Abstract
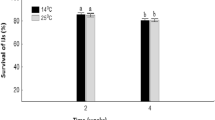
Large-scale use of entomopathogenic nematodes (EPNs) (Rhabditida: Steinernematidae and Heterorhabditidae) as biological control agents is impaired by their short shelf life. Three local South African EPN species, including Steinernema yirgalemense, S. jeffreyense and Heterorhabditis bacteriophora, were investigated for their role in formulations. Encapsulation of the infective juveniles (IJs) in alginate beads and diatomaceous earth (DE) was investigated. Survival of the IJs in the formulations was determined at 6 °C, 14 °C and 25 °C for four weeks. Of the IJs, 10-20% were observed to escape from the beads, depending on temperature, and readily survived the encapsulation process. DE did not cause the desiccation of the nematodes, with there still being a lower mortality rate by the 4th week of the study. In both formulations, the survival and virulence rates differed significantly at 6 °C, as compared to at 14 °C and 25 °C, with a drastic decrease over time for S. yirgalemense. The EPN species revealed poor survival and loss of virulence at low temperatures in both formulations. The alginate beads successfully retained most of the IJs and can be stored for a longer time.




Similar content being viewed by others
References
Addis T, Mijušković N, Ehlers R-U, Strauch O (2016) Life history traits, liquid culture production and storage temperatures of Steinernema yirgalemense. Nematology 18:367–376
Akhurst R, Smith K (2002) Regulation and safety. In: Gaugler R (ed) Entomopathogenic nematology. CABI Publishing, Wallingford, pp 311–332
Akhurst RJ, Mourant RG, Baud L, Boemare NE (1996) Phenotypic and DNA relatedness between nematode symbionts and clinical strains of the genus Photorhabdus (Enterobacteriaceae). Int J Syst Bacteriol 46:1034–1041
Boemare NE, Akhurst RJ, Mourant RG (1993) DNA relatedness between Xenorhabdus spp. (Enterobacteriaceae), symbiotic bacteria of entomopathogenic nematodes, and a proposal to transfer Xenorhabdus luminescens to a new genus, Photorhabdus gen. nov. Int J Syst Bacteriol 43:249–255
Buchholz S, Merkel K, Spiewok S, Pettis JS, Duncan M, Spooner-Hart R, Ulrichs C, Ritter W, Neumann P (2009) Alternative control of Aethina tumida Murray (Coleoptera: Nitidulidae) with lime and diatomaceous earth. Apidologie 40:535–548
Chen S, Glazer I (2005) A novel method for long-term storage of the entomopathogenic nematode Steinernema feltiae at room temperature. Biol Control 32:104–110
Ehlers R-U (2007) Entomopathogenic nematodes: from science to commercial use. In: Vincent C, Goettel MS, Lazarovits G (eds) Biological control: a global perspective: case studies from around the world. CABI Publishing, Wallingford, pp 136–151
Golob P (1997) Current status and future perspectives for inert dusts for control of stored product insects. J Stored Prod Res 33:69–79
Grewal PS (2000a) Enhanced ambient storage stability of an entomopathogenic nematode through anhydrobiosis. Pest Manag Sci 56:401–406
Grewal PS (2000b) Anhydrobiotic potential and long-term storage of entomopathogenic nematodes (Rhabditida: Steinernematidae). Int J Parasitol 30:995–1000
Grewal PS (2002) Formulation and application technology. In: Gaugler R (ed) Entomopathogenic nematology. CABI Publishing, Wallingford, pp 265–287
Grewal PS, Jagdale GB (2002) Enhanced trehalose accumulation and desiccation survival of entomopathogenic nematodes through cold preacclimation. Biocontrol Sci Tech 12:533–545
Grewal PS, Peters A (2005) Formulation and quality. In: Grewal PS, Ehlers R-U, Shapiro-Ilan DI (eds) Nematodes as biocontrol agents. CABI Publishing, Wallingford, pp 79–89
Hatting JL, Moore SD, Malan AP (2019) Microbial control of phytophagous invertebrate pests in South Africa: current status and future prospects. J Invertebr Pathol (in press). https://doi.org/10.1016/j.jip.2018.02.004
Hiltpold I, Hibbard BE, French BW, Turlings TCJ (2012) Capsules containing entomopathogenic nematodes as a Trojan horse approach to control the western corn rootworm. Plant Soil 358:11–25
Kagimu N, Ferreira T, Malan AP (2017) The attributes of survival in the formulation of entomopathogenic nematodes utilised as insect biocontrol agents. African Entomology 25:275–291
Kaya HK, Nelsen CE (1985) Encapsulation of steinernematid and heterorhabditid nematodes with calcium alginate: a new approach for insect control and other applications. Environ Entomol 14:572–574
Kaya HK, Stock SP (1997) Techniques in insect nematology. In: Lacey L (ed) Manual of techniques in insect pathology. Academic Press, San Diego, pp 281–324
Kaya HK, Mannion CM, Burlando TM, Nelsen CE (1987) Escape of Steinernema feltiae from alginate capsules containing tomato seeds. Journal of Nematology 19:287–291
