin derivative having undecylenic acid at the 6-position of its glucose moiety, was enzymatically synthesized. Its inhibitory activity was studied in vitro by using catechol and phenol as substrates. The IC50 value of the arbutin ester on tyrosinase using catechol (4 × 10−4 M) was 1% of that when arbutin (4 × 10−2 M) was used. Using phenol, IC50 of the arbutin ester (3 × 10−4 M) as substrate was 10% of that of arbutin (3 × 10−3 M). These results suggest that the arbutin ester inhibits the latter part of the tyrosinase reaction, which consists of hydroxylation and oxidation.





Similar content being viewed by others
References
Espin JC, Varon R, Fnoll LG, Gilabert MA, Garcia-Ruiz PA, Tudela J, Garcia-Canovas FG (2000) Kinetics characterization of the substrate specificity and mechanism of mushroom tyrosinase. Eur J Biochem 267:1270–1279
Hearing VJ, Tsukamoto K (1991) Enzymatic control of pigmentation in mammals. FEBS J 5:2902–2909
Hori I, Nihei K, Kubo I (2004) Structural criteria for depigmenting mechanism of arbutin. Phytother Res 18:475–479
Jin YH, Lee SJ, Chung MH, Park J, Park YI, Cho TH, Lee SK (1999) Aloesin and arbutin inhibit tyrosinase activity in a synergistic manner via a different action mechanism. Arch Pharm Res 22:232–236
Kadokawa J, Nishikura T, Tagaya H, Fukuoka N (2001) Synthesis of arbutin derivative with a kojic acid moiety. Nippon Kagaku Kaishi (Japanese): 533–535
Maeda K, Fukuda M (1996) Arbutin: Mechanism of its depigmenting action in human melanocyte culture. J Pharmacol Exp Therap 278:765–769
Marmol V, Beermann F (1996) Tyrosinase and related proteins in mammalian pigmentation. FEBS Lett 381:165–168
McLain N, Ascanio R, Baker C, Strohaver RA, Dolan JW (2000) Undecylenic acid inhibits morphogenesis of Candida albicans. Antimicrob Agents Chemother 44:2873–2875
Mellors A, Tappel AL (1966) The inhibition of mitochondrial peroxidation by ubiquinone and ubiquinol. J Biol Chem 241:4353–4356
Morikawa H, Kasai R, Otsuka H, Hirata E, Shinzato T, Aramoto M, Takeda Y (2004) Terpenic and phenolic glycosides from leaves of Breynia officinalis HEMSL. Chem Pharm Bull 52:1086–1090
Nakajima N, Ishihara K, Matsumura S, Hamada H, Nakamura K, Furuya T (1997) Lipase-catalyzed synthesis of arbutin cinnamate in an organic solvent and application of transesterification to stabilize plant pigment. Biosci Biotechnol Biochem 61:1921–1928
Raku T, Kitagawa M, Shimakawa H, Tokiwa Y (2003) Enzymatic synthesis of hydrophilic undecylenic acid esters and their biodegradability. Biotechnol Lett 16:161–166
Ros JR, Rodriguez-Lopez JN, Garcia-Canovas F (1993) Effect of L-ascorbic acid on the monophenolase activity of tyrosinase. Biochem J 295:309–312
Rzepecki M, Waite JH (1989) A chromogenic assay for catecholoxidases based on the addition of L-proline to quinones. Anal Biochem 179:375–381
Shafran SD, Sacks SL, Aoki FY, Tyrell DL, Schlech WF 3rd, Mendelson J, Rosenthal D, Gill MJ, Bader RL, Chang I (1997) Topical undecylenic acid for herpes simplex labialis: a multicenter, placebo-controlled trial. J Infect Dis 176:78–83
Takeuchi M, Takahashi H, Uritani I (1998) Mechanism on hydroxylation of L-tyrosine by mushroom tyrosinase. Nippon Shokuhin Kagaku Kaishi (Japanese) 45:415–419
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tokiwa, Y., Kitagawa, M. & Raku, T. Enzymatic synthesis of arbutin undecylenic acid ester and its inhibitory effect on mushroom tyrosinase. Biotechnol Lett 29, 481–486 (2007). https://doi.org/10.1007/s10529-006-9267-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-006-9267-4