Abstract
In the present study, we investigated the signaling pathways implicated in the induction of apoptosis by two modified nucleosides, 5-phenylselenyl-methyl-2′-deoxyuridine (PhSe-T) and 5-methylselenyl-methyl-2′-deoxyuridine (MeSe-T), using human cancer cell lines. The induction of apoptosis was associated with proteolytic activation of caspase-3 and -9, PARP cleavage, and decreased levels of IAP family members, including c-IAP-1 and c-IAP-2, but had no effect on XIAP and survivin. PhSe-T and MeSe-T also enhanced the activities of caspase-2 and -8, Bid cleavage, and the conformational activation of Bax. Additionally, nucleoside derivative-induced apoptosis was inhibited by the selective inhibitors of caspase-2, -3, -8, and -9 and also by si-RNAs against caspase-2, -3, -8, and -9; however, inhibition of caspase-2 and -3 was more effective at preventing apoptosis than inhibition of caspase-8 and -9. Moreover, the inhibition of caspase-2 activation by the pharmacological inhibitor z-VDVAD-fmk or by the knockdown of protein expression using siRNA suppressed nucleoside derivative-induced caspase-3 activation, but not vice versa. PhSe-T and MeSe-T also induced a Δψm loss via a CsA-insensitive mechanism, ROS production, and DNA damage, including strand breaks. Moreover, ROS scavengers such as NAC, tiron, and quercetin inhibited nucleoside derivative-induced ROS generation and apoptosis by blocking the sequential activation of caspase-2 and -3, indicating the role of ROS in caspase-2-mediated apoptosis. Taken together, these results indicate that caspase-2 acts upstream of caspase-3 and that caspase-2 functions in response to DNA damage in both PhSe-T- and MeSe-T-induced apoptosis. Our results also suggest that ROS are critical regulators of the sequential activation of caspase-2 and -3 in nucleoside derivative-treated cancer cells.








Similar content being viewed by others
Abbreviations
- ANOVA:
-
Analysis of variance
- CsA:
-
Cyclosporin A
- DAPI:
-
4′-6-Diamidino-2-phenylindole
- DiOC6:
-
3,3′-Dihexyloxacarbocyanine
- DSBs:
-
Double-strand breaks
- FACS:
-
Fluorescence-activated cell sorter
- IAP:
-
Inhibitors of apoptosis proteins
- LDH:
-
Lactate dehydrogenase
- MeSe-T:
-
5-Methylselenyl-methyl-2′-deoxyuridine
- MMP:
-
Mitochondrial membrane potential
- NAC:
-
N-acetylcysteine
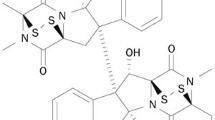
- PhSe-T:
-
5-Phenylselenyl-methyl-2′-deoxyuridine
- PI:
-
Propidium iodide
- pNA:
-
p-Nitroanilide
- ROS:
-
Reactive oxygen species
- SCGE:
-
Single-cell gel electrophoresis
- SSBs:
-
Single-strand breaks
- tiron:
-
4,5-Dihydroxy-1,3-benzenedisulfonic acid
- z-DEVD-fmk:
-
N-benzyloxycarbonyl-Asp-Glu-Val-Asp-fmk
- z-IETD-fmk:
-
N-benzyloxycarbonyl-Ile-Glu-Thr-Asp-fmk
- z-LEHD-fmk:
-
N-benzyloxycarbonyl-Leu-Glu-His-Asp-fmk
- z-VDVAD-fmk:
-
N-benzyloxycarbonyl-Val-Asp-Val-Ala-Asp-fmk
References
Zhu C, Johansson M, Karlsson A (2000) Incorporation of nucleoside analogs into nuclear or mitochondrial DNA is determined by the intracellular phosphorylation site. J Biol Chem 275:26727–26731
Zhivotovsky B, Orrenius S (2005) Caspase-2 function in response to DNA damage. Biochem Biophys Res Commun 331:859–867
Troy CM, Shelanski ML (2003) Caspase-2 redux. Cell Death Differ 10:101–107
Shi M, Vivian CJ, Lee KJ, Ge C, Morotomi-Yano K, Manzl C et al (2009) DNA-PKcs-PIDDosome: a nuclear caspase-2-activating complex with role in G2/M checkpoint maintenance. Cell 136:508–520
Lin CF, Chen CL, Chang WT, Jan MS, Hsu LJ, Wu RH et al (2004) Sequential caspase-2 and caspase-8 activation upstream of mitochondria during ceramide and etoposide-induced apoptosis. J Biol Chem 279:40755–40761
Kim BM, Hong SH (2011) Sequential caspase-2 and caspase-8 activation is essential for saikosaponin a-induced apoptosis of human colon carcinoma cell lines. Apoptosis 16:184–197
Burney S, Niles JC, Dedon PC, Tannenbaum SR (1999) DNA damage in deoxynucleosides and oligonucleotides treated with peroxynitrite. Chem Res Toxicol 12:513–520