Kim J, Jaffuel G, Turlings TCJ (2015) Enhanced alginate capsule properties as a formulation of entomopathogenic nematodes. BioControl 60:527–535
Malan AP, Hatting JL (2015) Entomopathogenic nematode exploitation: case studies in laboratory and field applications from South Africa. In: Campos-Herrera R (ed) Nematode pathogenesis of insects and other pests. Springer International Publishing, Dordrecht, pp 477–508
Malan AP, Knoetze R, Tiedt LR (2016) Steinernema jeffreyense n. sp. (Rhabditida: Steinernematidae), a new entomopathogenic nematode from South Africa. J Helminthol 90:262–278
Matadamas-Ortiz PT, Ruiz-Vega J, Vazquez-Feijoo JA, Cruz-Martinez H, Cortes-Martinez CI (2014) Mechanical production of pellets for the application of entomopathogenic nematodes: factors that determine survival time of Steinernema glaseri. Biocontrol Sci Technol 24:145–157
Navon A, Keren S, Salame L, Glazer I (1998) An edible-to-insects calcium alginate gel as a carrier for entomopathogenic nematodes. Biocontrol Sci Technol 8:429–437
Navon A, Nagalakshmi VK, Levski S, Salame L, Glazer I (2002) Effectiveness of entomopathogenic nematodes in an alginate gel formulation against lepidopterous pests. Biocontrol Sci Technol 12:737–746
Nguyen, Tesfamariam, Gozel, Gaugler, Adams (2004) Steinernema yirgalemense n. sp. (Rhabditida: Steinernematidae) from Ethiopia. Nematology 6:839–856
Odendaal D, Addison MF, Malan AP (2015) Control of codling moth (Cydia pomonella) (Lepidoptera: Tortricidae) in South Africa with special emphasis on using entomopathogenic nematodes. African Entomol 23:259–274
Odendaal D, Addison MF, Malan AP (2016) Entomopathogenic nematodes for the control of the codling moth (Cydia pomonella L.) in field and laboratory trials. J Helminthol 90:615–623
Patel AV, Vorlop K-D (1994) Entrapment of biological control agents applied to entomopathogenic nematodes. Biotechnol Tech 8:569–574
Peters A (2004) Protocol quality assessment of entomopathogenic nematodes. COST Action 850 meeting programme and mushroom protocols. Wädenswil, Switzerland, pp 4–6
Poinar GO (1975) Description and biology of a new insect parasite rhabditoid, Heterorhabditis bacteriophora n. gen. n. sp. (Rhabditida; Heterorhabditidae. fam.). Nematologica 21:463–470
Silver S, Dunlop D, Grove D (1995) WIPO Patent No. WO 95/0577. World Intellectual Property Organization, Geneva
Stuart RJ, Barbercheck ME, Grewal PS (2015) Entomopathogenic nematodes in the soil environment: distributions, interactions and the influence of biotic and abiotic factors. In: Campos-Herrera R (ed) Nematode pathogenesis of insects and other pests. Springer International Publishing, Dordrecht, pp 97–137
Surrey MR, Wharton DA (1995) Desiccation survival of the infective larvae of the insect parasitic nematode, Heterorhabditis zealandica Poinar. Int J Parasitol 25:749–752
Thomas GM, Poinar GO (1983) Amended description of the genus Xenorhabdus Thomas and Poinar. Int J Syst Bacteriol 33:878–879
Travassos L (1927) Sobre o Genera Oxystomatium. Boletim Biologico, Sao Paula. Brasil 5:20–21
van Zyl C, Malan AP (2015) Cost-effective culturing of Galleria mellonella and Tenebrio molitor and entomopathogenic nematode production in various hosts. African Entomol 23:361–375
Vemmer M, Patel AV (2013) Review of encapsulation methods suitable for microbial biological control agents. Biol Control 67:380–389
Wakil W, Riasat TM, Ghazanfar U, Kwon YJ, Shaheen FA (2011) Aptness of Beauveria bassiana and enhanced diatomaceous earth (DEBBM) for control of Rhyzopertha dominica F. Entomol Res 41:233–241
Womersley CZ (1990) Dehydration survival and anhydrobiotic potential. In: Gaugler R, Kaya HK (eds) Entomopathogenic nematodes in biological control. CRC Press Inc, Boca Raton, pp 117–137
Ziaee M, Atapour M, Marouf A (2016) Insecticidal efficacy of Iranian diatomaceous earths on adults of Oryzaephilus surinamensis. J Agric Sci Technol 18:361–370
Acknowledgements
The authors wish to thank D.G. Nel (Centre for Statistical Consultation, Stellenbosch University) for assistance with the statistical analyses. The financial support of NemaBio (Pty) (Ltd) and National Research Foundation (THRIP-TP14062571871) are greatly appreciated.
Funding
This study was funded by NemaBio (Pty) (Ltd) and National Research Foundation (Grant No. THRIP-TP14062571871).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human and animal participants
The research does not involve human participant or animals.
Additional information
Handling Editor: Ralf Ehlers
Rights and permissions
About this article
Cite this article
Kagimu, N., Malan, A.P. Formulation of South African entomopathogenic nematodes using alginate beads and diatomaceous earth. BioControl 64, 413–422 (2019). https://doi.org/10.1007/s10526-019-09945-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10526-019-09945-1