Abstract
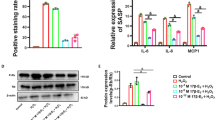
This study investigates the influence of 17beta-estradiol (E2) on hydrogen peroxide (H2O2)-induced human vascular endothelial cell (HUVEC) senescence. HUVECs were divided into four groups, namely control group, H2O2 stimulation group, E2 intervention group and ICI182780 (ICI) intervention group. The aging-related β-galactosidase activities, cytochrome C oxidase activities, intracellular ATP levels, intracellular reactive oxygen species (ROS) levels and phosphorylated Rb protein expressions were mainly observed. Of which, senescence-associated β-galactosidase activities were detected using immunohistochemical staining, cytochrome C oxidase activities and intracellular ATP levels were detected using commercial kits, ROS levels were detected by fluorescence microscopy and fluorescence microplate reader, immunoblotting was used to quantitatively detect the expressions of phosphorylated Rb proteins. After continuous treatment of H2O2, the senescent phenotypes appeared in the HUVECs. The percentage of positive SA-βgal staining cells and the phosphorylated Rb expressions were significantly increased; intracellular ROS levels, cytochrome C oxidase activities and intracellular ATP levels were elevated. Compared with the H2O2 stimulation group, E2 intervention significantly decreased the positive rate of SA-β-gal staining, the phosphorylated Rb protein levels, the intracellular ROS levels, cytochrome C oxidase activities and intracellular ATP levels. Pretreatment of estrogen receptor blocker ICI182780 weakened the role of E2. These results indicated that H2O2 could induce HUVEC senescence; 17beta-E2 might relieve H2O2-induced mitochondrial damage through estrogen receptor and delay the vascular endothelial cell senescence.




Similar content being viewed by others
References
Atamna H, Liu J, Ames BN (2001) Heme deficiency selectively interrupts assembly of mitochondrial complex IV in human fibroblasts: revelance to aging. J Biol Chem 276:48410–48416. doi:10.1074/jbc.M108362200
Attardi LD, Sage J (2013) RB goes mitochondrial. Genes Dev 27:975–979. doi:10.1101/gad.219451.113
Balaban RS, Nemoto S, Finkel T (2005) Mitochondria, oxidants, and aging. Cell 120:483–495. doi:10.1016/j.cell.2005.02.001
Bianco S, Lanvin O, Tribollet V, Macari C, North S, Vanacker JM (2009) Modulating estrogen receptor-related receptor-alpha activity inhibits cell proliferation. J Biol Chem 284:23286–23292. doi:10.1074/jbc.M109.028191
Blacher J, Agnoletti D, Protogerou AD, Iaria P, Czernichow S, Zhang Y, Safar ME (2012) Aortic stiffness, inflammation, denutrition and prognosis in the oldest people. J Hum Hypertens 26:518–524. doi:10.1038/jhh.2011.73
Chen JH, Stoeber K, Kingsbury S, Ozanne SE, Williams GH, Hales CN (2004) Loss of proliferative capacity and induction of senescence in oxidatively stressed human fibroblasts. J Biol Chem 279:49439–49446. doi:10.1074/jbc.M409153200
Cobrinik D (2005) Pocket proteins and cell cycle control. Oncogene 24:2796–2809. doi:10.1038/sj.onc.1208619
Denti L (2010) The hormone replacement therapy (HRT) of menopause: focus on cardiovascular implications. Acta Bio-med Atenei Parm 81(Suppl 1):73–76
Farsetti A, Grasselli A, Bacchetti S, Gaetano C, Capogrossi MC (2009) The telomerase tale in vascular aging: regulation by estrogens and nitric oxide signaling. J Appl Physiol 106:333–337. doi:10.1152/japplphysiol.91360.2008
Fauser BC et al (2011) Sex steroid hormones and reproductive disorders: impact on women’s health. Reprod Sci 18:702–712. doi:10.1177/1933719111405068
Finkel T, Holbrook NJ (2000) Oxidants, oxidative stress and the biology of ageing. Nature 408:239–247. doi:10.1038/35041687
Gemma C, Vila J, Bachstetter A, Bickford PC (2007) Oxidative stress and the aging brain: from theory to prevention. In: Riddle DR (ed) Brain aging: models, methods, and mechanisms. Frontiers in Neuroscience, Boca Raton
Hara E, Smith R, Parry D, Tahara H, Stone S, Peters G (1996) Regulation of p16CDKN2 expression and its implications for cell immortalization and senescence. Mol Cell Biol 16:859–867
James AM, Murphy MP (2002) How mitochondrial damage affects cell function. J Biomed Sci 9:475–487. doi:10.1007/BF02254975
Kokoszka JE, Coskun P, Esposito LA, Wallace DC (2001) Increased mitochondrial oxidative stress in the Sod2 (±) mouse results in the age-related decline of mitochondrial function culminating in increased apoptosis. Proc Natl Acad Sci USA 98:2278–2283. doi:10.1073/pnas.051627098
Kujoth GC et al (2005) Mitochondrial DNA mutations, oxidative stress, and apoptosis in mammalian aging. Science 309:481–484. doi:10.1126/science.1112125