Prasad V, Chandele A, Jagtap JC, Sudheer Kumar P, Shastry P (2006) ROS-triggered caspase-2 activation and feedback amplification loop in beta-carotene-induced apoptosis. Free Radic Biol Med 41:431–442
Madesh M, Zong WX, Hawkins BJ, Ramasamy S, Venkatachalam T, Mukhopadhyay P et al (2009) Execution of superoxide-induced cell death by the proapoptotic Bcl-2-related proteins Bid and Bak. Mol Cell Biol 29:3099–3112
Hong IS, Greenberg MM (2004) Mild generation of 5-(2′-deoxyuridinyl)methyl radical from a phenyl selenide precursor. Org Lett 6:5011–5013
Hong IS, Greenberg MM (2005) Efficient DNA interstrand cross-link formation from a nucleotide radical. J Am Chem Soc 127:3692–3693
Hong IS, Greenberg MM (2005) DNA interstrand cross-link formation initiated by reaction between singlet oxygen and a modified nucleotide. J Am Chem Soc 127:10510–10511
Hong IS, Ding H, Greenberg MM (2006) Oxygen independent DNA interstrand cross-link formation by a nucleotide radical. J Am Chem Soc 128:485–491
Peng X, Hong IS, Li H, Seidman MM, Greenberg MM (2008) Interstrand cross-link formation in duplex and triplex DNA by modified pyrimidines. J Am Chem Soc 130:10299–10306
Rode AB, Kim BM, Park SH, Hong IS, Hong SH (2011) Potent radiosensitizing agents: 5-methylselenyl- and 5-phenylselenyl-methyl-2′-deoxyuridine. Bioorg Med Chem Lett 21:1151–1154
Yamaguchi H, Wang HG (2001) The protein kinase PKB/Akt regulates cell survival and apoptosis by inhibiting Bax conformational change. Oncogene 20:7779–7786
Gao Z, Tian Y, Wang J, Yin Q, Wu H, Li YM et al (2007) A dimeric Smac/diablo peptide directly relieves caspase-3 inhibition by XIAP. Dynamic and cooperative regulation of XIAP by Smac/Diablo. J Biol Chem 282:30718–30727
Mehmet H (2000) Caspases find a new place to hide. Nature 403:29–30
Li H, Zhu H, Xu CJ, Yuan J (1998) Cleavage of BID by caspase 8 mediates the mitochondrial damage in the Fas pathway of apoptosis. Cell 94:491–501
Rogakou EP, Pilch DR, Orr AH, Ivanova VS, Bonner WM (1998) DNA double-stranded breaks induce histone H2AX phosphorylation on serine 139. J Biol Chem 273:5858–5868
Burma S, Chen BP, Murphy M, Kurimasa A, Chen DJ (2001) ATM phosphorylates histone H2AX in response to DNA double-strand breaks. J Biol Chem 276:42462–42467
Sedelnikova OA, Pilch DR, Redon C, Bonner WM (2003) Histone H2AX in DNA damage and repair. Cancer Biol Ther 2:233–235
Rogakou EP, Boon C, Redon C, Bonner WM (1999) Megabase chromatin domains involved in DNA double-strand breaks in vivo. J Cell Biol 146:905–916
Zamzami N, Marchetti P, Castedo M, Zanin C, Vayssière JL, Petit PX et al (1995) Reduction in mitochondrial potential constitutes an early irreversible step of programmed lymphocyte death in vivo. J Exp Med 181:1661–1672
Zhang D, Berry MD, Paterson IA, Boulton AA (1999) Loss of mitochondrial membrane potential is dependent on the apoptotic program activated: prevention by R-2HMP. J Neurosci Res 58:284–292
Düssmann H, Rehm M, Kögel D, Prehn JH (2003) Outer mitochondrial membrane permeabilization during apoptosis triggers caspase-independent mitochondrial and caspase-dependent plasma membrane potential depolarization: a single-cell analysis. J Cell Sci 116:525–536
Guo Y, Srinivasula SM, Druilhe A, Fernandes-Alnemri T, Alnemri ES (2002) Caspase-2 induces apoptosis by releasing proapoptotic proteins from mitochondria. J Biol Chem 277:13430–13437
Paroni G, Henderson C, Schneider C, Brancolini C (2002) Caspase-2 can trigger cytochrome C release and apoptosis from the nucleus. J Biol Chem 277:15147–15161
Lassus P, Opitz-Araya X, Lazebnik Y (2002) Requirement for caspase-2 in stress-induced apoptosis before mitochondrial permeabilization. Science 297:1352–1354
Demple B, Harrison L (1994) Repair of oxidative damage to DNA: enzymology and biology. Annu Rev Biochem 63:915–948
Zha S, Sekiguchi J, Brush JW, Bassing CH, Alt FW (2008) Complementary functions of ATM and H2AX in development and suppression of genomic instability. Proc Natl Acad Sci USA 105:9302–9306
Dickey JS, Redon CE, Nakamura AJ, Baird BJ, Sedelnikova OA, Bonner WM (2009) H2AX: functional roles and potential applications. Chromosoma 118:683–692
