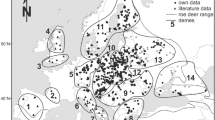
Partial sequences of the mitochondrial control region of northeastern China roe deer were analyzed to determine the degree of genetic diversity. Fourteen haplotypes were observed. The haplotype diversity was high (h = 0.872), nucleotide diversity was medium (p i = 0.0108), and the average Tamura–Nei nucleotide distance among them was 1.9%, indicating that genetic diversity of roe deer from northeastern China was relatively high and that the effective population size was large historically. To clarify the northeastern China roe deer's taxonomic status, these 14 haplotypes were compared with 31 haplotypes published in Genbank from Europe, Siberia, and Korea. The average genetic distance between haplogroups of northeastern China and European roe deer (5.8%) was more than twice that between northeastern China and Siberian roe deer (2.7%), indicating sufficient variation to consider roe deer of northeastern China and Siberia as a single species (Capreolus pygargus), distinct from European roe deer (Capreolus capreolus). This is the first presentation of mtDNA data for roe deer in northeastern China, which will be helpful in investigations of genetic diversity and clarifications of the taxonomic status of roe deer in the whole of China.



Similar content being viewed by others
REFERENCES
Bandelt, H. L., Forster, P., and Rohl, A. (1999). Median-joining networks for inferring intraspecific phylogenies. Mol. Biol. Evol. 16:37–48.
Blagojevic, J., and Vujosevic, M. (2004). B chromosomes and developmental homeostasis in the yellow-necked mouse, Apodemus flavicollis (Rodentia, Mammalia): Effects on nonmetric traits. Heredity 93:249–254.
Douzery, E., and Randi, E. (1997). The mitochondrial control region of cervidae: Evolutionary patterns and phylogenetic content. Mol. Biol. Evol. 14:1154–l166.
Fagundes, V., Camacho, J. P., and Yonenaga-Yassuda, Y. (2004). Are the dotlike chromosomes in Trinomys iheringi (Rodentia, Echimyidae) B chromosomes? Cytogenet. Genome Res. 106:159–164.
Fakler, P., and Schreiber, A. (1997). Allozyme heterozygosity in two isolated populations of roe deer (Capreolus capreolus) from the Netherlands. Neth. J. Zool. 47:1–8.
Graphodatsky, A. S. (1990). Karyotypical relationships between Cervidae. J. Zool. 69:101–114.
Groves, C. P., and Grubb P. (1987). Relationships of the living deer. In Wemmer, C. M. (ed.), Biology and Management of the Cervidae, Smithsonian Institution Press, Washington, DC, pp. 21–59.
Gustavsson, I., and Sundt, C. O. (1968). Karyotypes in five species of deer (Alces alces L., Capreolus capreolus L., Cervus elaphus L., Cervus nippon nippon Temm., and Dama dama L.). Hereditas 60:233–248.
Hartl, G. B., Markov, G., Rubin, A., Findo, S., Lang, G., and Willing, R. (1993). Allozyme diversity within and among populations of three ungulate species (Cervus elaphus, Capreolus capreolus, Sus scrofa) of Southeastern and Central Europe. Z. Säugetierkunde 58:352–361.
Hartl, G. B., and Reimoser, F. (1988). Biochemical variation in roe deer (Capreolus capreolus L.): Are r-strategists among deer genetically less variable than K-strategists? Heredity 60:221–227.
Hartl, G. B., Reimoser, F., Willing, R., and Köller, J. (1991). Genetic variability and differentiation in roe deer (Capreolus capreolus L.) of Central Europe. Genet. Sel. Evol. 23:281–299.
Herzog, S. (1988). The karyotype of the European roe deer (Capreolus capreolus L.). Z. Säugetierkunde 53:102–107.
Hewison, A. J. M., and Danilkin, A. (2001). Evidence for separate specific status of European (Capreolus capreolus) and Siberian (C. pygargus) roe deer. Mamm. Biol. 66:13–21.
Hewison, A. J. M. (1995). Isozyme variation in roe deer in relation to their population history in Britain. J. Zool. 235:279–288.
Jacob, B. B., Isaäc, J. N., Carla, Z., and Johannes, A. L. (1998). A satellite DNA element specific for roe deer (Capreolus capreolus). Chromosoma 107:1–5
Kartavtseva, I. V., and Roslik, G. V. (2004). A complex B chromosome system in the Korean field mouse, Apodemus peninsulae. Cytogenet. Genome Res. 106:271–278.
Koh, H. S., and Randi, E. (2001). Genetic distinction of roe deer (Capreolus pygargus Pallas) sampled in Korea. Mamm. Biol. 66:371–375.
Kumar, S., Tamura, K., Jakobsen, I. B., and Nei, M. (2001). MEGA2.1: Molecular evolutionary genetics analysis software, Arizona State University, Tempe.
Li, C. S., Ma, L. J., Zhou, H. M., and Yu, F. G. (2002). Biologic attributes of roe deer. Anim. Sci. Vet. Med. 19:54–57.
Lorenzini, R., Lovari, S., and Masseti, M. (2002). The rediscovery of the Italian roe deer: Genetic differentiation and management implications. Ital. J. Zool. 69:367–379.
Lorenzini, R., Patalano, M., Apollonio, M., and Lazzarone, V. (1993). Genetic variability of roe deer Capreolus capreolus in Italy: Electrophoretic survey on populations of different origin. Acta Theriol. 38(2):141–151.
