Abstract
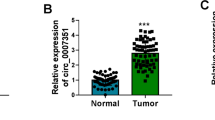
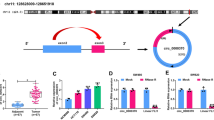
Colorectal cancer (CRC) remains one of the most frequent neoplasms of digestive tract worldwide. Circular RNAs (circRNAs) have been identified to serve crucial regulatory roles in the pathogenesis of human cancers. However, the role and regulatory mechanism of circ_0000467 in the progression of CRC are still unclear. The expression levels of circ_0000467, microRNA-330-5p (miR-330-5p), and tyrosine receptor kinase 3 (TYRO3) were measured by quantitative real-time polymerase chain reaction (qRT-PCR). The interaction between miR-330-5p and circ_0000467 or TYRO3 was validated by dual-luciferase reporter assay and RNA immunoprecipitation (RIP) assay. Xenograft tumor assay and Immunohistochemistry (IHC) assay were implemented to analyze CRC tumor growth in vivo. Circ_0000467 was a stable circRNA and was highly expressed in CRC tumor tissues and cells. Silencing of circ_0000467 could inhibit the proliferation, migration, invasion, and glycolysis and accelerated the apoptosis of CRC cells in vitro and hindered tumor growth in vivo. Mechanistically, circ_0000467 directly interacted with miR-330-5p and circ_0000467 depletion inhibited CRC cell malignant progression by regulating miR-330-5p. Furthermore, TYRO3 was a target of miR-330-5p and circ_0000467 upregulated TYRO3 expression by sponging miR-330-5p. Moreover, TYRO3 overexpression counteracted the inhibitory effect of miR-330-5p overexpression or circ_0000467 knockdown on CRC cell progression. Altogether, circ_0000467 knockdown suppressed CRC cell malignant development through modulating the miR-330-5p/TYRO3 network, providing a novel molecular target of CRC therapy.







Similar content being viewed by others
References
Avilla E, Guarino V, Visciano C, Liotti F, Svelto M, Krishnamoorthy G, Franco R, Melillo RM (2011) Activation of TYRO3/AXL tyrosine kinase receptors in thyroid cancer. Can Res 71:1792–1804
Berindan-Neagoe I, Monroig Pdel C, Pasculli B, Calin GA (2014) MicroRNAome genome: a treasure for cancer diagnosis and therapy. CA 64:311–336
Bhuyan R, Bagchi A (2020) Prediction of the differentially expressed circRNAs to decipher their roles in the onset of human colorectal cancers. Gene 762:145035
Castells A (2016) Hereditary forms of colorectal cancer. Gastroenterol Hepatol 39(Suppl 1):62–67
Chen LL, Yang L (2015) Regulation of circRNA biogenesis. RNA Biol 12:381–388
Chen L, Yang X, Zhao J, Xiong M, Almaraihah R, Chen Z, Hou T (2020) Circ_0008532 promotes bladder cancer progression by regulation of the miR-155-5p/miR-330-5p/MTGR1 axis. J Exp Clin Cancer Res 39:94
Chen H, Wu C, Luo L, Wang Y, Peng F (2021) circ_0000467 promotes the proliferation, metastasis, and angiogenesis in colorectal cancer cells through regulating KLF12 expression by sponging miR-4766-5p. Open Med (warsaw, Poland) 16:1415–1427
Chien CW, Hou PC, Wu HC, Chang YL, Lin SC, Lin SC, Lin BW, Lee JC, Chang YJ, Sun HS, Tsai SJ (2016) Targeting TYRO3 inhibits epithelial-mesenchymal transition and increases drug sensitivity in colon cancer. Oncogene 35:5872–5881
Connell LC, Mota JM, Braghiroli MI, Hoff PM (2017) The rising incidence of younger patients with colorectal cancer: questions about screening, biology, and treatment. Curr Treat Options Oncol 18:23
Cunningham D, Atkin W, Lenz HJ, Lynch HT, Minsky B, Nordlinger B, Starling N (2010) Colorectal cancer. Lancet 375:1030–1047
Du J, Xu J, Chen J, Liu W, Wang P, Ye K (2020) circRAE1 promotes colorectal cancer cell migration and invasion by modulating miR-338-3p/TYRO3 axis. Cancer Cell Int 20:430
Dufour F, Silina L, Neyret-Kahn H, Moreno-Vega A, Krucker C, Karboul N, Dorland-Galliot M, Maille P, Chapeaublanc E, Allory Y, Stransky N, Haegel H, Menguy T, Duong V, Radvanyi F, Bernard-Pierrot I (2019) TYRO3 as a molecular target for growth inhibition and apoptosis induction in bladder cancer. Br J Cancer 120:555–564