Kruidering M, Evan GI (2000) Caspase-8 in apoptosis: the beginning of “the end”? IUBMB Life 50:85–90
Luo X, Budihardjo I, Zou H, Slaughter C, Wang X (1998) Bid, a Bcl2 interacting protein, mediates cytochrome c release from mitochondria in response to activation of cell surface death receptors. Cell 94:481–490
Desagher S, Osen-Sand A, Nichols A, Eskes R, Montessuit S, Lauper S et al (1999) Bid-induced conformational change of Bax is responsible for mitochondrial cytochrome c release during apoptosis. J Cell Biol 144:891–901
Eskes R, Desagher S, Antonsson B, Martinou JC (2000) Bid induces the oligomerization and insertion of Bax into the outer mitochondrial membrane. Mol Cell Biol 20:929–935
Li P, Nijhawan D, Budihardjo I, Srinivasula SM, Ahmad M, Alnemri ES et al (1997) Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 91:479–489
Jiang X, Wang X (2000) Cytochrome c promotes caspase-9 activation by inducing nucleotide binding to Apaf-1. J Biol Chem 275:31199–31203
Hsu TC, Wu WJ, Chen MC, Tsay GJ (2004) Human parvovirus B19 non-structural protein (NS1) induces apoptosis through mitochondria cell death pathway in COS-7 cells. Scand J Infect Dis 36:570–577
Poole BD, Zhou J, Grote A, Schiffenbauer A, Naides SJ (2006) Apoptosis of liver-derived cells induced by parvovirus B19 nonstructural protein. J Virol 80:4114–4121
Olsson M, Vakifahmetoglu H, Abruzzo PM, Högstrand K, Grandien A, Zhivotovsky B (2009) DISC-mediated activation of caspase-2 in DNA damage-induced apoptosis. Oncogene 28:1949–1959
Robertson JD, Enoksson M, Suomela M, Zhivotovsky B, Orrenius S (2002) Caspase-2 acts upstream of mitochondria to promote cytochrome c release during etoposide-induced apoptosis. J Biol Chem 277:29803–29809
Panaretakis T, Laane E, Pokrovskaja K, Björklund AC, Moustakas A, Zhivotovsky B et al (2005) Doxorubicin requires the sequential activation of caspase-2, protein kinase Cdelta, and c-Jun NH2-terminal kinase to induce apoptosis. Mol Biol Cell 16:3821–3831
Arnaudeau C, Lundin C, Helleday T (2001) DNA double-strand breaks associated with replication forks are predominantly repaired by homologous recombination involving an exchange mechanism in mammalian cells. J Mol Biol 307:1235–1245
Roos WP, Kaina B (2006) DNA damage-induced cell death by apoptosis. Trends Mol Med 12:440–450
Hsieh YH, Su IJ, Wang HC, Chang WW, Lei HY, Lai MD et al (2004) Pre-S mutant surface antigens in chronic hepatitis B virus infection induce oxidative stress and DNA damage. Carcinogenesis 25:2023–2032
Mori K (2000) Tripartite management of unfolded proteins in the endoplasmic reticulum. Cell 101:451–454
Harding HP, Zhang Y, Bertolotti A, Zeng H, Ron D (2000) Perk is essential for translational regulation and cell survival during the unfolded protein response. Mol Cell 5:897–904
Oyadomari S, Mori M (2004) Roles of CHOP/GADD153 in endoplasmic reticulum stress. Cell Death Differ 11:381–389
Szegezdi E, Logue SE, Gorman AM, Samali A (2006) Mediators of endoplasmic reticulum stress-induced apoptosis. EMBO Rep 7:880–885
Hitomi J, Katayama T, Eguchi Y, Kudo T, Taniguchi M, Koyama Y (2004) Involvement of caspase-4 in endoplasmic reticulum stress-induced apoptosis and Abeta-induced cell death. J Cell Biol 165:347–356
Kim SJ, Zhang Z, Hitomi E, Lee YC, Mukherjee AB (2006) Endoplasmic reticulum stress-induced caspase-4 activation mediates apoptosis and neurodegeneration in INCL. Hum Mol Genet 15:1826–1834
Krumschnabel G, Sohm B, Bock F, Manzl C, Villunger A (2009) The enigma of caspase-2: the laymen’s view. Cell Death Differ 16:195–207
Acknowledgment
This study was supported by the National Nuclear R&D Program of the Ministry of Education, Science, and Technology (MEST) of the Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, B.M., Rode, A.B., Han, E.J. et al. 5-Phenylselenyl- and 5-methylselenyl-methyl-2′-deoxyuridine induce oxidative stress, DNA damage, and caspase-2-dependent apoptosis in cancer cells. Apoptosis 17, 200–216 (2012). https://doi.org/10.1007/s10495-011-0665-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-011-0665-2