Lakatta EG, Levy D (2003) Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises: Part I: aging arteries: a “set up” for vascular disease. Circulation 107:139–146
Lee BY et al (2006) Senescence-associated beta-galactosidase is lysosomal beta-galactosidase. Aging Cell 5:187–195. doi:10.1111/j.1474-9726.2006.00199.x
Liu J, Wu S, Wei H, Zhou K, Ruan Y, Lai W (2002) Effects of sex hormones and their balance on the proliferation of rat vascular endothelial cells. Horm Res 58:16–20. 10.1159/000063211
Minamino T, Miyauchi H, Yoshida T, Tateno K, Kunieda T, Komuro I (2004) Vascular cell senescence and vascular aging. J Mol Cell Cardiol 36:175–183. doi:10.1016/j.yjmcc.2003.11.010
Moiseeva O, Bourdeau V, Roux A, Deschenes-Simard X, Ferbeyre G (2009) Mitochondrial dysfunction contributes to oncogene-induced senescence. Mol Cell Biol 29:4495–4507. doi:10.1128/MCB.01868-08
Murphy E (2011) Estrogen signaling and cardiovascular disease. Circ Res 109:687–696. doi:10.1161/CIRCRESAHA.110.236687
Nakaya M, Tachibana H, Yamada K (2005) Isoflavone genistein and daidzein up-regulate LPS-induced inducible nitric oxide synthase activity through estrogen receptor pathway in RAW264.7 cells. Biochem Pharmacol 71:108–114. doi:10.1016/j.bcp.2005.10.002
Payne JA, Reckelhoff JF, Khalil RA (2003) Role of oxidative stress in age-related reduction of NO-cGMP-mediated vascular relaxation in SHR. Am J Physiol Regul Integr Comp Physiol 285:R542–R551. doi:10.1152/ajpregu.00056.2003
Pedram A, Razandi M, Evinger AJ, Lee E, Levin ER (2009) Estrogen inhibits ATR signaling to cell cycle checkpoints and DNA repair. Mol Biol Cell 20:3374–3389. doi:10.1091/mbc.E09-01-0085
Pitkin J (2012) Alternative and complementary therapies for the menopause. Menopause Int 18:20–27. doi:10.1258/mi.2012.012001
Psarra AM, Solakidi S, Sekeris CE (2006) The mitochondrion as a primary site of action of steroid and thyroid hormones: presence and action of steroid and thyroid hormone receptors in mitochondria of animal cells. Mol Cell Endocrinol 246:21–33. doi:10.1016/j.mce.2005.11.025
Sena LA, Chandel NS (2012) Physiological roles of mitochondrial reactive oxygen species. Mol Cell 48:158–167. doi:10.1016/j.molcel.2012.09.025
Serra V, von Zglinicki T, Lorenz M, Saretzki G (2003) Extracellular superoxide dismutase is a major antioxidant in human fibroblasts and slows telomere shortening. J Biol Chem 278:6824–6830. doi:10.1074/jbc.M207939200
Shimokawa H, Takeshita A (1995) Endothelium-dependent regulation of the cardiovascular system. Intern Med 34:939–946
Stice JP, Lee JS, Pechenino AS, Knowlton AA (2009) Estrogen, aging and the cardiovascular system. Future Cardiol 5:93–103. doi:10.2217/14796678.5.1.93
Toda N (2012) Age-related changes in endothelial function and blood flow regulation. Pharmacol Ther 133:159–176. doi:10.1016/j.pharmthera.2011.10.004
Velarde MC (2013) Pleiotropic actions of estrogen: a mitochondrial matter. Physiol Genomics 45:106–109. doi:10.1152/physiolgenomics.00155.2012
Wang JC, Bennett M (2012) Aging and atherosclerosis: mechanisms, functional consequences, and potential therapeutics for cellular senescence. Circ Res 111:245–259. doi:10.1161/CIRCRESAHA.111.261388
Wu S, Ruan Y, Zhu X, Lai W (2000) Estrogen receptors and the activity of nitric oxide synthase in the artery of female rats receiving hormone replacement therapy. Horm Res 53:144–147.
Wu S, Ruan Y, Yin M, Lai W (2007) Research on the age-related changes in the nitric oxide pathway in the arteries of rats and the intervention effect of dehydroepiandrosterone. Gerontology 53:234–237. doi:10.1159/000100961
Yildiz O (2007) Vascular smooth muscle and endothelial functions in aging. Ann NY Acad Sci 1100:353–360. doi:10.1196/annals.1395.038
Zhao Z et al (2009) p21-Activated kinase mediates rapid estradiol-negative feedback actions in the reproductive axis. Proc Natl Acad Sci USA 106:7221–7226. doi:10.1073/pnas.0812597106
Acknowledgments
We declare no conflict of interest. This work was supported by the Nature Science Foundation of Guangdong Province (S2013010011949).
Author information
Authors and Affiliations
Corresponding author
Additional information
Yunjun Ruan and Saizhu Wu have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Ruan, Y., Wu, S., Zhang, L. et al. Retarding the senescence of human vascular endothelial cells induced by hydrogen peroxide: effects of 17beta-estradiol (E2) mediated mitochondria protection. Biogerontology 15, 367–375 (2014). https://doi.org/10.1007/s10522-014-9507-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-014-9507-2