Ekyalongo RC, Mukohara T, Kataoka Y, Funakoshi Y, Tomioka H, Kiyota N, Fujiwara Y, Minami H (2013) Mechanisms of acquired resistance to insulin-like growth factor 1 receptor inhibitor in MCF-7 breast cancer cell line. Invest New Drugs 31:293–303
Garzon R, Marcucci G, Croce CM (2010) Targeting microRNAs in cancer: rationale, strategies and challenges. Nat Rev Drug Discov 9:775–789
Jiang D, Xie X, Lu Z, Liu L, Qu Y, Wu S, Li Y, Li G, Wang H, Xu G (2020) Establishment of a colorectal cancer-related MicroRNA-mRNA regulatory network by microarray and bioinformatics. Front Genet 11:560186
Kabir TD, Ganda C, Brown RM, Beveridge DJ, Richardson KL, Chaturvedi V, Candy P, Epis M, Wintle L, Kalinowski F, Kopp C, Stuart LM, Yeoh GC, George J, Leedman PJ (2018) A microRNA-7/growth arrest specific 6/TYRO3 axis regulates the growth and invasiveness of sorafenib-resistant cells in human hepatocellular carcinoma. Hepatology 67:216–231
Kasinski AL, Slack FJ (2011) Epigenetics and genetics. MicroRNAs en route to the clinic: progress in validating and targeting microRNAs for cancer therapy. Nat Rev Cancer 11:849–864
Kristensen LS, Hansen TB, Veno MT, Kjems J (2018) Circular RNAs in cancer: opportunities and challenges in the field. Oncogene 37:555–565
Kristensen LS, Andersen MS, Stagsted LVW, Ebbesen KK, Hansen TB, Kjems J (2019) The biogenesis, biology and characterization of circular RNAs. Nat Rev Genet 20:675–691
Lai FJ, Yu H, Xie YY, He N (2021) Circ_0000317/microRNA-520g/HOXD10 axis affects the biological characteristics of colorectal cancer. Kaohsiung J Med Sci 37:951–963
Lee C (2015) Overexpression of Tyro3 receptor tyrosine kinase leads to the acquisition of taxol resistance in ovarian cancer cells. Mol Med Rep 12:1485–1492
Li Y, Zang H, Zhang X, Huang G (2020) circ_0136666 Facilitates the progression of colorectal cancer via miR-383/CREB1 Axis. Cancer Manag Res 12:6795–6806
Liu HZ, Shan TD, Han Y, Liu XS (2020) Silencing long non-coding RNA CASC9 inhibits colorectal cancer cell proliferation by acting as a competing endogenous RNA of miR-576-5p to regulate AKT3. Cell Death Discov 6:115
Lu J, Zhang PY, Xie JW, Wang JB, Lin JX, Chen QY, Cao LL, Huang CM, Li P, Zheng CH (2019) Hsa_circ_0000467 promotes cancer progression and serves as a diagnostic and prognostic biomarker for gastric cancer. J Clin Lab Anal 33:e22726
Mansoori B, Mohammadi A, Naghizadeh S, Gjerstorff M, Shanehbandi D, Shirjang S, Najafi S, Holmskov U, Khaze V, Duijf PHG, Baradaran B (2020) miR-330 suppresses EMT and induces apoptosis by downregulating HMGA2 in human colorectal cancer. J Cell Physiol 235:920–931
Mo WL, Jiang JT, Zhang L, Lu QC, Li J, Gu WD, Cheng Y, Wang HT (2020) Circular RNA hsa_circ_0000467 promotes the development of gastric cancer by competitively binding to microRNA miR-326-3p. Biomed Res Int 2020:4030826
Myers KV, Amend SR, Pienta KJ (2019) Targeting Tyro3, Axl and MerTK (TAM receptors): implications for macrophages in the tumor microenvironment. Mol Cancer 18:94
Panda AC (2018) Circular RNAs Act as miRNA sponges. Adv Exp Med Biol 1087:67–79
Qin A, Qian W (2018) MicroRNA-7 inhibits colorectal cancer cell proliferation, migration and invasion via TYRO3 and phosphoinositide 3-kinase/protein B kinase/mammalian target of rapamycin pathway suppression. Int J Mol Med 42:2503–2514
Shao T, Wang G, Chen H, Xie Y, Jin X, Bai J, Xu J, Li X, Huang J, Jin Y, Li Y (2019) Survey of miRNA-miRNA cooperative regulation principles across cancer types. Brief Bioinform 20:1621–1638
Smart SK, Vasileiadi E, Wang X, DeRyckere D, Graham DK (2018) The emerging role of TYRO3 as a therapeutic target in cancer. Cancers 10:474
Wang G, Li Y, Zhu H, Huo G, Bai J, Gao Z (2020) Circ-PRKDC facilitates the progression of colorectal cancer through miR-198/DDR1 regulatory axis. Cancer Manag Res 12:12853–12865
Xie L, Pan Z (2021) Circular RNA circ_0000467 regulates colorectal cancer development via miR-382-5p/EN2 axis. Bioengineered 12:886–897
Xu W, Jiang H, Zhang F, Gao J, Hou J (2017) MicroRNA-330 inhibited cell proliferation and enhanced chemosensitivity to 5-fluorouracil in colorectal cancer by directly targeting thymidylate synthase. Oncol Lett 13:3387–3394
