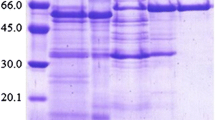
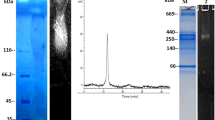
Abstract
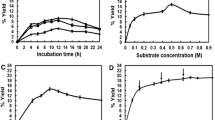
Hydroquinone-α-glucoside was synthesised from hydroquinone and maltose as glucosyl donor by transglucosylation in a water system with α-glucosidase from baker’s yeast. Only one phenolic –OH group was α-anomer-selectively glucosylated. The optimum conditions for transglucosylation reaction were at 30 °C for 20 h with 50 mM hydroquinone and 1.5 M maltose in 100 mM sodium citrate/phosphate buffer at pH 5.5. The glucoside was obtained at 0.6 mg/ml with a 4.6% molar yield with respect to hydroquinone.
Similar content being viewed by others
References
M Funayama H Arakawa R Yamamoto T Nishino T Shin S Murao (1995) ArticleTitleEffects of α- and β-arbutin on activity of tyrosinases from mushroom, and mouse melanoma Biosci. Biotech. Biochem. 59 143–144 Occurrence Handle1:CAS:528:DyaK2MXjslKrtbs%3D
S Kitao H Sekine (1994) ArticleTitleα-D-Glucosyl transfer to phenolic compounds by sucrose phosphorylase from Leuconostoc mesenteroides and production of α-arbutin Biosci. Biotech. Biochem. 58 38–42 Occurrence Handle1:CAS:528:DyaK2cXitFWhu70%3D
H Nakagawa M Yoshiyama S Shimura K Kirimura S Usami (1998) ArticleTitleAnomer-selective glucosylation of l-menthol by yeast α-glucosidase Biosci. Biotechnol. Biochem. 62 1332–1336 Occurrence Handle10.1271/bbb.62.1332 Occurrence Handle1:CAS:528:DyaK1cXltFGht7c%3D Occurrence Handle9720215
BR Needleman JH Federoff R Eccleshall B Buchferer J Marmur (1978) ArticleTitlePurification and characterization of an α-glucosidase from Saccharomyces carlsbergensis Biochemistry 17 4657–4661 Occurrence Handle10.1021/bi00615a011 Occurrence Handle1:CAS:528:DyaE1MXit1er Occurrence Handle365220
T Nishimura T Kometani H Takii Y Terada S Okada (1994) ArticleTitlePurification and some properties of α-amylase from Bacillus subtilis X-23 that glucosylates phenolic compounds such as hydroquinone J. Ferm. Bioeng. 78 31–36 Occurrence Handle10.1016/0922-338X(94)90174-0 Occurrence Handle1:CAS:528:DyaK2cXmsV2ju7g%3D
MJ Petit F Paquet MJ Beau (1991) ArticleTitleSyntheses of β-2-deoxy-D-glycosides assisted by glycosidases Tetrahedron Lett. 32 6125–6128 Occurrence Handle10.1016/0040-4039(91)80769-3 Occurrence Handle1:CAS:528:DyaK38XlvFOksw%3D%3D
R Prodanović S Jovanović Z Vujčić (2001) ArticleTitleImmobilization of invertase on a new type of macroporous glycidyl methacrylate Biotechnol. Lett. 23 1171–1174 Occurrence Handle10.1023/A:1010560911400
M Rodde M Franssen A Padt R Boom (2003) ArticleTitlePerspectives for the industrial enzymatic production of glycosides Biotechnol. Prog. 19 1391–1402 Occurrence Handle10.1021/bp030038q Occurrence Handle14524698
T Sato H Nakagawa J Kurosu K Yoshida T Tsugane S Shimura K Kirimura K Kino S Usami (2000) ArticleTitleα-Anomer-selective glucosylation of (+)-catechin by the crude enzyme, showing glucosyl transfer activity, of Xanthomonas campestris WU-9701 J. Biosci. Bioeng 90 625–630 Occurrence Handle10.1263/jbb.90.625 Occurrence Handle1:CAS:528:DC%2BD3MXhs12nurs%3D
KH Shin YJ Kong DJ Lee HT Lee (2000) ArticleTitleSyntheses of hydroxybenzyl-α-glucosides by amyloglucosidase-catalyzed transglycosylation Biotechnol. Lett. 22 321–325 Occurrence Handle10.1023/A:1005699104814 Occurrence Handle1:CAS:528:DC%2BD3cXitFems7g%3D
D Sladić M Gašić (1994) ArticleTitleEffects of iron(II) compounds on the amount of DNA damage in Friend erythroleukemia cells induced by avarol Role of hydroxyl radicals J Serb. Chem. Soc. 59 915–920
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Prodanović, R.M., Milosavić, N.B., Sladić, D. et al. Synthesis of hydroquinone-α-glucoside by α-glucosidasefrom baker’s yeast. Biotechnol Lett 27, 551–554 (2005). https://doi.org/10.1007/s10529-005-2880-9
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10529-005-2880-9