Lorenzini, R., San Josè, C., Braza, F., and Aragón, S. (2003). Genetic differentiation and phylogeography of roe deer in Spain, as suggested by mitochondrial DNA and microanalysis. Ital. J. Zool. 70:89–99.
Ma, Y. Q. (1986). Mammal List in Heilongjiang Province, Heilongjiang Science and Technology Press, Harbin, China.
Nies, G., Zachos, F. E., and Hartl, G. B. (2005). The impact of female philopatry on population differentiation in the European roe deer (Capreolus capreolus) as revealed by mitochondrial DNA and allozymes. Mamm. Biol. 70:130–134.
Petrosian, V. G., Tokarskaia, O. N., Danilkin, A. A., and Ryskov, A. P. (2002). Quantitative analysis of genetic parameters in populations of European (Capreolus capreolus L.) and Siberian (Capreolus pygargus Pall.) roe deer with RAPD markers. Genetika 38:812–819.
Randi, E., Alves, P. C., Carranza, J., Milosevic-Zlatanovic, S., Sfougaris, A., and Mucci, N. (2004). Phylogeography of roe deer (Capreolus capreolus) populations: The effects of historical genetic subdivisions and recent nonequilibrium dynamics. Mol. Ecol. 13:3071–3083.
Randi, E., Lucchini, V., and Diong, C. H. (1996). Evolutionary genetics of the Suiformes as reconstructed using mtDNA sequencing. J. Mamm. Evol. 3:163–194.
Randi, E., Pierpaoli, M., and Danikin, A. (1998). Mitochondrial DNA polymorphism in populations of Siberian and European roe deer (Capreolus pygargus and C. capreolus). Heredity 80:429–437.
Rozas, J., and Rozas, R. (2003). DNASP: DNA polymorphism analysis by the coalescent and other methods. Bioinformatics 19:2496–2497.
Sheng, H. L. (1992). Chinese Deer, East China Normal University Press, Shanghai.
Shou, Z. H. (1962). List of Chinese Economic Animals: Mammals, Science Press, Beijing
Sokolov, V. E. (1978). Chromosome differences of two subspecies of roe deer Capreolus capreolus capreolus L. and Capreolus capreolus pygargus Pallas. J. Zool. 57:1109–1112.
Sokolov, V. E., and Gromov, V. S. (1990). The contemporary ideas on roe deer (Capreolus Gray, 1821) systematization: Morphological, ethological and hybridolohigical analysis. Mammalia 54:431–444.
Tamura, K., and Nei, M. (1993). Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Mol. Biol. Evol. 10:512–526.
Thompson, J. D., Gibson, T. J., Plewniak, F., Jeanmougin, F., and Higgins, D. G. (1997). The clustal × windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 24:4876–4882.
Tokarskaia, O. N., Efremova, D. A., Kan, N. G., Danilkin, A. A., Sempere, A., Petrosian, V. G., Semenova, S. K., and Ryskov, A. P. (2000). Variability of multilocus DNA markers in populations of the Siberian (Capreolus pygargus Pall.) and European (C. capreolus L.) roe deer. Genetika 36:1520–1530.
Vernesi, C., Pecchioli, E., Caramelli, D., Tiedemann, R., Randi, E., and Bertorelle, G. (2002). The genetic structure of natural and reintroduced roe deer (Capreolus capreolus) populations in the Alps and central Italy, with reference to the mitochondrial DNA phylogeography of Europe. Mol. Ecol. 11:1285–1297.
Vujosevic, M., and Blagojevic, J. (2004). B chromosomes in populations of mammals. Cytogenet. Genome Res. 106:247–256.
Wang, M., Lang, G., and Schreiber, A. (2002). Temporal shifts of DNA-microsatellite allele profiles in roe deer (Capreolus capreolus L.) within three decades. J. Zool. Syst. Evol. Res. 40:232–236.
Wang, M., and Schreiber, A. (2001). The impact of social structure and habitat fragmentation on the population genetics of roe deer (Capreolus capreolus L.) in Central Europe. Heredity 86:703–715.
Wang, Y. X. (2003). A Complete Checklist of Mammal Species and Subspecies in China: A Taxonomic and Geographic Reference, China Forestry Publishing House, Beijing.
Wang, Z. R., and Du, R. F. (1983). Karyotypes of Cervidae and their evolution. Acta Zool. Sinica 29:214–221.
Wilson, D. E., and Reeder D. M. (1993). Mammal Species of the World: A Taxonomic and Geographic Reference, 2nd ed., Smithsonian Institution Press, Washington, DC.
Zhang, M. H., Xiao, C. T., and Koh, H. S. (2005). Taxonomic status of roe deer in northeastern China based on mitochondrial DNA sequences. Acta Theriol. Sinica 25:14–19.
Zheng, Z. X. (1982). Taxonomy of Vertebrate Animals in China, Science Press, Beijing.
ACKNOWLEDGMENTS
This work was supported by the Rhinoceros and Tiger Conservation Fund, USA (98210-2-G191).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xiao, CT., Zhang, MH., Fu, Y. et al. Mitochondrial DNA Distinction of Northeastern China Roe Deer, Siberian Roe Deer, and European Roe Deer, to Clarify the Taxonomic Status of Northeastern China Roe Deer. Biochem Genet 45, 93–102 (2007). https://doi.org/10.1007/s10528-006-9052-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-006-9052-z