Xu D, Wu Y, Wang X, Hu X, Qin W, Li Y, Wang Y, Zhang Z, Lu S, Sun T, Wu Z, Fu D, Fu B, Zhang J, Chen Q, Wei M, Zhao L, Wu H (2020) Identification of functional circRNA/miRNA/mRNA regulatory network for exploring prospective therapy strategy of colorectal cancer. J Cell Biochem. https://doi.org/10.1002/jcb.29703
Xu S, Lei SL, Liu KJ, Yi SG, Yang ZL, Yao HL (2020) circSFMBT1 promotes pancreatic cancer growth and metastasis via targeting miR-330–5p/PAK1 axis. Cancer Gene Ther. https://doi.org/10.1038/s41417-020-00215-2
Yoo HI, Kim BK, Yoon SK (2016) MicroRNA-330-5p negatively regulates ITGA5 expression in human colorectal cancer. Oncol Rep 36:3023–3029
Yu X, He Y, Wang Y, Tang Y, Guo Y, Yuan J, Bai J, Yao T, Wu X (2020) MicroRNA-199a-5p suppresses the cell growth of colorectal cancer by targeting oncogene Caprin1. 3 Biotech 10:453
Yuan W, Peng S, Wang J, Wei C, Ye Z, Wang Y, Wang M, Xu H, Jiang S, Sun D, Dai C, Jiang L, Li X (2019) Identification and characterization of circRNAs as competing endogenous RNAs for miRNA-mRNA in colorectal cancer. PeerJ 7:e7602
Zeng K, Wang S (2020) Circular RNAs: the crucial regulatory molecules in colorectal cancer. Pathol Res Pract 216:1861
Zhang HD, Jiang LH, Sun DW, Hou JC, Ji ZL (2018) CircRNA: a novel type of biomarker for cancer. Breast Cancer (tokyo, Japan) 25:1–7
Zhao G, Dai GJ (2020) Hsa_circRNA_000166 promotes cell proliferation, migration and invasion by regulating miR-330-5p/ELK1 in colon cancer. Onco Targets Ther 13:5529–5539
Acknowledgements
None.
Funding
None.
Author information
Authors and Affiliations
Contributions
HH designed and performed the research; ZC and XZ analyzed the data; YH wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Data Availability
Not applicable.
Ethical Approval
Written informed consents were obtained from all participants and this study was permitted by the Ethics Committee of Huizhou Municipal Central Hospital.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10528_2021_10171_MOESM1_ESM.tif
Supplementary file1 (TIF 5992 KB) The effects of miR-382-5p and miR-330-5p on the malignant behaviors of CRC cells. HCT116 and LoVo cells transfected with anti-miR-NC, anti-miR-330-5p, anti-miR-382-5p, and anti-miR-330-5p + anti-miR-382-5p. (A and B) CCK8 assay was used to evaluate the proliferation of transfected cells. (C) Cell migration ability was detected by transwell assay. *P < 0.05
10528_2021_10171_MOESM2_ESM.tif
Supplementary file2 (TIF 450 KB) The protein levels of miR-330-5p-targeted genes in CRC cells with miR-330-5p transfection. (A and B) The protein levels of ERBB4, FZD5, TYRO3, PAK2, and XIAP in HCT116 and LoVo cells transfected with miR-NC or miR-330-5p were detected by western blot assay. *P < 0.05, **P < 0.01, ***P < 0.001
10528_2021_10171_MOESM3_ESM.tif
Supplementary file3 (TIF 7681 KB) Circ_0000467 regulated the progression of CRC by regulating TYRO3 expression. HCT116 and LoVo cells were transfected with si-NC, si-circ_0000467#2, si-circ_0000467#2 + pcDNA, or si-circ_0000467#2 + TYRO3, respectively. (A and B) The proliferation of transfected cells was detected by CCK8 assay. (C–E) Flow cytometry assay was employed to detect the apoptosis and cell cycle distribution of transfected cells. (F and G) The migration and invasion abilities of transfected cells were detected by transwell assay. (H and I) The protein levels of E-cadherin, N-cadherin, and Vimentin were analyzed by western blot assay. (J–L) The levels of glucose uptake, lactate production, and ATP production were evaluated by corresponding kits. *P < 0.05
Rights and permissions
About this article
Cite this article
Huang, Y., Chen, Z., Zhou, X. et al. Circ_0000467 Exerts an Oncogenic Role in Colorectal Cancer via miR-330-5p-Dependent Regulation of TYRO3. Biochem Genet 60, 1488–1510 (2022). https://doi.org/10.1007/s10528-021-10171-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-021-10171-7